Nanoarchitecture and Dynamics of the Mouse Enteric Glycocalyx Examined by Freeze-Etching Electron Tomography and Intravital Microscopy
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Comparative Study of the Ultrastructure of Microvilli in the Epithelium of Small and Large Intestine of Mice
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by PubMed Central A COMPARATIVE STUDY OF THE ULTRASTRUCTURE OF MICROVILLI IN THE EPITHELIUM OF SMALL AND LARGE INTESTINE OF MICE T. M. MUKHERJEE and A. WYNN WILLIAMS From the Electron Microscope Laboratory, the Departlnent of Pathology, the University of Otago Medical School, Dunedin, New Zealand ABSTRACT A comparative analysis of the fine structure of the microvilli on jejunal and colonic epi- thelial cells of the mouse intestine has been made. The microvilli in these two locations demonstrate a remarkably similar fine structure with respect to the thickness of the plasma membrane, the extent of the filament-free zone, and the characteristics of the microfila- ments situated within the microvillous core. Some of the core microfilaments appear to continue across the plasma membrane limiting the tip of the microvillus. The main differ- ence between the microvilli of small intestine and colon is in the extent and organization of the surface coat. In the small intestine, in addition to the commonly observed thin surface "fuzz," occasional areas of the jejunal villus show a more conspicuous surface coat covering the tips of the microvilli. Evidence has been put forward which indicates that the surface coat is an integral part of the epithelial cells. In contrast to the jejunal epithelium, the colonic epithelium is endowed with a thicker surface coat. Variations in the organization of the surface coat at different levels of the colonic crypts have also been noted. The func- tional significance of these variations in the surface coat is discussed. -

Gastrointestinal Stem Cells in Health and Disease: from Flies to Humans Hongjie Li1,2 and Heinrich Jasper1,*
© 2016. Published by The Company of Biologists Ltd | Disease Models & Mechanisms (2016) 9, 487-499 doi:10.1242/dmm.024232 REVIEW SUBJECT COLLECTION: TRANSLATIONAL IMPACT OF DROSOPHILA Gastrointestinal stem cells in health and disease: from flies to humans Hongjie Li1,2 and Heinrich Jasper1,* ABSTRACT is Barrett’s metaplasia (see Box 1), in which the esophageal The gastrointestinal tract of complex metazoans is highly squamous epithelium acquires properties that are reminiscent of the compartmentalized. It is lined by a series of specialized epithelia gastric or intestinal columnar epithelium. This transformation has that are regenerated by specific populations of stem cells. To maintain been associated with acid reflux disease and is believed to be a cause tissue homeostasis, the proliferative activity of stem and/or progenitor of esophageal adenocarcinomas (Falk, 2002; Hvid-Jensen et al., cells has to be carefully controlled and coordinated with regionally 2011). Dysplasia (see Box 1), another type of epithelial lesion that distinct programs of differentiation. Metaplasias and dysplasias, commonly affects the human GI tract, is characterized by aberrant precancerous lesions that commonly occur in the human cell proliferation and differentiation. Dysplastic changes are often gastrointestinal tract, are often associated with the aberrant found at later stages during epithelial carcinogenesis than are proliferation and differentiation of stem and/or progenitor cells. The metaplasias, and eventually lead to invasive carcinoma (see Box 1) increasingly sophisticated characterization of stem cells in the (Correa and Houghton, 2007; Ullman et al., 2009). Much remains to gastrointestinal tract of mammals and of the fruit fly Drosophila has be learnt about intestinal metaplasias and dysplasias, not least provided important new insights into these processes and into the because of their clinical significance, such as the exact cellular mechanisms that drive epithelial dysfunction. -

