(ACIP) General Best Guidance for Immunization
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Safety of Immunization During Pregnancy a Review of the Evidence
Safety of Immunization during Pregnancy A review of the evidence Global Advisory Committee on Vaccine Safety © World Health Organization 2014 All rights reserved. Publications of the World Health Organization are available on the WHO website (www.who.int) or can be purchased from WHO Press, World Health Organization, 20 Avenue Appia, 1211 Geneva 27, Switzerland (tel.: +41 22 791 3264; fax: +41 22 791 4857; e-mail: [email protected]). Requests for permission to reproduce or translate WHO publications –whether for sale or for non-commercial distribution– should be addressed to WHO Press through the WHO website (www.who.int/about/licensing/copyright_form/en/index.html). The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines for which there may not yet be full agreement. The mention of specific companies or of certain manufacturers’ products does not imply that they are endorsed or recommended by the World Health Organization in preference to others of a similar nature that are not mentioned. Errors and omissions excepted, the names of proprietary products are distinguished by initial capital letters. All reasonable precautions have been taken by the World Health Organization to verify the information contained in this publication. However, the published material is being distributed without warranty of any kind, either expressed or implied. -

Frequently Asked Questions About Measles Immunizations
Immunization Unit Assessment, Compliance, and Evaluation Group Measles FAQs Q: When did the vaccine for measles become available? A: The first measles vaccines were available in 1963, and were replaced with a much better vaccine in 1968. Q: Do people who received MMR in the 1960s need to have their dose repeated? A: Not necessarily. People who have documentation of receiving a live measles vaccine in the 1960s do not need to be revaccinated. People who were vaccinated with a killed or inactivated measles vaccine, or vaccine of unknown type, should be revaccinated with at least 1 dose of MMR. Q: What kind of vaccine is it? A: The vaccines available in the United States that protect against measles are the MMR and MMRV, which protects against measles, mumps, and rubella or measles, mumps, rubella, and varicella (chickenpox). They are both live attenuated, or weakened, vaccines. Q: Which vaccine can I get, the MMR or the MMRV? A: The MMRV is only approved for people aged 12 months – 12 years. The MMR is approved for anyone 6 months of age and older, but it is routinely recommended for use in people 12 months of age and older. Any dose of MMR received before 12 months of age is not considered valid and should be repeated in 4 weeks or at age 12 months, whichever is later. Q: How is the vaccine given? A: The vaccine is administered in the fatty layer of tissue underneath the skin. Q: Is the measles shot just a one-time dose? A: The measles vaccine is very effective. -

Adult Vaccines
What do flu, whooping cough, measles, shingles and pneumonia have in common? 1 They’re viruses that can make you very sick. 2 Vaccines can help prevent them. Protect yourself and those you care about. Get vaccinated at a network pharmacy near you. • Ask your pharmacist which vaccines are right for you. • Find out if your pharmacist can administer the recommended vaccinations. • Many vaccinations are covered by your plan at participating retail pharmacies. • Don’t forget to present your member ID card to the pharmacist at the time of service! The following vaccines are available and can be administered by pharmacists at participating network pharmacies: • Flu (seasonal influenza) • Meningitis • Travel Vaccines (typhoid, yellow • Tetanus/Diphtheria/Pertussis • Pneumonia fever, etc.) • Hepatitis • Rabies • Childhood Vaccines (MMR, etc.) • Human Papillomavirus (HPV) • Shingles/Zoster See other side for recommended adult vaccinations. The vaccinations you need ALL adults should get vaccinated for1: • Flu, every year. It’s especially important for pregnant women, older adults and people with chronic health conditions. • Tetanus, diphtheria and pertussis (whooping cough). Adults should get a one-time dose of the Tdap vaccine. It’s different from the tetanus vaccine (Td), which is given every 10 years. You may need additional vaccinations depending on your age1: Young adults not yet vaccinated need: Human papillomavirus (HPV) vaccine series (3 doses) if you are: • Female age 26 or younger • Male age 21 or younger • Male age 26 or younger who has sex with men, who is immunocompromised or who has HIV Adults born in the U.S. in 1957 or after need: Measles, mumps, rubella (MMR) vaccine2 Adults should get at least one dose of MMR vaccine, unless they’ve already gotten this vaccine or have immunity to measles, mumps and rubella Adults born in the U.S. -

