Table 2. Recommended Empiric Regimens to Reduce Fluoroquinolone
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Efficacy of Fosfomycin Trometamol and Cefuroxime Axetil in the Treatment of Kashmir, India Urinary Tract Infections During Pregnancy
International Journal of Surgery Science 2019; 3(2): 33-35 E-ISSN: 2616-3470 P-ISSN: 2616-3462 © Surgery Science Efficacy of fosfomycin trometamol and cefuroxime axetil www.surgeryscience.com 2019; 3(2): 33-35 in the treatment of UTI during pregnancy: A Received: 18-11-2018 Accepted: 22-12-2018 comparative study Dr. Robindera Kour In Charge Consultant Surgery, Dr. Robindera Kour, Dr. Gurpreet Kour, Dr. Iqbal Singh, Dr. KK Gupta Govt. Hospital Sarwal, Jammu, and Dr. Rajiv Sharma Jammu and Kashmir, India Dr. Gurpreet Kour DOI: https://doi.org/10.33545/surgery.2019.v3.i2a.373 Medical Officer, Acharya Shri Chander College of Medical Abstract Sciences, Jammu, Jammu and Objective: To compare the efficacy of fosfomycin trometamol and cefuroxime axetil in the treatment of Kashmir, India urinary tract infections during pregnancy. Methods: Prospective clinical study was conducted among pregnant women who were followed as part of Dr. Iqbal Singh routine prenatal care at Government Hospital Sarwal, Jammu, India with complaints of lower UTI were Assistant Professor, Public Health assessed. 60 patients were enrolled, divided into 2 equal groups for treatment with single-dose fosfomycin Dentistry, Indira Gandhi Government Dental College, trometamol and 5-day courses of cefuroxime axetil. Follow-up was carried out after 5 weeks. Jammu, Jammu and Kashmir, Results: The treatment groups did not differ significantly in terms of demographics, clinical compliance India rate and adverse effects. But the patients who were taking 5-day courses of cefuroxime axetil showed statistically significant higher cure rate as well as lower recurrence rate as compared to fosfomycin Dr. -

Severe Sepsis and Septic Shock Antibiotic Guide
Stanford Health Issue Date: 05/2017 Stanford Antimicrobial Safety and Sustainability Program Severe Sepsis and Septic Shock Antibiotic Guide Table 1: Antibiotic selection options for healthcare associated and/or immunocompromised patients • Healthcare associated: intravenous therapy, wound care, or intravenous chemotherapy within the prior 30 days, residence in a nursing home or other long-term care facility, hospitalization in an acute care hospital for two or more days within the prior 90 days, attendance at a hospital or hemodialysis clinic within the prior 30 days • Immunocompromised: Receiving chemotherapy, known systemic cancer not in remission, ANC <500, severe cell-mediated immune deficiency Table 2: Antibiotic selection options for community acquired, immunocompetent patients Table 3: Antibiotic selection options for patients with simple sepsis, community acquired, immunocompetent patients requiring hospitalization. Risk Factors for Select Organisms P. aeruginosa MRSA Invasive Candidiasis VRE (and other resistant GNR) Community acquired: • Known colonization with MDROs • Central venous catheter • Liver transplant • Prior IV antibiotics within 90 day • Recent MRSA infection • Broad-spectrum antibiotics • Known colonization • Known colonization with MDROs • Known MRSA colonization • + 1 of the following risk factors: • Prolonged broad antibacterial • Skin & Skin Structure and/or IV access site: ♦ Parenteral nutrition therapy Hospital acquired: ♦ Purulence ♦ Dialysis • Prolonged profound • Prior IV antibiotics within 90 days ♦ Abscess -

