Chapter 03 Lecture
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Review Article: Inhibition of Methanogenic Archaea by Statins As a Targeted Management Strategy for Constipation and Related Disorders
Alimentary Pharmacology and Therapeutics Review article: inhibition of methanogenic archaea by statins as a targeted management strategy for constipation and related disorders K. Gottlieb*, V. Wacher*, J. Sliman* & M. Pimentel† *Synthetic Biologics, Inc., Rockville, SUMMARY MD, USA. † Gastroenterology, Cedars-Sinai Background Medical Center, Los Angeles, CA, USA. Observational studies show a strong association between delayed intestinal transit and the production of methane. Experimental data suggest a direct inhibitory activity of methane on the colonic and ileal smooth muscle and Correspondence to: a possible role for methane as a gasotransmitter. Archaea are the only con- Dr K. Gottlieb, Synthetic Biologics, fi Inc., 9605 Medical Center Drive, rmed biological sources of methane in nature and Methanobrevibacter Rockville, MD 20850, USA. smithii is the predominant methanogen in the human intestine. E-mail: [email protected] Aim To review the biosynthesis and composition of archaeal cell membranes, Publication data archaeal methanogenesis and the mechanism of action of statins in this context. Submitted 8 September 2015 First decision 29 September 2015 Methods Resubmitted 7 October 2015 Narrative review of the literature. Resubmitted 20 October 2015 Accepted 20 October 2015 Results EV Pub Online 11 November 2015 Statins can inhibit archaeal cell membrane biosynthesis without affecting This uncommissioned review article was bacterial numbers as demonstrated in livestock and humans. This opens subject to full peer-review. the possibility of a therapeutic intervention that targets a specific aetiologi- cal factor of constipation while protecting the intestinal microbiome. While it is generally believed that statins inhibit methane production via their effect on cell membrane biosynthesis, mediated by inhibition of the HMG- CoA reductase, there is accumulating evidence for an alternative or addi- tional mechanism of action where statins inhibit methanogenesis directly. -

Vocabulario De Morfoloxía, Anatomía E Citoloxía Veterinaria
Vocabulario de Morfoloxía, anatomía e citoloxía veterinaria (galego-español-inglés) Servizo de Normalización Lingüística Universidade de Santiago de Compostela COLECCIÓN VOCABULARIOS TEMÁTICOS N.º 4 SERVIZO DE NORMALIZACIÓN LINGÜÍSTICA Vocabulario de Morfoloxía, anatomía e citoloxía veterinaria (galego-español-inglés) 2008 UNIVERSIDADE DE SANTIAGO DE COMPOSTELA VOCABULARIO de morfoloxía, anatomía e citoloxía veterinaria : (galego-español- inglés) / coordinador Xusto A. Rodríguez Río, Servizo de Normalización Lingüística ; autores Matilde Lombardero Fernández ... [et al.]. – Santiago de Compostela : Universidade de Santiago de Compostela, Servizo de Publicacións e Intercambio Científico, 2008. – 369 p. ; 21 cm. – (Vocabularios temáticos ; 4). - D.L. C 2458-2008. – ISBN 978-84-9887-018-3 1.Medicina �������������������������������������������������������������������������veterinaria-Diccionarios�������������������������������������������������. 2.Galego (Lingua)-Glosarios, vocabularios, etc. políglotas. I.Lombardero Fernández, Matilde. II.Rodríguez Rio, Xusto A. coord. III. Universidade de Santiago de Compostela. Servizo de Normalización Lingüística, coord. IV.Universidade de Santiago de Compostela. Servizo de Publicacións e Intercambio Científico, ed. V.Serie. 591.4(038)=699=60=20 Coordinador Xusto A. Rodríguez Río (Área de Terminoloxía. Servizo de Normalización Lingüística. Universidade de Santiago de Compostela) Autoras/res Matilde Lombardero Fernández (doutora en Veterinaria e profesora do Departamento de Anatomía e Produción Animal. -

