Material Safety Data Sheet
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
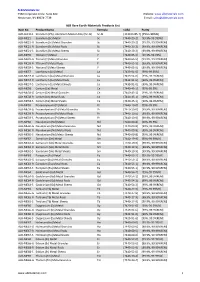
ALB Materials Inc Product List.Xlsx
ALB Materials Inc 2360 Corporate Circle. Suite 400 Website: www.albmaterials.com Henderson, NV 89074-7739 E-mail: [email protected] ALB Rare Earth Materials Products List Item No. Product Name Formula CAS# Purity ALB-AL2113 Scandium (2%)- Aluminum Master Alloy (Sc-Al) Sc-Al [113413-85-7] [2%Sc+98%Al] ALB-ME21 Scandium (Sc) Metal Sc [7440-20-2] [99.9%-99.999%] ALB-ME21-G Scandium (Sc) Metal Granules Sc [7440-20-2] [99.9%, 99.99%REM] ALB-ME21-R Scandium (Sc) Metal Rods Sc [7440-20-2] [99.9%, 99.99%REM] ALB-ME21-S Scandium (Sc) Metal Sheets Sc [7440-20-2] [99.9%, 99.99%REM] ALB-ME39 Yttrium (Y) Metal Y [7440-65-5] [99.9%-99.99%] ALB-ME39-G Yttrium (Y) Metal Granules Y [7440-65-5] [99.9%, 99.99%REM] ALB-ME39-R Yttrium (Y) Metal Rods Y [7440-65-5] [99.9%, 99.99%REM] ALB-ME39-S Yttrium (Y) Metal Sheets Y [7440-65-5] [99.9%, 99.99%REM] ALB-ME57 Lanthanum (La) Metal La [7439-91-0] [99%-99.95%] ALB-ME57-G Lanthanum (La) Metal Granules La [7439-91-0] [99%, 99.9%REM] ALB-ME57-R Lanthanum (La) Metal Rods La [7439-91-0] [99%, 99.9%REM] ALB-ME57-S Lanthanum (La) Metal Sheets La [7439-91-0] [99%, 99.9%REM] ALB-ME58 Cerium (Ce) Metal Ce [7440-45-1] [99%-99.9%] ALB-ME58-G Cerium (Ce) Metal Granules Ce [7440-45-1] [99%, 99.9%REM] ALB-ME58-R Cerium (Ce) Metal Rods Ce [7440-45-1] [99%, 99.9%REM] ALB-ME58-S Cerium (Ce) Metal Sheets Ce [7440-45-1] [99%, 99.9%REM] ALB-ME59 Praseodymium (Pr) Metal Pr [7440-10-0] [99%-99.9%] ALB-ME59-G Praseodymium (Pr) Metal Granules Pr [7440-10-0] [99.9%, 99.99%REM] ALB-ME59-R Praseodymium (Pr) Metal Rods Pr [7440-10-0] -

A Study of the Mechanical Properties of Silicon-Based Thin Films
MECHANICAL PROPERTIES OF SILICON-BASED FILMS FABRICATED BY PECVD A STUDY OF THE MECHANICAL PROPERTIES OF SILICON-BASED THIN FILMS DEPOSITED BY ECR-PECVD AND ICP-CVD By OWEN TAGGART, B.ENG. A Thesis Submitted to the School of Graduate Studies in Partial Fulfillment of the Requirements for the Degree Master of Applied Science McMaster University © Copyright by Owen Taggart, April 2013 McMaster University MASTER OF APPLIED SCIENCE (2013) Hamilton, Ontario (Engineering Physics) Title: A Study of the Mechanical Properties of Silicon-Based Thin Films Deposited by ECR-PECVD and ICP-CVD AUTHOR: Owen Taggart SUPERVISOR: Professor Peter Mascher Number of pages: xv, 151 ii Abstract Silicon-based dielectric thin films including amorphous hydrogenated aluminium-doped silicon oxides (a-SiAlxOy:H), amorphous hydrogenated silicon nitrides (a-SiNx:H), and amorphous hydrogenated silicon carbides (a-SiCx:H) were deposited by remote plasma chemical vapour deposition (RPECVD) techniques including electron cyclotron resonance plasma enhanced chemical vapour deposition (ECR-PECVD) and inductively-coupled-plasma chemical vapour deposition (ICP-CVD) on silicon (Si) wafers, soda-lime glass microscope slides, and glassy carbon (C) plates. Aluminium (Al) in the SiAlO films was incorporated by way of a metalorganic Al(TMHD)3 precursor. Thickness, refractive index, and growth rate of the films were measured using variable angle spectroscopic ellipsometry (VASE). Film composition was measured using energy dispersive X-ray spectroscopy (EDX) for the SiAlO films and Rutherford backscattering spectrometry (RBS) for the SiCx films. Elastic modulus and hardness of the SiAlO and SiCx films were measured using nanoindentation and their adhesion was characterized via progressive load scratch testing. -