In Vitro Susceptibility of Multi-Drug Resistant Klebsiella Pneumoniae Strains Causing Nosocomial Infections to Fosfomycin. a Comparison of Determination Methods
pathogens Article In Vitro Susceptibility of Multi-Drug Resistant Klebsiella pneumoniae Strains Causing Nosocomial Infections to Fosfomycin. A Comparison of Determination Methods Beata M ˛aczy´nska 1,* , Justyna Paleczny 1 , Monika Oleksy-Wawrzyniak 1 , Irena Choroszy-Król 2 and Marzenna Bartoszewicz 1 1 Department of Pharmaceutical Microbiology and Parasitology, Faculty of Pharmacy, Medical University, 50-367 Wroclaw, Poland; [email protected] (J.P.); [email protected] (M.O.-W.); [email protected] (M.B.) 2 Department of Basic Sciences, Faculty of Health Sciences, Medical University, 50-367 Wroclaw, Poland; [email protected] * Correspondence: [email protected] Abstract: Introduction: Over the past few decades, Klebsiella pneumoniae strains increased their pathogenicity and antibiotic resistance, thereby becoming a major therapeutic challenge. One of the few available therapeutic options seems to be intravenous fosfomycin. Unfortunately, the determination of sensitivity to fosfomycin performed in hospital laboratories can pose a significant problem. Therefore, the aim of the present research was to evaluate the activity of fosfomycin against clinical, multidrug-resistant Klebsiella pneumoniae strains isolated from nosocomial infections between Citation: M ˛aczy´nska,B.; Paleczny, J.; 2011 and 2020, as well as to evaluate the methods routinely used in hospital laboratories to assess Oleksy-Wawrzyniak, M.; bacterial susceptibility to this antibiotic. Materials and Methods: 43 multidrug-resistant Klebsiella Choroszy-Król, I.; Bartoszewicz, M. strains isolates from various infections were tested. All the strains had ESBL enzymes, and 20 In Vitro Susceptibility of Multi-Drug also showed the presence of carbapenemases. Susceptibility was determined using the diffusion Resistant Klebsiella pneumoniae Strains method (E-test) and the automated system (Phoenix), which were compared with the reference Causing Nosocomial Infections to method (agar dilution). -
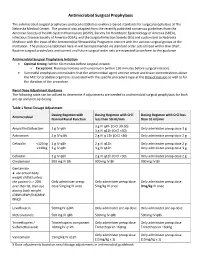
Antimicrobial Surgical Prophylaxis
Antimicrobial Surgical Prophylaxis The antimicrobial surgical prophylaxis protocol establishes evidence-based standards for surgical prophylaxis at The Nebraska Medical Center. The protocol was adapted from the recently published consensus guidelines from the American Society of Health-System Pharmacists (ASHP), Society for Healthcare Epidemiology of America (SHEA), Infectious Disease Society of America (IDSA), and the Surgical Infection Society (SIS) and customized to Nebraska Medicine with the input of the Antimicrobial Stewardship Program in concert with the various surgical groups at the institution. The protocol established here-in will be implemented via standard order sets utilized within One Chart. Routine surgical prophylaxis and current and future surgical order sets are expected to conform to this guidance. Antimicrobial Surgical Prophylaxis Initiation Optimal timing: Within 60 minutes before surgical incision o Exceptions: Fluoroquinolones and vancomycin (within 120 minutes before surgical incision) Successful prophylaxis necessitates that the antimicrobial agent achieve serum and tissue concentrations above the MIC for probable organisms associated with the specific procedure type at the time of incision as well as for the duration of the procedure. Renal Dose Adjustment Guidance The following table can be utilized to determine if adjustments are needed to antimicrobial surgical prophylaxis for both pre-op and post-op dosing. Table 1 Renal Dosage Adjustment Dosing Regimen with Dosing Regimen with CrCl Dosing Regimen with -

Guidelines on Urinary and Male Genital Tract Infections
European Association of Urology GUIDELINES ON URINARY AND MALE GENITAL TRACT INFECTIONS K.G. Naber, B. Bergman, M.C. Bishop, T.E. Bjerklund Johansen, H. Botto, B. Lobel, F. Jimenez Cruz, F.P. Selvaggi TABLE OF CONTENTS PAGE 1. INTRODUCTION 5 1.1 Classification 5 1.2 References 6 2. UNCOMPLICATED UTIS IN ADULTS 7 2.1 Summary 7 2.2 Background 8 2.3 Definition 8 2.4 Aetiological spectrum 9 2.5 Acute uncomplicated cystitis in pre-menopausal, non-pregnant women 9 2.5.1 Diagnosis 9 2.5.2 Treatment 10 2.5.3 Post-treatment follow-up 11 2.6 Acute uncomplicated pyelonephritis in pre-menopausal, non-pregnant women 11 2.6.1 Diagnosis 11 2.6.2 Treatment 12 2.6.3 Post-treatment follow-up 12 2.7 Recurrent (uncomplicated) UTIs in women 13 2.7.1 Background 13 2.7.2 Prophylactic antimicrobial regimens 13 2.7.3 Alternative prophylactic methods 14 2.8 UTIs in pregnancy 14 2.8.1 Epidemiology 14 2.8.2 Asymptomatic bacteriuria 15 2.8.3 Acute cystitis during pregnancy 15 2.8.4 Acute pyelonephritis during pregnancy 15 2.9 UTIs in post-menopausal women 15 2.10 Acute uncomplicated UTIs in young men 16 2.10.1 Pathogenesis and risk factors 16 2.10.2 Diagnosis 16 2.10.3 Treatment 16 2.11 References 16 3. UTIs IN CHILDREN 20 3.1 Summary 20 3.2 Background 20 3.3 Aetiology 20 3.4 Pathogenesis 20 3.5 Signs and symptoms 21 3.5.1 New-borns 21 3.5.2 Children < 6 months of age 21 3.5.3 Pre-school children (2-6 years of age) 21 3.5.4 School-children and adolescents 21 3.5.5 Severity of a UTI 21 3.5.6 Severe UTIs 21 3.5.7 Simple UTIs 21 3.5.8 Epididymo orchitis 22 3.6 Diagnosis 22 3.6.1 Physical examination 22 3.6.2 Laboratory tests 22 3.6.3 Imaging of the urinary tract 23 3.7 Schedule of investigation 24 3.8 Treatment 24 3.8.1 Severe UTIs 25 3.8.2 Simple UTIs 25 3.9 References 26 4. -