IB DIPLOMA PROGRAMME Debora M
OXFORD IB PREPARED BIOLOGY IB DIPLOMA PROGRAMME Debora M. Primrose Contents Introduction iv 9 Plant biology (AHL) 1 Cell biology 9.1 Transport in the xylem of plants 103 9.2 Transport in the phloem of plants 107 1.1 Introduction to cells 2 9.3 Growth in plants 110 1.2 Ultrastructure of cells 4 9.4 Reproduction in plants 113 1.3 Membrane structure 6 1.4 Membrane transport 7 10 Genetics and evolution (AHL) 1.5 The origin of cells 9 10.1 Meiosis 117 1.6 Cell division 11 10.2 Inheritance 121 2 Molecular biology 10.3 Gene pools and speciation 125 2.1 Molecules to metabolism 14 11 Animal physiology (AHL) 2.2 Water 15 11.1 Antibody production and vaccination 128 2.3 Carbohydrates and lipids 16 11.2 Movement 133 2.4 Proteins 20 11.3 The kidney and osmoregulation 137 2.5 Enzymes 21 11.4 Sexual reproduction 141 2.6 Structure of DNA and RNA 23 2.7 DNA replication, transcription and translation 24 12 Data-based and practical questions 147 2.8 Cell respiration 26 2.9 Photosynthesis 28 A Neurobiology and behaviour 3 Genetics A.1 Neural development 157 A.2 The human brain 159 3.1 Genes 30 A.3 Perception of stimuli 161 3.2 Chromosomes 32 A.4 Innate and learned behaviour (AHL) 165 3.3 Meiosis 33 A.5 Neuropharmacology (AHL) 167 3.4 Inheritance 35 A.6 Ethology (AHL) 169 3.5 Genetic modification and biotechnology 37 B Biotechnology and bioinformatics 4 Ecology B.1 Microbiology: organisms in industry 172 4.1 Species, communities and ecosystems 40 B.2 Biotechnology in agriculture 174 4.2 Energy flow 43 B.3 Environmental protection 178 4.3 Carbon cycling 45 -
Cell Structure and Function
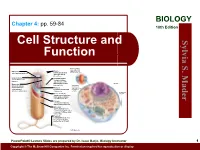
BIOLOGY Chapter 4: pp. 59-84 10th Edition Cell Structure and S. Sylvia Function Plasma membrane: outer surface that Ribosome: Fimbriae: regulates entrance site of protein synthesis hairlike bristles that and exit of molecules allow adhesion to Mader the surfaces Inclusion body: Conjugation pilus: stored nutrients for elongated, hollow later use appendage used for DNA transfer to other Nucleus: Mesosome: bacterial cells plasma membrane Cytoskeleton: maintains cell that folds into the Nucleoid: shape and assists cytoplasm and location of the bacterial movement of increases surface area chromosome cell parts: Endoplasmic Plasma membrane: reticulum: sheath around cytoplasm that regulates entrance and exit of molecules Cell wall: covering that supports, shapes, and protects cell Glycocalyx: gel-like coating outside cell wall; if compact, called a capsule; if diffuse, called a slime layer Flagellum: rotating filament present in some bacteria that pushes the cell forward *not in plant cells PowerPoint® Lecture Slides are prepared by Dr. Isaac Barjis, Biology Instructor 1 Copyright © The McGraw Hill Companies Inc. Permission required for reproduction or display Outline Cellular Level of Organization Cell theory Cell size Prokaryotic Cells Eukaryotic Cells Organelles Nucleus and Ribosome Endomembrane System Other Vesicles and Vacuoles Energy related organelles Cytoskeleton Centrioles, Cilia, and Flagella 2 Cell Theory Detailed study of the cell began in the 1830s A unifying concept in biology Originated from the work of biologists Schleiden and Schwann in 1838-9 States that: All organisms are composed of cells German botanist Matthais Schleiden in 1838 German zoologist Theodor Schwann in 1839 All cells come only from preexisting cells German physician Rudolph Virchow in 1850’s Cells are the smallest structural and functional unit of organisms 3 Organisms and Cells Copyright © The McGraw-Hill Companies, Inc. -