Study of Mucin Turnover in the Small Intestine by in Vivo Labeling Hannah Schneider, Thaher Pelaseyed, Frida Svensson & Malin E
www.nature.com/scientificreports OPEN Study of mucin turnover in the small intestine by in vivo labeling Hannah Schneider, Thaher Pelaseyed, Frida Svensson & Malin E. V. Johansson Mucins are highly glycosylated proteins which protect the epithelium. In the small intestine, the goblet Received: 23 January 2018 cell-secreted Muc2 mucin constitutes the main component of the loose mucus layer that traps luminal Accepted: 26 March 2018 material. The transmembrane mucin Muc17 forms part of the carbohydrate-rich glycocalyx covering Published: xx xx xxxx intestinal epithelial cells. Our study aimed at investigating the turnover of these mucins in the small intestine by using in vivo labeling of O-glycans with N-azidoacetylgalactosamine. Mice were injected intraperitoneally and sacrifced every hour up to 12 hours and at 24 hours. Samples were fxed with preservation of the mucus layer and stained for Muc2 and Muc17. Turnover of Muc2 was slower in goblet cells of the crypts compared to goblet cells along the villi. Muc17 showed stable expression over time at the plasma membrane on villi tips, in crypts and at crypt openings. In conclusion, we have identifed diferent subtypes of goblet cells based on their rate of mucin biosynthesis and secretion. In order to protect the intestinal epithelium from chemical and bacterial hazards, fast and frequent renewal of the secreted mucus layer in the villi area is combined with massive secretion of stored Muc2 from goblet cells in the upper crypt. Te small intestinal epithelium is constantly aiming to balance efective nutritional uptake with minimal damage due to exposure to ingested, secreted and resident agents. -

Vocabulario De Morfoloxía, Anatomía E Citoloxía Veterinaria
Vocabulario de Morfoloxía, anatomía e citoloxía veterinaria (galego-español-inglés) Servizo de Normalización Lingüística Universidade de Santiago de Compostela COLECCIÓN VOCABULARIOS TEMÁTICOS N.º 4 SERVIZO DE NORMALIZACIÓN LINGÜÍSTICA Vocabulario de Morfoloxía, anatomía e citoloxía veterinaria (galego-español-inglés) 2008 UNIVERSIDADE DE SANTIAGO DE COMPOSTELA VOCABULARIO de morfoloxía, anatomía e citoloxía veterinaria : (galego-español- inglés) / coordinador Xusto A. Rodríguez Río, Servizo de Normalización Lingüística ; autores Matilde Lombardero Fernández ... [et al.]. – Santiago de Compostela : Universidade de Santiago de Compostela, Servizo de Publicacións e Intercambio Científico, 2008. – 369 p. ; 21 cm. – (Vocabularios temáticos ; 4). - D.L. C 2458-2008. – ISBN 978-84-9887-018-3 1.Medicina �������������������������������������������������������������������������veterinaria-Diccionarios�������������������������������������������������. 2.Galego (Lingua)-Glosarios, vocabularios, etc. políglotas. I.Lombardero Fernández, Matilde. II.Rodríguez Rio, Xusto A. coord. III. Universidade de Santiago de Compostela. Servizo de Normalización Lingüística, coord. IV.Universidade de Santiago de Compostela. Servizo de Publicacións e Intercambio Científico, ed. V.Serie. 591.4(038)=699=60=20 Coordinador Xusto A. Rodríguez Río (Área de Terminoloxía. Servizo de Normalización Lingüística. Universidade de Santiago de Compostela) Autoras/res Matilde Lombardero Fernández (doutora en Veterinaria e profesora do Departamento de Anatomía e Produción Animal. -
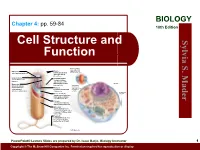
Cell Structure and Function
BIOLOGY Chapter 4: pp. 59-84 10th Edition Cell Structure and S. Sylvia Function Plasma membrane: outer surface that Ribosome: Fimbriae: regulates entrance site of protein synthesis hairlike bristles that and exit of molecules allow adhesion to Mader the surfaces Inclusion body: Conjugation pilus: stored nutrients for elongated, hollow later use appendage used for DNA transfer to other Nucleus: Mesosome: bacterial cells plasma membrane Cytoskeleton: maintains cell that folds into the Nucleoid: shape and assists cytoplasm and location of the bacterial movement of increases surface area chromosome cell parts: Endoplasmic Plasma membrane: reticulum: sheath around cytoplasm that regulates entrance and exit of molecules Cell wall: covering that supports, shapes, and protects cell Glycocalyx: gel-like coating outside cell wall; if compact, called a capsule; if diffuse, called a slime layer Flagellum: rotating filament present in some bacteria that pushes the cell forward *not in plant cells PowerPoint® Lecture Slides are prepared by Dr. Isaac Barjis, Biology Instructor 1 Copyright © The McGraw Hill Companies Inc. Permission required for reproduction or display Outline Cellular Level of Organization Cell theory Cell size Prokaryotic Cells Eukaryotic Cells Organelles Nucleus and Ribosome Endomembrane System Other Vesicles and Vacuoles Energy related organelles Cytoskeleton Centrioles, Cilia, and Flagella 2 Cell Theory Detailed study of the cell began in the 1830s A unifying concept in biology Originated from the work of biologists Schleiden and Schwann in 1838-9 States that: All organisms are composed of cells German botanist Matthais Schleiden in 1838 German zoologist Theodor Schwann in 1839 All cells come only from preexisting cells German physician Rudolph Virchow in 1850’s Cells are the smallest structural and functional unit of organisms 3 Organisms and Cells Copyright © The McGraw-Hill Companies, Inc. -