Penicillin Allergy Guidance Document
Penicillin Allergy Guidance Document Key Points Background Careful evaluation of antibiotic allergy and prior tolerance history is essential to providing optimal treatment The true incidence of penicillin hypersensitivity amongst patients in the United States is less than 1% Alterations in antibiotic prescribing due to reported penicillin allergy has been shown to result in higher costs, increased risk of antibiotic resistance, and worse patient outcomes Cross-reactivity between truly penicillin allergic patients and later generation cephalosporins and/or carbapenems is rare Evaluation of Penicillin Allergy Obtain a detailed history of allergic reaction Classify the type and severity of the reaction paying particular attention to any IgE-mediated reactions (e.g., anaphylaxis, hives, angioedema, etc.) (Table 1) Evaluate prior tolerance of beta-lactam antibiotics utilizing patient interview or the electronic medical record Recommendations for Challenging Penicillin Allergic Patients See Figure 1 Follow-Up Document tolerance or intolerance in the patient’s allergy history Consider referring to allergy clinic for skin testing Created July 2017 by Macey Wolfe, PharmD; John Schoen, PharmD, BCPS; Scott Bergman, PharmD, BCPS; Sara May, MD; and Trevor Van Schooneveld, MD, FACP Disclaimer: This resource is intended for non-commercial educational and quality improvement purposes. Outside entities may utilize for these purposes, but must acknowledge the source. The guidance is intended to assist practitioners in managing a clinical situation but is not mandatory. The interprofessional group of authors have made considerable efforts to ensure the information upon which they are based is accurate and up to date. Any treatments have some inherent risk. Recommendations are meant to improve quality of patient care yet should not replace clinical judgment. -

Fosfomycin Resistance in Escherichia Coli Isolates from South Korea and in Vitro Activity of Fosfomycin Alone and in Combination with Other Antibiotics
antibiotics Article Fosfomycin Resistance in Escherichia coli Isolates from South Korea and in vitro Activity of Fosfomycin Alone and in Combination with Other Antibiotics Hyeri Seok 1 , Ji Young Choi 2, Yu Mi Wi 3, Dae Won Park 1, Kyong Ran Peck 4 and Kwan Soo Ko 2,* 1 Division of Infectious Diseases, Department of Medicine, Korea University Ansan Hospital, Korea University College of Medicine, Ansan 15355, Korea; [email protected] (H.S.); [email protected] (D.W.P.) 2 Department of Microbiology, Sungkyunkwan University School of Medicine, Suwon 16419, Korea; [email protected] 3 Division of Infectious Diseases, Samsung Changwon Hospital, Sungkyunkwan University School of Medicine, Changwon 51353, Korea; [email protected] 4 Division of Infectious Diseases, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul 06351, Korea; [email protected] * Correspondence: [email protected]; Tel.: +82-31-299-6223 Received: 17 February 2020; Accepted: 3 March 2020; Published: 6 March 2020 Abstract: We investigated fosfomycin susceptibility in Escherichia coli clinical isolates from South Korea, including community-onset, hospital-onset, and long-term care facility (LTCF)-onset isolates. The resistance mechanisms and genotypes of fosfomycin-resistant isolates were also identified. Finally, the in vitro efficacy of combinations of fosfomycin with other antibiotics were examined in susceptible or extended spectrum β-lactamase (ESBL)-producing E. coli isolates. The fosfomycin resistance rate was 6.7% and was significantly higher in LTCF-onset isolates than community-onset and hospital-onset isolates. Twenty-one sequence types (STs) were identified among 19 fosfomycin-resistant E. coli isolates, showing diverse genotypes. fosA3 was found in only two isolates, and diverse genetic variations were identified in three genes associated with fosfomycin resistance, namely, GlpT, UhpT, and MurA. -