Solvo-Hydrothermal Growth and Photoluminescence Studies of Micro and Nano Structured Zinc Sulfide for Bio-Imaging Applications
Solvo-hydrothermal growth and photoluminescence studies of micro and nano structured Zinc Sulfide for bio-imaging applications Thesis submitted to COCHIN UNIVERSITY OF SCIENCE AND TECHNOLOGY in partial fulfillment of the requirements for the award of the degree of DOCTOR OF PHILOSOPHY in Physics by Sajan P. Nano Functional Materials Lab Department of Physics Cochin University of Science and Technology Cochin - 682 022, Kerala, India June 2016 Solvo-hydrothermal growth and photoluminescence studies of micro and nano structured Zinc Sulfide for bio-imaging applications Ph.D. thesis in the field of Physics Author: Sajan P. Nano Functional Materials Lab Department of Physics Cochin University of Science and Technology Cochin - 682 022, Kerala, India. Email: [email protected] Supervisor: Dr. M. Junaid Bushiri Professor Nano Functional Materials Lab Department of Physics Cochin University of Science and Technology Cochin-682 022, Kerala, India. Email: [email protected] June 2016 Dedicated to my mother Department of Physics Cochin University of Science and Technology Cochin – 682 022 Dr. M. Junaid Bushiri Professor Department of Physics Cochin University of Science and Technology Cochin 682 022, India. Certified that the work presented in this thesis entitled “Solvo-hydrothermal growth and photoluminescence studies of micro and nano structured Zinc Sulfide for bio- imaging applications” is based on the bonafide record of research work done by Sajan P., under my guidance in the Department of Physics, Cochin University of Science and Technology, Cochin 682 022 in partial fulfillment of the requirements for the award of degree of Doctor of Philosophy and has not been included in any other thesis submitted for the award of any degree. -

Development of Nanostructure Photocatalysts Based Perovskite and Carbon Nitride Materials for CO2 Reduction and H2 Production Using Solar Energy
Development of nanostructure photocatalysts based perovskite and carbon nitride materials for CO2 reduction and H2 production using solar energy Thèse Nhu Nang Vu Doctorat en génie chimique Philosophiæ doctor (Ph. D.) Québec, Canada © Nhu Nang Vu, 2020 Development of nanostructure photocatalysts based perovskite and carbon nitride materials for CO2 reduction and H2 production using solar energy Thèse Nhu-Nang Vu Sous la direction de : Trong-On Do, directeur de recherche Serge Kaliaguine, codirecteur de recherche Résumé Cette thèse passe en revue la production de carburant à partir d’énergie solaire de H2O et de CO2 à l'aide d'un photocatalyseur, ce qui est considéré comme l'une des solutions les plus prometteuses pour la lutte contre le réchauffement climatique et la crise du carburant. Cette thèse propose quatre nouvelles approches pour développer des photocatalyseurs nanostructures efficaces pour la réduction photocatalytique du CO2 et la production de H2. L’absorption de la lumière, la séparation des charges et les réactions de surface sont des aspects critiques qui ont un impact énorme sur la photoréduction du CO2 et la production photocatalytique de H2. Le g-C3N4 et les matériaux pérovskites sont des candidats appropriés pour ces