Vaccines You Might Need?
Do You Know Which Adult Vaccines You Might Need? Vaccines are recommended for all adults based on factors such as age, travel, occupation, medical history, and vaccines they have had in the past. Below are the main vaccines you might need. But, this list may not include every vaccine that you need. Find out which vaccines you need by taking the quiz at: www.cdc.gov/vaccines/AdultQuiz/ ALL adults 19 and older, including pregnant women, need: Influenza vaccine every year • A flu vaccine is especially important for people with chronic health conditions, pregnant women, and older adults Tetanus, diphtheria, and pertussis (whooping cough) vaccine (Tdap) • Adults should get a one-time dose of Tdap. Adults can get Tdap no matter when they got their last tetanus vaccine (Td), which is given every 10 years • Pregnant women should get Tdap to protect themselves and their newborn babies from whooping cough In addition to influenza and Tdap vaccines, you may also need other vaccines depending on your age or other factors. Flip the page to learn more. Talk to your healthcare provider about which vaccines are right for you. National Center for Immunization and Respiratory Diseases Immunization Services Division CS235267 In addition to influenza and Tdap vaccines, you may also need other vaccines depending on your age… Young adults not yet vaccinated need: Adults born in the US in 1980 or after need: Human papillomavirus (HPV) vaccine series Varicella “chickenpox” vaccine* (3 doses) if you are: • Adults should get 2 doses of chickenpox vaccine • -

Measles: Chapter 7.1 Chapter 7: Measles Paul A
VPD Surveillance Manual 7 Measles: Chapter 7.1 Chapter 7: Measles Paul A. Gastanaduy, MD, MPH; Susan B. Redd; Nakia S. Clemmons, MPH; Adria D. Lee, MSPH; Carole J. Hickman, PhD; Paul A. Rota, PhD; Manisha Patel, MD, MS I. Disease Description Measles is an acute viral illness caused by a virus in the family paramyxovirus, genus Morbillivirus. Measles is characterized by a prodrome of fever (as high as 105°F) and malaise, cough, coryza, and conjunctivitis, followed by a maculopapular rash.1 The rash spreads from head to trunk to lower extremities. Measles is usually a mild or moderately severe illness. However, measles can result in complications such as pneumonia, encephalitis, and death. Approximately one case of encephalitis2 and two to three deaths may occur for every 1,000 reported measles cases.3 One rare long-term sequelae of measles virus infection is subacute sclerosing panencephalitis (SSPE), a fatal disease of the central nervous system that generally develops 7–10 years after infection. Among persons who contracted measles during the resurgence in the United States (U.S.) in 1989–1991, the risk of SSPE was estimated to be 7–11 cases/100,000 cases of measles.4 The risk of developing SSPE may be higher when measles occurs prior to the second year of life.4 The average incubation period for measles is 11–12 days,5 and the average interval between exposure and rash onset is 14 days, with a range of 7–21 days.1, 6 Persons with measles are usually considered infectious from four days before until four days after onset of rash with the rash onset being considered as day zero. -