Fosfomycin for Lower Urinary Tract Infections: Comparative Clinical and Cost-Effectiveness
CADTH RAPID RESPONSE REPORT: SUMMARY OF ABSTRACTS Fosfomycin for Lower Urinary Tract Infections: Comparative Clinical and Cost-Effectiveness Service Line: Rapid Response Service Version: 1.0 Publication Date: April 12, 2018 Report Length: 8 Pages Authors: Kelsey Seal, Caitlyn Ford Cite As: Fosfomycin for Lower Urinary Tract Infections: Comparative Clinical and Cost-Effectiveness. Ottawa: CADTH; 2018 Apr. (CADTH rapid response report: summary of abstracts). Acknowledgments: Disclaimer: The inf ormation in this document is intended to help Canadian health care decision-makers, health care prof essionals, health sy stems leaders, and policy -makers make well-inf ormed decisions and thereby improv e the quality of health care serv ices. While patients and others may access this document, the document is made av ailable f or inf ormational purposes only and no representations or warranties are made with respect to its f itness f or any particular purpose. The inf ormation in this document should not be used as a substitute f or prof essional medical adv ice or as a substitute f or the application of clinical judgment in respect of the care of a particular patient or other prof essional judgment in any decision-making process. The Canadian Agency f or Drugs and Technologies in Health (CADTH) does not endorse any inf ormation, drugs, therapies, treatments, products, processes, or serv ic es. While care has been taken to ensure that the inf ormation prepared by CADTH in this document is accurate, complete, and up-to-date as at the applicable date the material was f irst published by CADTH, CADTH does not make any guarantees to that ef f ect. -

Safety and Efficacy of Ceftaroline Fosamil in the Management of Community-Acquired Bacterial Pneumonia Heather F
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Scholarly Papers 2014 Safety and Efficacy of Ceftaroline Fosamil in the Management of Community-Acquired Bacterial Pneumonia Heather F. DeBellis Kimberly L. Barefield Philadelphia College of Osteopathic Medicine, [email protected] Follow this and additional works at: https://digitalcommons.pcom.edu/scholarly_papers Part of the Medicine and Health Sciences Commons Recommended Citation DeBellis, Heather F. and Barefield, Kimberly L., "Safety and Efficacy of Ceftaroline Fosamil in the Management of Community- Acquired Bacterial Pneumonia" (2014). PCOM Scholarly Papers. 1913. https://digitalcommons.pcom.edu/scholarly_papers/1913 This Article is brought to you for free and open access by DigitalCommons@PCOM. It has been accepted for inclusion in PCOM Scholarly Papers by an authorized administrator of DigitalCommons@PCOM. For more information, please contact [email protected]. Open Access: Full open access to Clinical Medicine Reviews this and thousands of other papers at http://www.la-press.com. in Therapeutics Safety and Efficacy of Ceftaroline Fosamil in the Management of Community- Acquired Bacterial Pneumonia Heather F. DeBellis and Kimberly L. Tackett South University School of Pharmacy, Savannah, GA, USA. ABSTR ACT: Ceftaroline fosamil is a new fifth-generation cephalosporin indicated for the treatment of community-acquired bacterial pneumonia (CABP). It possesses antimicrobial effects against both Gram-positive and Gram-negative bacteria, including methicillin-resistant Staphylococcus aureus (MRSA), but not against anaerobes. Organisms covered by this novel agent that are commonly associated with CABP are Streptococcus pneumoniae, Staphylococcus aureus, Haemophilus influenzae, Moraxella catarrhalis, and Klebsiella pneumoniae; however, ceftaroline fosamil lacks antimicrobial activity against Pseudomonas and Acinetobacter species. -