PEX5 Regulates Autophagy Via the Mtorc1-TFEB Axis During Starvation
Eun et al. Experimental & Molecular Medicine (2018) 50:4 DOI 10.1038/s12276-017-0007-8 Experimental & Molecular Medicine ARTICLE Open Access PEX5 regulates autophagy via the mTORC1-TFEB axis during starvation So Young Eun1,JoonNoLee2,In-KooNam2, Zhi-qiang Liu1,Hong-SeobSo 1, Seong-Kyu Choe1 and RaeKil Park2 Abstract Defects in the PEX5 gene impair the import of peroxisomal matrix proteins, leading to nonfunctional peroxisomes and other associated pathological defects such as Zellweger syndrome. Although PEX5 regulates autophagy process in a stress condition, the mechanisms controlling autophagy by PEX5 under nutrient deprivation are largely unknown. Herein, we show a novel function of PEX5 in the regulation of autophagy via Transcription Factor EB (TFEB). Under serum-starved conditions, when PEX5 is depleted, the mammalian target of rapamycin (mTORC1) inhibitor TSC2 is downregulated, which results in increased phosphorylation of the mTORC1 substrates, including 70S6K, S6K, and 4E- BP-1. mTORC1 activation further suppresses the nuclear localization of TFEB, as indicated by decreased mRNA levels of TFEB, LIPA, and LAMP1. Interestingly, peroxisomal mRNA and protein levels are also reduced by TFEB inactivation, indicating that TFEB might control peroxisome biogenesis at a transcriptional level. Conversely, pharmacological inhibition of mTOR resulting from PEX5 depletion during nutrient starvation activates TFEB by promoting nuclear localization of the protein. In addition, mTORC1 inhibition recovers the damaged-peroxisome biogenesis. These data suggest that PEX5 may be a critical regulator of lysosomal gene expression and autophagy through the mTOR-TFEB- autophagy axis under nutrient deprivation. 1234567890():,; 1234567890():,; Introduction Mitochondrial antiviral-signaling protein (MAVS) func- Peroxisome is an essential cellular organelle for per- tions as an antiviral signaling platform to induce the forming various metabolic activities, including oxidation interferon-independent signaling pathways4. -

Phagocytosis Dynamics Depends on Target Shape
Biophysical Journal Volume 105 September 2013 1143–1150 1143 Phagocytosis Dynamics Depends on Target Shape Debjani Paul,†‡6* Sarra Achouri,†6 Young-Zoon Yoon,† Jurgen Herre,§ Clare E. Bryant,{ and Pietro Cicuta† † ‡ Cavendish Laboratory, University of Cambridge, Cambridge, United Kingdom; Department of Biosciences and Bioengineering, § { Indian Institute of Technology Bombay, Powai, Mumbai, India; Department of Medicine and Department of Veterinary Medicine, University of Cambridge, Cambridge, United Kingdom ABSTRACT A complete understanding of phagocytosis requires insight into both its biochemical and physical aspects. One of the ways to explore the physical mechanism of phagocytosis is to probe whether and how the target properties (e.g., size, shape, surface states, stiffness, etc.) affect their uptake. Here we report an imaging-based method to explore phagocytosis kinetics, which is compatible with real-time imaging and can be used to validate existing reports using fixed and stained cells. We measure single-event engulfment time from a large number of phagocytosis events to compare how size and shape of targets determine their engulfment. The data shows an increase in the average engulfment time for increased target size, for spherical particles. The uptake time data on nonspherical particles confirms that target shape plays a more dominant role than target size for phagocytosis: Ellipsoids with an eccentricity of 0.954 and much smaller surface areas than spheres were taken up five times more slowly than spherical targets. INTRODUCTION Macrophages can engulf a wide variety of targets, such as the degradation of phagosomal contents and the induction invading pathogens, dead cells, foreign airborne particles of the appropriate immune responses. -

Actin Nucleator Spire 1 Is a Regulator of Ectoplasmic Specialization in the Testis Qing Wen1,Nanli1,Xiangxiao 1,2,Wing-Yeelui3, Darren S
Wen et al. Cell Death and Disease (2018) 9:208 DOI 10.1038/s41419-017-0201-6 Cell Death & Disease ARTICLE Open Access Actin nucleator Spire 1 is a regulator of ectoplasmic specialization in the testis Qing Wen1,NanLi1,XiangXiao 1,2,Wing-yeeLui3, Darren S. Chu1, Chris K. C. Wong4, Qingquan Lian5,RenshanGe5, Will M. Lee3, Bruno Silvestrini6 and C. Yan Cheng 1 Abstract Germ cell differentiation during the epithelial cycle of spermatogenesis is accompanied by extensive remodeling at the Sertoli cell–cell and Sertoli cell–spermatid interface to accommodate the transport of preleptotene spermatocytes and developing spermatids across the blood–testis barrier (BTB) and the adluminal compartment of the seminiferous epithelium, respectively. The unique cell junction in the testis is the actin-rich ectoplasmic specialization (ES) designated basal ES at the Sertoli cell–cell interface, and the apical ES at the Sertoli–spermatid interface. Since ES dynamics (i.e., disassembly, reassembly and stabilization) are supported by actin microfilaments, which rapidly converts between their bundled and unbundled/branched configuration to confer plasticity to the ES, it is logical to speculate that actin nucleation proteins play a crucial role to ES dynamics. Herein, we reported findings that Spire 1, an actin nucleator known to polymerize actins into long stretches of linear microfilaments in cells, is an important regulator of ES dynamics. Its knockdown by RNAi in Sertoli cells cultured in vitro was found to impede the Sertoli cell tight junction (TJ)-permeability barrier through changes in the organization of F-actin across Sertoli cell cytosol. Unexpectedly, Spire 1 knockdown also perturbed microtubule (MT) organization in Sertoli cells cultured in vitro. -