Vaccine Information for PARENTS and CAREGIVERS
NATIONAL INSTITUTE FOR COMMUNICABLE DISEASES Division of the National Health Laboratory Service VACCINE INFOR MATION FOR PARENTS & CAREGIVERS First Edition November 2016 Editors-in-Chief Nkengafac Villyen Motaze (MD, MSc, PhD fellow), Melinda Suchard (MBBCh, FCPath (SA), MMed) Edited by: Cheryl Cohen, (MBBCh, FCPath (SA) Micro, DTM&H, MSc (Epi), Phd) Lee Baker, (Dip Pharm) Lucille Blumberg, (MBBCh, MMed (Micro) ID (SA) FFTM (RCPS, Glasgow) DTM&H DOH DCH) Published by: Ideas Wise and Wonderful (IWW) for National Institute for Communicable Diseases (NICD) First Edition: Copyright © 2016 Contributions by: Clement Adu-Gyamfi (BSc Hons, MSc), Jayendrie Thaver (BSc), Kerrigan McCarthy, (MBBCh, FCPath (SA), DTM&H, MPhil (Theol) Kirsten Redman (BSc Hons), Nishi Prabdial-Sing (PhD), Nonhlanhla Mbenenge (MBBCh, MMED) Philippa Hime (midwife), Vania Duxbury (BSc Hons), Wayne Howard (BSc Hons) Acknowledgments: Amayeza, Vaccine Information Centre (http://www.amayeza-info.co.za/) Centre for Communicable Diseases Fact Sheets (http://www.cdc.gov/vaccines/hcp/vis/) World Health Organization Fact Sheets (http://www.who.int/mediacentre/factsheets/en/) National Department of Health, South Africa (http://www.health.gov.za/) Disclaimer This book is intended as an educational tool only. Information may be subject to change as schedules or formulations are updated. Summarized and simplified information is presented in this booklet. For full prescribing information and contraindications for vaccinations, please consult individual package inserts. There has been -
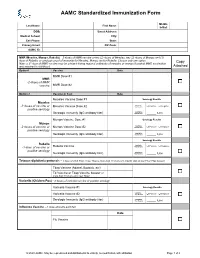
AAMC Standardized Immunization Form
AAMC Standardized Immunization Form Middle Last Name: First Name: Initial: DOB: Street Address: Medical School: City: Cell Phone: State: Primary Email: ZIP Code: AAMC ID: MMR (Measles, Mumps, Rubella) – 2 doses of MMR vaccine or two (2) doses of Measles, two (2) doses of Mumps and (1) dose of Rubella; or serologic proof of immunity for Measles, Mumps and/or Rubella. Choose only one option. Copy Note: a 3rd dose of MMR vaccine may be advised during regional outbreaks of measles or mumps if original MMR vaccination was received in childhood. Attached Option1 Vaccine Date MMR Dose #1 MMR -2 doses of MMR vaccine MMR Dose #2 Option 2 Vaccine or Test Date Measles Vaccine Dose #1 Serology Results Measles Qualitative -2 doses of vaccine or Measles Vaccine Dose #2 Titer Results: Positive Negative positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Mumps Vaccine Dose #1 Serology Results Mumps Qualitative -2 doses of vaccine or Mumps Vaccine Dose #2 Titer Results: Positive Negative positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Serology Results Rubella Qualitative Positive Negative -1 dose of vaccine or Rubella Vaccine Titer Results: positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Tetanus-diphtheria-pertussis – 1 dose of adult Tdap; if last Tdap is more than 10 years old, provide date of last Td or Tdap booster Tdap Vaccine (Adacel, Boostrix, etc) Td Vaccine or Tdap Vaccine booster (if more than 10 years since last Tdap) Varicella (Chicken Pox) - 2 doses of varicella vaccine or positive serology Varicella Vaccine #1 Serology Results Qualitative Varicella Vaccine #2 Titer Results: Positive Negative Serologic Immunity (IgG antibody titer) Quantitative Titer Results: _____ IU/ml Influenza Vaccine --1 dose annually each fall Date Flu Vaccine © 2020 AAMC. -