NIH Public Access Author Manuscript Mucosal Immunol
NIH Public Access Author Manuscript Mucosal Immunol. Author manuscript; available in PMC 2014 March 01. NIH-PA Author ManuscriptPublished NIH-PA Author Manuscript in final edited NIH-PA Author Manuscript form as: Mucosal Immunol. 2013 March ; 6(2): 379–392. doi:10.1038/mi.2012.81. Molecular Organization of the Mucins and Glycocalyx Underlying Mucus Transport Over Mucosal Surfaces of the Airways Mehmet Kesimer*,‡,▫, Camille Ehre*, Kimberly A. Burns*, C. William Davis*,†, John K. Sheehan*,‡, and Raymond J. Pickles*,§ *Cystic Fibrosis/Pulmonary Research and Treatment Center, University of North Carolina, Chapel Hill, NC 27599-7248 ‡Department of Biochemistry and Biophysics, University of North Carolina, Chapel Hill, NC 27599-7248 †Department of Cell and Molecular Physiology, University of North Carolina, Chapel Hill, NC 27599-7248 §Department of Microbiology and Immunology, University of North Carolina, Chapel Hill, NC 27599-7248 Abstract Mucus, with its burden of inspired particulates, and pathogens, is cleared from mucosal surfaces of the airways by cilia beating within the periciliary layer (PCL). The PCL is held to be ‘watery’ and free of mucus by thixotropic-like forces arising from beating cilia. With radii of gyration ~250 nm, however, polymeric mucins should reptate readily into the PCL, so we assessed the glycocalyx for barrier functions. The PCL stained negative for MUC5AC and MUC5B, but it was positive for keratan sulfate, a glycosaminoglycan commonly associated with glycoconjugates. Shotgun proteomics showed keratan sulfate-rich fractions from mucus containing abundant tethered mucins, MUC1, MUC4, and MUC16, but no proteoglycans. Immuno-histology by light and electron microscopy localized MUC1 to microvilli, MUC4 and MUC20 to cilia, and MUC16 to goblet cells. -

Nomina Histologica Veterinaria, First Edition
NOMINA HISTOLOGICA VETERINARIA Submitted by the International Committee on Veterinary Histological Nomenclature (ICVHN) to the World Association of Veterinary Anatomists Published on the website of the World Association of Veterinary Anatomists www.wava-amav.org 2017 CONTENTS Introduction i Principles of term construction in N.H.V. iii Cytologia – Cytology 1 Textus epithelialis – Epithelial tissue 10 Textus connectivus – Connective tissue 13 Sanguis et Lympha – Blood and Lymph 17 Textus muscularis – Muscle tissue 19 Textus nervosus – Nerve tissue 20 Splanchnologia – Viscera 23 Systema digestorium – Digestive system 24 Systema respiratorium – Respiratory system 32 Systema urinarium – Urinary system 35 Organa genitalia masculina – Male genital system 38 Organa genitalia feminina – Female genital system 42 Systema endocrinum – Endocrine system 45 Systema cardiovasculare et lymphaticum [Angiologia] – Cardiovascular and lymphatic system 47 Systema nervosum – Nervous system 52 Receptores sensorii et Organa sensuum – Sensory receptors and Sense organs 58 Integumentum – Integument 64 INTRODUCTION The preparations leading to the publication of the present first edition of the Nomina Histologica Veterinaria has a long history spanning more than 50 years. Under the auspices of the World Association of Veterinary Anatomists (W.A.V.A.), the International Committee on Veterinary Anatomical Nomenclature (I.C.V.A.N.) appointed in Giessen, 1965, a Subcommittee on Histology and Embryology which started a working relation with the Subcommittee on Histology of the former International Anatomical Nomenclature Committee. In Mexico City, 1971, this Subcommittee presented a document entitled Nomina Histologica Veterinaria: A Working Draft as a basis for the continued work of the newly-appointed Subcommittee on Histological Nomenclature. This resulted in the editing of the Nomina Histologica Veterinaria: A Working Draft II (Toulouse, 1974), followed by preparations for publication of a Nomina Histologica Veterinaria. -