processus, car ils offrent des caractéristiques structurelles et des propriétés exceptionnelles. Un grand nombre de photocatalyseurs nanostructures sont actuellement développés pour la photoréduction du CO2 et la production photocatalytique de H2. Les nanostructures 2D et les nanocomposites hétérostructures sont largement étudiés en raison de leurs excellentes propriétés telles que la séparation efficace et la longue durée de vie des porteurs de charge. De manière prometteuse, les nanostructures 2D et les nanocomposites des matériaux g-C3N4 et pérovskites présentent d'excellentes performances photocatalytiques, selon la littérature scientifique. -

(12) United States Patent (10) Patent No.: US 8,940,855 B2 Hedricket Al
USOO8940855B2 (12) United States Patent (10) Patent No.: US 8,940,855 B2 Hedricket al. (45) Date of Patent: Jan. 27, 2015 (54) POLYMERS BEARING PENDANT (56) References Cited PENTAFLUOROPHENYL ESTER GROUPS, AND METHODS OF SYNTHESIS AND U.S. PATENT DOCUMENTS FUNCTIONALIZATION THEREOF 3,274,214. A 9, 1966 Prochaska 4,102.912 A 7, 1978 Carr 4,515,712 A 5/1985 Wiegers et al. (71) Applicant: International Business Machines 4,613,442 A 9, 1986 Yuki et al. Corporation, Armonk, NY (US) 4,782,131 A 1 1/1988 Sweeney 4,831,100 A 5/1989 Komatsu et al. 4,888.410 A 12, 1989 Komatsu et al. (72) Inventors: James L. Hedrick, Pleasanton, CA 5,091,543 A 2/1992 Grey (US); Alshakim Nelson, Fremont, CA 5,357,027 A 10, 1994 Komatsu (US); Daniel P. Sanders, San Jose, CA 5,424.473 A 6/1995 Galvan et al. (US) 5,523,399 A 6/1996 Asaka et al. 5,869,697 A 2f1999 Bhushan et al. 6,300,458 B1 10/2001 Vandenberg (73) Assignee: International Business Machines 6,664,372 B1 12/2003 Janda et al. Corporation, Armonk, NY (US) 8,013,065 B2 9, 2011 Hedricket al. 8,044,194 B2 10/2011 Dubois et al. 8, 158,281 B2 4/2012 Ishihara et al. (*) Notice: Subject to any disclaimer, the term of this 2007, OO15932 A1 1/2007 Fujita et al. 2007/0232751 A1 10/2007 Ludewig et al. patent is extended or adjusted under 35 2007,0298.066 A1 12/2007 Alferiev et al. -

Nomenclature of Inorganic Chemistry (IUPAC Recommendations 2005)
NOMENCLATURE OF INORGANIC CHEMISTRY IUPAC Recommendations 2005 IUPAC Periodic Table of the Elements 118 1 2 21314151617 H He 3 4 5 6 7 8 9 10 Li Be B C N O F Ne 11 12 13 14 15 16 17 18 3456 78910 11 12 Na Mg Al Si P S Cl Ar 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe 55 56 * 57− 71 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 Cs Ba lanthanoids Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn 87 88 ‡ 89− 103 104 105 106 107 108 109 110 111 112 113 114 115 116 117 118 Fr Ra actinoids Rf Db Sg Bh Hs Mt Ds Rg Uub Uut Uuq Uup Uuh Uus Uuo * 57 58 59 60 61 62 63 64 65 66 67 68 69 70 71 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu ‡ 89 90 91 92 93 94 95 96 97 98 99 100 101 102 103 Ac Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr International Union of Pure and Applied Chemistry Nomenclature of Inorganic Chemistry IUPAC RECOMMENDATIONS 2005 Issued by the Division of Chemical Nomenclature and Structure Representation in collaboration with the Division of Inorganic Chemistry Prepared for publication by Neil G. -

Recycling of Neodymium and Dysprosium from Permanent Magnets