Recommended Adult Immunization Schedule
Recommended Adult Immunization Schedule UNITED STATES for ages 19 years or older 2021 Recommended by the Advisory Committee on Immunization Practices How to use the adult immunization schedule (www.cdc.gov/vaccines/acip) and approved by the Centers for Disease Determine recommended Assess need for additional Review vaccine types, Control and Prevention (www.cdc.gov), American College of Physicians 1 vaccinations by age 2 recommended vaccinations 3 frequencies, and intervals (www.acponline.org), American Academy of Family Physicians (www.aafp. (Table 1) by medical condition and and considerations for org), American College of Obstetricians and Gynecologists (www.acog.org), other indications (Table 2) special situations (Notes) American College of Nurse-Midwives (www.midwife.org), and American Academy of Physician Assistants (www.aapa.org). Vaccines in the Adult Immunization Schedule* Report y Vaccines Abbreviations Trade names Suspected cases of reportable vaccine-preventable diseases or outbreaks to the local or state health department Haemophilus influenzae type b vaccine Hib ActHIB® y Clinically significant postvaccination reactions to the Vaccine Adverse Event Hiberix® Reporting System at www.vaers.hhs.gov or 800-822-7967 PedvaxHIB® Hepatitis A vaccine HepA Havrix® Injury claims Vaqta® All vaccines included in the adult immunization schedule except pneumococcal 23-valent polysaccharide (PPSV23) and zoster (RZV) vaccines are covered by the Hepatitis A and hepatitis B vaccine HepA-HepB Twinrix® Vaccine Injury Compensation Program. Information on how to file a vaccine injury Hepatitis B vaccine HepB Engerix-B® claim is available at www.hrsa.gov/vaccinecompensation. Recombivax HB® Heplisav-B® Questions or comments Contact www.cdc.gov/cdc-info or 800-CDC-INFO (800-232-4636), in English or Human papillomavirus vaccine HPV Gardasil 9® Spanish, 8 a.m.–8 p.m. -

Kids and COVID 19 Vaccination
Kids and COVID 19 Vaccination Q: What COVID vaccine is available for kids right now? A: Pfizer-BioNTech mRNA vaccine was initially approved for individuals to as young as age 16 years old and was recently approved for kids as young as 12 years of age. This is a two shot series, given 3 weeks apart. The dose is the same as that given to adults. Q: When will younger kids be able to get vaccinated? A: Pfizer is currently conducting studies on kids as young as 6 months of age. Moderna is in the process of submitting data on their studies for ages 12-18 yrs and have started the process for studies in younger kids. Many reputable sources suggest that kids as young as 6yrs of age may be eligible for vaccination this fall. Keep checking our website for more information on timeline and details for vaccination in younger kids and infants. Q: How do I get my child vaccinated? A: In North Idaho, there are many locations that offer the Pfizer vaccine. Many local pharmacies have the Pfizer vaccine available (check online or in person to ensure they have Pfizer brand available for those under age 18 yrs). The local Walgreen’s pharmacies are offering Pfizer vaccine and appointments can be scheduled at www.walgreens.com. Other option: www.panhandlehealth.org. This site will allow you to make appointments at the Kootenai county fairgrounds site as well as other sites in adjacent counties. We are not yet offering COVID vaccination at Coeur d’Alene Pediatrics, but hope to be able to do so in the future. -