Magnetosensitive Neurons Mediate Geomagnetic Orientation in Caenorhabditis Elegans
RESEARCH ARTICLE elifesciences.org Magnetosensitive neurons mediate geomagnetic orientation in Caenorhabditis elegans Andres´ Vidal-Gadea1†, Kristi Ward1, Celia Beron1, Navid Ghorashian2, Sertan Gokce3, Joshua Russell1, Nicholas Truong1, Adhishri Parikh1, Otilia Gadea1, Adela Ben-Yakar2, Jonathan Pierce-Shimomura1* 1Department of Neuroscience; Center for Brain, Behavior and Evolution; Center for Learning and Memory; Waggoner Center for Alcohol and Addiction Research; Institute of Cell and Molecular Biology, University of Texas at Austin, Austin, United States; 2Department of Mechanical Engineering, University of Texas at Austin, Austin, United States; 3Department of Electrical Engineering, University of Texas at Austin, Austin, United States Abstract Many organisms spanning from bacteria to mammals orient to the earth’s magnetic field. For a few animals, central neurons responsive to earth-strength magnetic fields have been identified; however, magnetosensory neurons have yet to be identified in any animal. We show that the nematode Caenorhabditis elegans orients to the earth’s magnetic field during vertical burrowing migrations. Well-fed worms migrated up, while starved worms migrated down. Populations isolated from around the world, migrated at angles to the magnetic vector that would optimize vertical translation in their native soil, with northern- and southern-hemisphere worms displaying opposite migratory preferences. Magnetic orientation and vertical migrations required the TAX-4 cyclic nucleotide-gated ion channel in the AFD sensory neuron pair. Calcium imaging showed that these *For correspondence: jonps@ neurons respond to magnetic fields even without synaptic input. C. elegans may have adapted austin.utexas.edu magnetic orientation to simplify their vertical burrowing migration by reducing the orientation Present address: †School of task from three dimensions to one. -

The Association of Peroxisomes with the Developing Cell Plate in Dividing Onion Root Cells Depends on Actin Microfilaments and Myosin
Planta (2003) 218: 204–216 DOI 10.1007/s00425-003-1096-2 ORIGINAL ARTICLE David A. Collings Æ John D. I. Harper Æ Kevin C. Vaughn The association of peroxisomes with the developing cell plate in dividing onion root cells depends on actin microfilaments and myosin Received: 5 April 2003 / Accepted: 23 June 2003 / Published online: 21 August 2003 Ó Springer-Verlag 2003 Abstract We have investigated changes in the distribu- cling of excess membranes from secretory vesicles via the tion of peroxisomes through the cell cycle in onion b-oxidation pathway. Differences in aggregation, a (Allium cepa L.) root meristem cells with immunofluo- phenomenon which occurs in onion, some other mono- rescence and electron microscopy, and in leek (Allium cots and to a lesser extent in tobacco BY-2 suspension porrum L.) epidermal cells with immunofluorescence and cells, but which is not obvious in the roots of Arabidopsis peroxisomal-targeted green fluorescent protein. During thaliana (L.) Heynh., may reflect differences within the interphase and mitosis, peroxisomes distribute randomly primary cell walls of these plants. throughout the cytoplasm, but beginning late in ana- phase, they accumulate at the division plane. Initially, Keywords Actin microfilaments Æ Allium Æ peroxisomes occur within the microtubule phragmoplast Microtubule Æ Cell plate Æ Peroxisome Æ Phragmoplast in two zones on either side of the developing cell plate. However, as the phragmoplast expands outwards to Abbreviations BDM: 2,3-butanedione monoxime Æ DAPI: form an annulus, peroxisomes redistribute into a ring 4¢,6-diamidino-2-phenylindole Æ ER: endoplasmic reti- immediately inside the location of the microtubules. -