Management of Penicillin and Beta-Lactam Allergy
Management of Penicillin and Beta-Lactam Allergy (NB Provincial Health Authorities Anti-Infective Stewardship Committee, September 2017) Key Points • Beta-lactams are generally safe; allergic and adverse drug reactions are over diagnosed and over reported • Nonpruritic, nonurticarial rashes occur in up to 10% of patients receiving penicillins. These rashes are usually not allergic and are not a contraindication to the use of a different beta-lactam • The frequently cited risk of 8 to 10% cross-reactivity between penicillins and cephalosporins is an overestimate based on studies from the 1970’s that are now considered flawed • Expect new intolerances (i.e. any allergy or adverse reaction reported in a drug allergy field) to be reported after 0.5 to 4% of all antimicrobial courses depending on the gender and specific antimicrobial. Expect a higher incidence of new intolerances in patients with three or more prior medication intolerances1 • For type-1 immediate hypersensitivity reactions (IgE-mediated), cross-reactivity among penicillins (table 1) is expected due to similar core structure and/or major/minor antigenic determinants, use not recommended without desensitization • For type-1 immediate hypersensitivity reactions, cross-reactivity between penicillins (table 1) and cephalosporins is due to similarities in the side chains; risk of cross-reactivity will only be significant between penicillins and cephalosporins with similar side chains • Only type-1 immediate hypersensitivity to a penicillin manifesting as anaphylaxis, bronchospasm, -

Fosfomycin Activity Versus Carbapenem-Resistant Enterobacteriaceae and Vancomycin-Resistant Enterococcus, Detroit, 2008–10
The Journal of Antibiotics (2013) 66, 625–627 & 2013 Japan Antibiotics Research Association All rights reserved 0021-8820/13 www.nature.com/ja NOTE Fosfomycin activity versus carbapenem-resistant Enterobacteriaceae and vancomycin-resistant Enterococcus, Detroit, 2008–10 Jason M Pogue1,2, Dror Marchaim2,3,4, Odaliz Abreu-Lanfranco2, Bharath Sunkara2, Ryan P Mynatt1, Jing J Zhao1, Suchitha Bheemreddy2, Kayoko Hayakawa2, Emily T Martin1, Sorabh Dhar2, Keith S Kaye2 and Paul R Lephart5 The Journal of Antibiotics (2013) 66, 625–627; doi:10.1038/ja.2013.56; published online 29 May 2013 Keywords: CRE; HAI; multidrug resistant; UTI; VRE; in-vitro Enterobacteriaceae and enterococci are among the most common culture coupled with potential clinical signs of infection such as causes of urinary tract infections (UTI) in adults.1 Carbapenem- mental status changes. In these scenarios, when CRE or VRE are the resistant Enterobacteriaceae (CRE) and vancomycin-resistant offending pathogens, often in elderly incontinent institutionalized Enterococcus (VRE) are multidrug-resistant (MDR) pathogens, individuals,12,13 patients are hospitalized and treated with intravenous which have become endemic in many locations worldwide.2,3 These (IV) broad-spectrum and sometime toxic agents, owing to lack of oral pathogens are associated with devastating outcomes among patients regimens. If these patients could have been managed in the outpatient and their continued dissemination and spread pose a threat to public settings, both the patients and the acute-care facilities -

Cephalosporins Can Be Prescribed Safely for Penicillin-Allergic Patients ▲
JFP_0206_AE_Pichichero.Final 1/23/06 1:26 PM Page 106 APPLIED EVIDENCE New research findings that are changing clinical practice Michael E. Pichichero, MD University of Rochester Cephalosporins can be Medical Center, Rochester, NY prescribed safely for penicillin-allergic patients Practice recommendations an allergic reaction to cephalosporins, ■ The widely quoted cross-allergy risk compared with the incidence of a primary of 10% between penicillin and (and unrelated) cephalosporin allergy. cephalosporins is a myth (A). Most people produce IgG and IgM antibodies in response to exposure to ■ Cephalothin, cephalexin, cefadroxil, penicillin1 that may cross-react with and cefazolin confer an increased risk cephalosporin antigens.2 The presence of of allergic reaction among patients these antibodies does not predict allergic, with penicillin allergy (B). IgE cross-sensitivity to a cephalosporin. ■ Cefprozil, cefuroxime, cefpodoxime, Even penicillin skin testing is generally not ceftazidime, and ceftriaxone do not predictive of cephalosporin allergy.3 increase risk of an allergic reaction (B). Reliably predicting cross-reactivity ndoubtedly you have patients who A comprehensive review of the evidence say they are allergic to penicillin shows that the attributable risk of a cross- U but have difficulty recalling details reactive allergic reaction varies and is of the reactions they experienced. To be strongest when the chemical side chain of safe, we often label these patients as peni- the specific cephalosporin is similar to that cillin-allergic without further questioning of penicillin or amoxicillin. and withhold not only penicillins but Administration of cephalothin, cepha- cephalosporins due to concerns about lexin, cefadroxil, and cefazolin in penicillin- potential cross-reactivity and resultant IgE- allergic patients is associated with a mediated, type I reactions.