University of Pennsylvania ScholarlyCommons Department of Chemical & Biomolecular Senior Design Reports (CBE) Engineering 4-2016 Recycling of Neodymium and Dysprosium from Permanent Magnets Alan X. Dai University of Pennsylvania, [email protected] Connor A. Lippincott University of Pennsylvania, [email protected] Michael E. Nissan University of Pennsylvania, [email protected] Richard Shim University of Pennsylvania Follow this and additional works at: https://repository.upenn.edu/cbe_sdr Part of the Biochemical and Biomolecular Engineering Commons Dai, Alan X.; Lippincott, Connor A.; Nissan, Michael E.; and Shim, Richard, "Recycling of Neodymium and Dysprosium from Permanent Magnets" (2016). Senior Design Reports (CBE). 81. https://repository.upenn.edu/cbe_sdr/81 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/cbe_sdr/81 For more information, please contact [email protected]. Recycling of Neodymium and Dysprosium from Permanent Magnets Abstract This project seeks to recycle neodymium and dysprosium from used neodymium-iron-boron, NdFeB, permanent magnets. Our alternative recycling strategy is based on a method developed by Dr. Eric Schelter of the University of Pennsylvania Department of Chemistry and his research group. This process involves the use of a novel tripodal nitroxide ligand, H3TriNOX. The proposed process design converts used NdFeB magnets to neodymium and dysprosium. The goal production of neodymium oxide is 126,000 kg/year and of dysprosium oxide is 14,000 kg/year, which would cost $12,500,000 with the current price rate of neodymium and dysprosium. Based on the results of the economic analysis, this project would be unprofitable in the near future. Disciplines Biochemical and Biomolecular Engineering | Chemical Engineering | Engineering This working paper is available at ScholarlyCommons: https://repository.upenn.edu/cbe_sdr/81 Professor Leonard Fabiano Dr. -

Memoria Congreso Xxvi
Sociedad Mexicana de Ciencia y Tecnología de Superficies y Materiales SMCTSM XXVI Congreso Nacional 25 al 29 de Septiembre Puebla, Pue, México 2006 Libro de Resúmenes h t t p : / / w w w . s m c s y v . o r g . m x / Sociedad Mexicana de Ciencia y Tecnología de Superficies y Materiales (SMCTSM) XXVI Congreso Nacional del 25 al 29 de Septiembre Puebla, Pue, México 2006 h t t p : / / w w w . s m c s y v . o r g . m x / Comité Organizador Organizing Committee Wilfrido Calleja Arriaga INAOE Presidente [email protected] Jesús Carrillo López CIDS- ICUAP Secretario [email protected] José Martín Yáñez CINVESTAV-Qro. Tesorero [email protected] Enrique Camps Carvajal ININ Vocal de Congresos [email protected] Víctor Hugo Méndez IICO-UASLP Vocal de Cursos [email protected] Gerardo Contreras Puente ESFM-IPN Vocal de publicaciones [email protected] Alberto Herrera Gómez Presidente pasado CINVESTAV-Querétaro [email protected] Comité Organizador Local Local Organizing Committee Nicolás Atenco (Presidente) UABJO Sara Carriillo Uribe UABJO Rubén Doroteo Castillejos UABJO Dante Arias Torres UABJO José Roberto Nicolás Carlock UABJO Wilfrido Calleja Arriaga INAOE Jesús Carrillo López CIDS-BUAP Aurelio Horacio Heredia UAPEP Salvador Alcántara Iniesta CIDS-BUAP Francisco Renero Carrillo INAOE S M C T S M Mensaje XXVI Congreso Nacional A nombre del Comité de Organización Local de la ciudad de Puebla y de la Mesa Directiva de la SMCTSM, damos la bienvenida a todos los participantes y les deseamos una semana de trabajo fructífero en esta bella, colonial y hospitalaria ciudad de Puebla. -
Nanoscale Heterogeneities in Visible Light Absorbing Photocatalysts