The Still Valid Fluid Mosaic Model for Molecular Organization of Biomembranes: Accumulating Data Confirm It
DISCOVERIES 2013, Oct-Dec; 1(1): e7 DOI: 10.15190/d.2013.7 The still actual fluid mosaic model for membrane organization Focused REVIEW The still valid fluid mosaic model for molecular organization of biomembranes: accumulating data confirm it Mircea Leabu1,* 1University of Medicine and Pharmacy “Carol Davila”, Department of Cellular and Molecular Medicine; “Victor Babes” National Institute of Pathology; University of Bucharest, Research Center for Applied Ethics *Correspondence to: Mircea Leabu, PhD, “Victor Babes” National Institute of Pathology, 99101, Splaiul Independentei, 050096, Bucharest, Romania; E-mail: [email protected] Citation: Leabu M. The still valid fluid mosaic model for molecular organization of biomembranes: accumulating data confirm it. Discoveries 2013, Oct-Dec; 1(1): e7. DOI: 10.15190/d.2013.7 ABSTRACT Keywords: membrane organization, membrane fluidity, membrane microdomains, Singer-Nicolson More than forty years passed since Singer and model, science history Nicolson launched the fluid mosaic model related to molecular organization and dynamics of cell Introduction membranes, applicable to endomembranes as well. For 20 years I and perhaps many other professors During this period of time, that will reach half a have been teaching students in medicine about the century soon, accumulating data all confirm, but not molecular organization of the cell membrane, infirm the brilliant idea of such a model. presenting with a high enthusiasm the fluid mosaic Sometimes, the results developed the model in a model launched by Seymour Jonathan Singer and very impacting manner, as was the case with the Garth L. Nicolson in 19721, and considering it as an introduction of the membrane microdomain concept inspired, still actual scientific and even pedagogic (mainly lipid rafts organization). -

Cell Structure and Function in the Bacteria and Archaea
4 Chapter Preview and Key Concepts 4.1 1.1 DiversityThe Beginnings among theof Microbiology Bacteria and Archaea 1.1. •The BacteriaThe are discovery classified of microorganismsinto several Cell Structure wasmajor dependent phyla. on observations made with 2. theThe microscope Archaea are currently classified into two 2. •major phyla.The emergence of experimental 4.2 Cellscience Shapes provided and Arrangements a means to test long held and Function beliefs and resolve controversies 3. Many bacterial cells have a rod, spherical, or 3. MicroInquiryspiral shape and1: Experimentation are organized into and a specific Scientificellular c arrangement. Inquiry in the Bacteria 4.31.2 AnMicroorganisms Overview to Bacterialand Disease and Transmission Archaeal 4.Cell • StructureEarly epidemiology studies suggested how diseases could be spread and 4. Bacterial and archaeal cells are organized at be controlled the cellular and molecular levels. 5. • Resistance to a disease can come and Archaea 4.4 External Cell Structures from exposure to and recovery from a mild 5.form Pili allowof (or cells a very to attach similar) to surfacesdisease or other cells. 1.3 The Classical Golden Age of Microbiology 6. Flagella provide motility. Our planet has always been in the “Age of Bacteria,” ever since the first 6. (1854-1914) 7. A glycocalyx protects against desiccation, fossils—bacteria of course—were entombed in rocks more than 3 billion 7. • The germ theory was based on the attaches cells to surfaces, and helps observations that different microorganisms years ago. On any possible, reasonable criterion, bacteria are—and always pathogens evade the immune system. have been—the dominant forms of life on Earth. -

The Dynamics and Function of the Endolysosomal/Lysosomal System
The Dynamics and Function of the Endolysosomal/Lysosomal System Luther John Davis University of Cambridge Homerton College This dissertation is submitted for the degree of Doctor of Philosophy September 2018 I Acknowledgements Firstly, I would like to thank Prof. J Paul Luzio for his invaluable guidance and effort towards the production and proofreading of this thesis. I would also like to thank Chloe Taylor, Ian Baines, Theo Dare, and Ashley Broom for their expertise, assistance and reassurance. I am grateful to the BBSRC and GSK for funding my project for four years. I thank Sally Gray, Nick Bright, and Lena Wartosch for their technical assistance and support. Finally I would like to express my gratitude to Matthew Gratian and Mark Bowen for training and assistance with microscopy, as well as Reiner Schulte, Chiara Cossetti, and Gabriela Grondys-Kotarba for their help with flow cytometry and cell sorting. II Preface This dissertation is the result of my own work and includes nothing which is the outcome of work done in collaboration except as declared in the Preface and specified in the text. It is not substantially the same as any that I have submitted, or, is being concurrently submitted for a degree or diploma or other qualification at the University of Cambridge or any other University or similar institution except as declared in the Preface and specified in the text. I further state that no substantial part of my dissertation has already been submitted, or, is being concurrently submitted for any such degree, diploma or other qualification at the University of Cambridge or any other University or similar institution except as declared in the Preface and specified in the text.