Varicella (Chickenpox): Questions and Answers Q&A Information About the Disease and Vaccines
Varicella (Chickenpox): Questions and Answers Q&A information about the disease and vaccines What causes chickenpox? more common in infants, adults, and people with Chickenpox is caused by a virus, the varicella-zoster weakened immune systems. virus. How do I know if my child has chickenpox? How does chickenpox spread? Usually chickenpox can be diagnosed by disease his- Chickenpox spreads from person to person by direct tory and appearance alone. Adults who need to contact or through the air by coughing or sneezing. know if they’ve had chickenpox in the past can have It is highly contagious. It can also be spread through this determined by a laboratory test. Chickenpox is direct contact with the fluid from a blister of a per- much less common now than it was before a vaccine son infected with chickenpox, or from direct contact became available, so parents, doctors, and nurses with a sore from a person with shingles. are less familiar with it. It may be necessary to perform laboratory testing for children to confirm chickenpox. How long does it take to show signs of chickenpox after being exposed? How long is a person with chickenpox contagious? It takes from 10 to 21 days to develop symptoms after Patients with chickenpox are contagious for 1–2 days being exposed to a person infected with chickenpox. before the rash appears and continue to be conta- The usual time period is 14–16 days. gious through the first 4–5 days or until all the blisters are crusted over. What are the symptoms of chickenpox? Is there a treatment for chickenpox? The most common symptoms of chickenpox are rash, fever, coughing, fussiness, headache, and loss of appe- Most cases of chickenpox in otherwise healthy children tite. -

An Insight Into the Immunization Coverage for Combined Vaccines in Albania
European Scientific Journal January 2017 edition vol.13, No.3 ISSN: 1857 – 7881 (Print) e - ISSN 1857- 7431 An Insight into the Immunization Coverage for Combined Vaccines in Albania Eftiola Pojani, PhD Student Department of Pharmaceutical Sciences Catholic University “Our Lady of Good Counsel” Tirana, Albania Erida Nelaj, PhD Department of Epidemiology and Infectious Disease Control Institute of Public Health, Tirana, Albania Alban Ylli, Associate Prof. University of Medicine, Faculty of Public Health, Tirana, Albania doi: 10.19044/esj.2016.v13n3p33 URL:http://dx.doi.org/10.19044/esj.2016.v13n3p33 Abstract We aim to Provide facts about the vaccination coverage for combined vaccines such as MMR (Measles, Mumps and Rubella) and DTP (Diphtheria, Tetanus, Pertussis) containing vaccines in use in Albania during the last 10 years, in order to confirm the stability of immunization program. One of the reasons for coverage improvement is the use of one or two-dose vials for the administration of MMR vaccine, enabling the vaccination of children at any time, without the need of gathering them on certain days when the multi-dose vaccine vials are opened. In the last three years, according to WHO recommendations, it was noted that vaccination coverage with three doses containing DTP is at sustainable levels, always above 95%, but for our country this value goes sometimes above 98%. As for the use of pentavalent vaccine, from 2008, DTP-HepB-Hib, also the coverage of Hepatitis b vaccine is always at upper levels due to its use on 5 in 1 combination. The application of this vaccine was associated with the use of one dose vial administration, therefore one vaccine to a child. -

Vaccine Information Program
1 How to access translation • On the toolbar at the bottom of your screen, select the “Interpretation” button. • Select the language you would like to listen to. Date Date • How to access Spanish and Portuguese translation • Introduction of panel • Overview of local data • How vaccines work • Vaccine development and safety • Side effects Welcome! • State vaccination strategy • Vaccine priority groups • After vaccination • Where to get vaccinated • Support for accessing vaccines • Q & A 3 Why I am choosing vaccination • Mayor Edward Bettencourt Why I am choosing vaccination • Dr. Alain Chaoui, MD • I chose to get the vaccine to protect my patients, my family and my community and to take part in eradicating Covid-19. Why I am choosing vaccination Dr. Laura Holland, MD • I took the vaccine because I researched the safety and concluded that it was safe and effective. • I took it to protect myself, my family and my patients. 7 Why I am choosing vaccination • Grace Martins, RN • I decided to get the vaccine because as a Registered Nurse I am in constant contact with patients and staff. I am also in the high risk category because I am a diabetic on Insulin. • For my own health and the safety of those I take care of, I decided I needed to be protected from this virus! Why I am choosing vaccination • Cari Pacheco, LPN • I had no doubts about getting the vaccine due to the love I have for my family, friends, people in the community and my work environment. • I need to protect myself in order to be able to care for others • I want to protect myself as much as I can so I will not bring any illnesses to the people I love.