Nanoscale Heterogeneities in Visible Light Absorbing Photocatalysts: Connecting Structure to Functionality Through Electron Microscopy and Spectroscopy by Diane Michelle Haiber A Dissertation Presented in Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy Approved November 2019 by the Graduate Supervisory Committee: Peter Crozier, Chair Candace Chan Jingyue Liu Michael Treacy ARIZONA STATE UNIVERSITY December 2019 ABSTRACT Photocatalytic water splitting over suspended nanoparticles represents a potential solution for achieving CO2-neutral energy generation and storage. To design efficient photocatalysts, a fundamental understanding of the material’s structure, electronic properties, defects, and how these are controlled via synthesis is essential. Both bulk and nanoscale materials characterization, in addition to various performance metrics, can be combined to elucidate functionality at multiple length scales. In this work, two promising visible light harvesting systems are studied in detail: Pt-functionalized graphitic carbon nitrides (g-CNxHys) and TiO2-supported CeO2-x composites. Electron energy-loss spectroscopy (EELS) is used to sense variations in the local concentration of amine moieties (defects believed to facilitate interfacial charge transfer) at the surface of a g-CNxHy flake. Using an aloof-beam configuration, spatial resolution is maximized while minimizing damage thus providing nanoscale vibrational fingerprints similar to infrared absorption spectra. Structural disorder in g-CNxHys is further studied using transmission electron microscopy at low electron fluence rates. In-plane structural fluctuations revealed variations in the local azimuthal orientation of the heptazine building blocks, allowing planar domain sizes to be related to the average polymer chain length. Furthermore, competing factors regulating photocatalytic performance in a series of Pt/g- CNxHys is elucidated. -

(12) Patent Application Publication (10) Pub. No.: US 2013/0251942 A1 Azimi Et Al
US 2013025 1942A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2013/0251942 A1 Azimi et al. (43) Pub. Date: Sep. 26, 2013 (54) HYDROPHOBIC MATERIALS C04B 35/622 (2006.01) NCORPORATING RARE EARTHELEMENTS B22F 3/12 (2006.01) AND METHODS OF MANUFACTURE BOSD5/00 (2006.01) B32B I5/00 (2006.01) (76) Inventors: Gisele Azimi, Waltham, MA (US); B32B33/00 (2006.01) Kripa K. Varanasi, Lexington, MA (52) U.S. Cl. (US); Rajeev Dhiman, Malden, MA USPC ........... 428/141; 428/689: 428/698; 428/702: (US); Adam T. Paxson, Cambridge, MA 428/336; 41.9/39: 420/416: 501/152 (US); Kyukmin Kwon, Cambridge, MA (US) (57) ABSTRACT This invention relates generally to an article that includes a (21) Appl. No.: 13/428,652 base Substrate and a hydrophobic coating on the base Sub strate, wherein the hydrophobic coating includes a rare earth (22) Filed: Mar. 23, 2012 element material (e.g., a rare earth oxide, a rare earth carbide, a rare earth nitride, a rare earth fluoride, and/or a rare earth Publication Classification boride). An exposed Surface of the hydrophobic coating has a dynamic contactangle with water of at least about 90 degrees. (51) Int. Cl. A method of manufacturing the article includes providing the B32B 9/00 (2006.01) base Substrate and forming a coating on the base Substrate B32B 8/00 (2006.01) (e.g., through sintering or Sputtering). Patent Application Publication Sep. 26, 2013 Sheet 1 of 13 US 2013/0251942 A1 FIG. FIG. 2 Patent Application Publication Sep. 26, 2013 Sheet 2 of 13 US 2013/0251942 A1 300 302 304 FIG. -