Mechanical Characterisation of Bone Cells and Their Glycocalyx
Mechanical characterisation of bone cells and their glycocalyx a dissertation presented by Stefania Marcotti to The Department of Mechanical Engineering University of Sheffield Sheffield, United Kingdom in partial fulfilment of the requirements for the degree of Doctor of Philosophy Supervisor: Prof. Damien Lacroix Co-Supervisor: Dr. Gwendolen C Reilly November 2017 Abstract Mechanotransductionreferstotheprocessbywhichacellisabletotranslatemech- anical stimulation into biochemical signals. In bone, mechanotransduction regu- lates how cells detect environmental stimuli and use these to direct towards bone deposition or resorption. The mechanical properties of bone cells have an impact on the way mechanical stimulation is sensed, however, little evidence is available about how these properties influence mechanotransduction. The aim of the present Thesis was to quantify the mechanical properties of bone cells with a combined experimental and computational approach. Atomic force microscopy was employed to quantify the stiffness of bone cells and their gly- cocalyx. Changes in cell stiffness during osteocytogenesis were explored. Single molecule force spectroscopy of glycocalyx components was performed to evalu- ate their anchoring to the cytoskeleton. A single cell finite element model was designed to discern the contributions of sub-cellular components in response to simulated cell nano-indentation. Wide ranges of variation were found for bone cell stiffness and a method was proposed to determine suitable sample sizes to capture population heterogeneity. Bytargetingsinglecomponentsoftheboneglycocalyx, itwaspossibletohypothes- ise different mechanotransduction mechanisms depending on the hyaluronic acid attachment to the cytoskeleton. The developed computational framework showed similar results to the nano-indentation experiments and highlighted the role of the actin cytoskeleton in withstanding compression and distributing strain within the cell. iii Publications and conference presentations Book chapters Marcotti S and Reilly GC (2017). -

The Intestinal Stem Cell
Downloaded from genesdev.cshlp.org on September 29, 2021 - Published by Cold Spring Harbor Laboratory Press REVIEW The intestinal stem cell Nick Barker, Marc van de Wetering, and Hans Clevers1 Hubrecht Institute and University Medical Center Utrecht, Uppsalalaan 8, 3584CT Utrecht, the Netherlands The epithelium of the adult mammalian intestine is in a Claudinot et al. 2005). Of note, multipotency can only be constant dialog with its underlying mesenchyme to di- definitively demonstrated when transplantation can be rect progenitor proliferation, lineage commitment, ter- performed with a single cell, which is rarely possible. As minal differentiation, and, ultimately, cell death. The an alternative strategy, candidate stem cells are geneti- epithelium is shaped into spatially distinct compart- cally marked in situ, after which the introduced marker ments that are dedicated to each of these events. While allows the visualization of the modified stem cell and its the intestinal epithelium represents the most vigorously clonal offspring over time. As an example of the latter renewing adult tissue in mammals, the stem cells that approach, a progesterone-responsive version of the Cre fuel this self-renewal process have been identified only recombinase enzyme was specifically expressed in cells recently. The unique epithelial anatomy makes the in- residing in the bulge region of hair follicles using a trans- testinal crypt one of the most accessible models for the genic Keratin-15 promoter (Morris et al. 2004). Activa- study of adult stem cell biology. This review attempts to tion of the Cre enzyme by progesterone irreversibly ac- provide a comprehensive overview of four decades of re- tivated the genetic marker R26R-LacZ in the bulge cells. -