Technological Gaps Hindering Uptake of Crms Substitution in Industrial Application
SCRREEN Coordination and Support Action (CSA) This project has received funding from the European Union's Horizon 2020 research and innovation programme under grant agreement No 730227. Start date : 2016-11-01 Duration : 38 Months www.scrreen.eu Technological gaps hindering uptake of CRMs substitution in industrial application Authors : Dr. Rocio BARROS (ICCRAM), Yongxiang Yang (TuDelf), Etienne Bouyer (CEA), Marjaana Karhu, Elina Yli-Rantala, Elina Huttunen Saarivirta (VTT), Michael Samouhos (NTUA), Lena Sundwvist, Xianfeng Hu (MEFOS) SCRREEN - D6.3 - Issued on 2019-11-25 14:45:41 by ICCRAM SCRREEN - D6.3 - Issued on 2019-11-25 14:45:41 by ICCRAM SCRREEN - Contract Number: 730227 Project officer: Jonas Hedberg Document title Technological gaps hindering uptake of CRMs substitution in industrial application Dr. Rocio BARROS, Yongxiang Yang (TuDelf), Etienne Bouyer (CEA), Marjaana Karhu, Elina Author(s) Yli-Rantala, Elina Huttunen Saarivirta (VTT), Michael Samouhos (NTUA), Lena Sundwvist, Xianfeng Hu (MEFOS) Number of pages 205 Document type Deliverable Work Package WP6 Document number D6.3 Issued by ICCRAM Date of completion 2019-11-25 14:45:41 Dissemination level Public Summary Technological gaps hindering uptake of CRMs substitution in industrial application Approval Date By 2019-11-29 14:54:36 Mrs. Maria TAXIARCHOU (LabMet, NTUA) 2019-12-02 07:30:59 Mr. Stéphane BOURG (CEA) SCRREEN - D6.3 - Issued on 2019-11-25 14:45:41 by ICCRAM SCRREEN Coordination and Support Actions (Coordinating) (CSA-CA) Co-funded by the European Commission under -

Coordination Chemistry Reviews 257 (2013) 2032–2062
Coordination Chemistry Reviews 257 (2013) 2032–2062 Contents lists available at SciVerse ScienceDirect Coordination Chemistry Reviews journa l homepage: www.elsevier.com/locate/ccr Review Tri-s-triazines (s-heptazines)—From a “mystery molecule” to industrially relevant carbon nitride materials ∗ Anke Schwarzer, Tatyana Saplinova, Edwin Kroke Institut für Anorganische Chemie, TU Bergakademie Freiberg, Leipziger Straße 29, 09596 Freiberg, Germany Contents 1. Introduction and historical overview . 2033 1.1. The heterocyclic systems s-triazine C3N3 and s-heptazine C6N7 – a brief comparison . 2033 1.2. Heptazines – a historical sketch. 2034 2. Melem, and melemium salts . 2036 2.1. Melem . 2036 2.1.1. Formation . 2036 2.1.2. Melem – structure and properties. 2038 2.2. Melemium salts . 2040 3. s-Heptazine-based salts and acids . 2041 3.1. Cyameluric acid and cyamelurates . 2041 3.2. Preparation . 2041 3.2.1. Physical and chemical properties . 2041 3.2.2. Thermal stability and spectroscopic properties . 2042 3.2.3. Applications. 2042 3.3. Melonates and hydromelonic acid . 2042 3.3.1. Introduction . 2042 3.3.2. Preparation . 2043 3.3.3. Physical properties . 2043 3.3.4. Vibrational and NMR spectroscopy . 2043 3.3.5. Crystal structures . 2043 3.3.6. Applications. 2045 4. Other molecular tri-s-triazine derivatives . 2045 4.1. C6N7H3 – the parent s-heptazine. 2045 4.2. Cyameluric halides – tri-chloro-tri-s-triazine . 2045 4.3. The azide C6N7(N3)3 . 2046 4.4. C6N7R3 – alkyl- and aryl s-heptazine derivatives . 2047 4.5. Symmetric and asymmetric cyameluric esters . 2047 4.6. Symmetric and asymmetric cyameluric amides . 2048 4.7.