Immuno-Electron and Confocal Laser Scanning Microscopy of the Glycocalyx
biology Article Immuno-Electron and Confocal Laser Scanning Microscopy of the Glycocalyx Shailey Gale Twamley 1,2, Anke Stach 1, Heike Heilmann 3, Berit Söhl-Kielczynski 4, Verena Stangl 1,2, Antje Ludwig 1,2,*,† and Agnieszka Münster-Wandowski 3,*,† 1 Medizinische Klinik für Kardiologie und Angiologie, Charité—Universitätsmedizin Berlin, Corporate Member of Freie Universität Berlin, Humboldt-Universität zu Berlin, and Berlin Institute of Health, 10117 Berlin, Germany; [email protected] (S.G.T.); [email protected] (A.S.); [email protected] (V.S.) 2 DZHK (German Centre for Cardiovascular Research), Partner Site, 10117 Berlin, Germany 3 Institute of Integrative Neuroanatomy, Charité—Universitätsmedizin Berlin, Corporate Member of Freie Universität Berlin, Humboldt-Universität zu Berlin, and Berlin Institute of Health, 10117 Berlin, Germany; [email protected] 4 Institute for Integrative Neurophysiology—Universitätsmedizin Berlin, Corporate Member of Freie Universität Berlin, Humboldt-Universität zu Berlin, and Berlin Institute of Health, 10117 Berlin, Germany; [email protected] * Correspondence: [email protected] (A.L.); [email protected] (A.M.-W.) † These authors equally contributed. Simple Summary: The glycocalyx (GCX) is a hydrated, gel-like layer of biological macromolecules attached to the cell membrane. The GCX acts as a barrier and regulates the entry of external substances into the cell. The function of the GCX is highly dependent on its structure and composition. Citation: Twamley, S.G.; Stach, A.; Pathogenic factors can affect the protective structure of the GCX. We know very little about the three- Heilmann, H.; Söhl-Kielczynski, B.; dimensional organization of the GXC. The tiny and delicate structures of the GCX are difficult Stangl, V.; Ludwig, A.; to study by microscopic techniques. -

Characteristics of a Glycoprotein in the Ocular Surface Glycocalyx
Investigative Ophthalmology & Visual Science, Vol. 33, No. 1, January 1992 Copyright © Association for Research in Vision and Ophthalmology Characteristics of a Glycoprotein in the Ocular Surface Glycocalyx llene K. Gipson,*t Michelle Yankauckas,* Sandra J. Spurr-Michaud,* Ann S. Tisdale,* and William Rineharr* A monoclonal antibody has been produced that binds to the apical squames (flattened cells) of the rat ocular surface epithelium and to the goblet cells of the conjunctiva. Immunoelectron microscopic localization of the antigen indicates that in apical cells it is present along the apical-microplical mem- brane in the region of the glycocalyx. In subapical squames, the antigen is in cytoplasmic vesicles. In some goblet cells, the antigen is in the Golgi network, and in others, it is located primarily in the membrane of the mucous granules. SDS-PAGE and immunoblot analysis demonstrate that the molecu- lar weight of the antigen is greater than 205 kD, and the electrophoretic band stains with Alcian blue followed by silver stain. Periodate oxidation of immunoblots and cryostat sections removes antibody binding. Neuraminidase treatment of cryostat sections does not remove antibody binding, whereas N-glycanase does. Taken together, these data indicate that the antigen recognized by the monoclonal antibody is a carbohydrate epitope on a high-molecular-weight, highly glycosylated glycoprotein in the glycocalyx of the ocular surface epithelium and goblet cell mucin granule membrane. The antigen appears to be stored within cytoplasmic vesicles and reaches the glycocalyx when cells differentiate to the apical-most position where the glycocalyx interfaces with the mucin layer of the tear film. Invest Ophthalmol Vis Sci 33:218-227,1992 The ocular surface epithelium that covers the con- tissue stained with tannic acid demonstrates that the junctiva and cornea is nonkeratinizing, stratified, and glycocalyx is a fine, filamentous layer. -

Intestinal Organoids Generated from Human Pluripotent Stem Cells
DOI: 10.31662/jmaj.2019-0027 https://www.jmaj.jp/ Review Article Intestinal Organoids Generated from Human Pluripotent Stem Cells Satoru Tsuruta1),2), Hajime Uchida3), and Hidenori Akutsu2) Abstract: The gastrointestinal system is one of the most complex organ systems in the human body, and consists of numerous cell types originating from three germ layers. To understand intestinal development and homeostasis and elucidate the patho- genesis of intestinal disorders, including unidentified diseases, several in vitro models have been developed. Human pluripo- tent stem cells (PSCs), including embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), have remarkable developmental plasticity and possess the potential for a wide variety of applications. Three-dimensional organs, termed organoids and produced in vitro by PSCs, contain not only epithelium but also mesenchymal tissue and partially recapitu- late intestinal functions. Such intestinal organoids have begun to be applied in disease models and drug development and have contributed to a detailed analysis of molecular interactions and findings in the synergistic development of biomedicine for human digestive organs. In this review, we describe gastrointestinal organoid technology derived from PSCs and consid- er its potential applications. Key Words: intestinal organoids, embryonic stem cells, induced pluripotent stem cells, gastrointestinal disease, drug discovery Introduction the anterior to posterior axis. Accompanied by the develop- ment of the fetus, repeated gut