Fenoxaprop-P-Ethyl Susceptibility and Mutation Point Detection of Acetyl-Coa
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Historical Study on the Relation Between Ancient Chinese Cuju and Modern Football
2018 4th International Conference on Innovative Development of E-commerce and Logistics (ICIDEL 2018) Historical Study on the Relation between Ancient Chinese Cuju and Modern Football Xiaoxue Liu1, Yanfen Zhang2, and Xuezhi Ma3 1Department of Physical Education, China University of Geosciences, Xueyuan Road, Haidian District, Beijing, P. R. China 2Department of Life Sciences; Xinxiang University, Xinxiang Henan Province, Eastern Section of Hua Lan Road, Hongqi District, Xinxiang City, Henan, China 3Beijing Sport University Wushu School, Information Road, Haidian District, Beijing, China [email protected], [email protected], [email protected] Keywords: Ancient Chinese Cuju, Modern Football, Relationship, Development, The Same Origin Abstract: This paper studies on the origin and development of Chinese Cuju through document retrieval. Born in the period of Dongyi civilization, Chinese Cuju began to take shape during the Spring and Autumn and Warring States Period, and gradually flourished during the Qin, Han, Tang and Song dynasties. Through the economic and cultural exchange between China and the West in the past ages, Cuju was introduced into Europe when Mongol expedited westward in Yuan Dynasty. Finally, it has become the modern football, which originated from ancient Chinese Cuju and developed from European competition rules and now is widely accepted and popular in the world. 1. The Cultural Background of the Study On July 15th, 2004, Mr. Blatter, the president of FIFA (Fédération Internationale de Football Association) officially announced in the 3rd session of Soccerex Fair, that football originated in Zibo, the capital of Qi State during the Spring and Autumn Period of ancient China. Cuju (ancient football game) began in China, while modern football (eleven -player game) originated in England. -

Download This Article PDF Format
RSC Advances View Article Online PAPER View Journal | View Issue Panax notoginseng saponins radiosensitize colorectal cancer cells by regulating the SNHG6/ Cite this: RSC Adv.,2019,9,38558 miR-137 axis Caihui Xu,a Teng Liu,b Haiyan Liu,a Gongbin Chena and Yinmou Guo *a Panax notoginseng saponins (PNS) have recently attracted great attention for their anti-cancer activity in colorectal cancer (CRC). The aim of this study was to explore the functional role and underlying mechanisms of PNS on CRC radiosensitivity. Cell viability was assessed by a Cell Counting kit-8 assay. Cell survival and apoptosis were determined using colony formation assay and flow cytometry, respectively. Quantitative real-time PCR was used to quantify the levels of SNHG6 and miR-137. The targeted correlation between SNHG6 and miR-137 was validated by dual-luciferase reporter and RNA immunoprecipitation assays. Our data supported that PNS weakened the viability of CRC cells. Moreover, PNS promoted the radiosensitivity of CRC cells. Mechanistically, PNS enhanced CRC cell radiosensitivity Creative Commons Attribution-NonCommercial 3.0 Unported Licence. by upregulating SNHG6. SNHG6 directly targeted miR-137 and inhibited miR-137 expression. MiR-137 Received 20th September 2019 was involved in the regulatory effect of SNHG6 on CRC cell radiosensitivity. Furthermore, PNS increased Accepted 11th November 2019 miR-137 expression through SNHG6 in CRC cells. Our study suggested that PNS promoted DOI: 10.1039/c9ra07622k radiosensitivity in CRC cells at least partly through regulating the SNHG6/miR-137 axis, providing a novel rsc.li/rsc-advances understanding of the anti-cancer mechanism of PNS in CRC. -
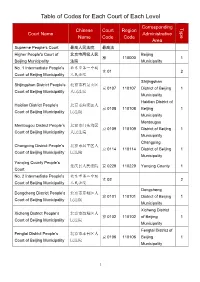
Table of Codes for Each Court of Each Level
Table of Codes for Each Court of Each Level Corresponding Type Chinese Court Region Court Name Administrative Name Code Code Area Supreme People’s Court 最高人民法院 最高法 Higher People's Court of 北京市高级人民 Beijing 京 110000 1 Beijing Municipality 法院 Municipality No. 1 Intermediate People's 北京市第一中级 京 01 2 Court of Beijing Municipality 人民法院 Shijingshan Shijingshan District People’s 北京市石景山区 京 0107 110107 District of Beijing 1 Court of Beijing Municipality 人民法院 Municipality Haidian District of Haidian District People’s 北京市海淀区人 京 0108 110108 Beijing 1 Court of Beijing Municipality 民法院 Municipality Mentougou Mentougou District People’s 北京市门头沟区 京 0109 110109 District of Beijing 1 Court of Beijing Municipality 人民法院 Municipality Changping Changping District People’s 北京市昌平区人 京 0114 110114 District of Beijing 1 Court of Beijing Municipality 民法院 Municipality Yanqing County People’s 延庆县人民法院 京 0229 110229 Yanqing County 1 Court No. 2 Intermediate People's 北京市第二中级 京 02 2 Court of Beijing Municipality 人民法院 Dongcheng Dongcheng District People’s 北京市东城区人 京 0101 110101 District of Beijing 1 Court of Beijing Municipality 民法院 Municipality Xicheng District Xicheng District People’s 北京市西城区人 京 0102 110102 of Beijing 1 Court of Beijing Municipality 民法院 Municipality Fengtai District of Fengtai District People’s 北京市丰台区人 京 0106 110106 Beijing 1 Court of Beijing Municipality 民法院 Municipality 1 Fangshan District Fangshan District People’s 北京市房山区人 京 0111 110111 of Beijing 1 Court of Beijing Municipality 民法院 Municipality Daxing District of Daxing District People’s 北京市大兴区人 京 0115 -

Announcement of Annual Results for the Year Ended December 31, 2019
Hong Kong Exchanges and Clearing Limited and The Stock Exchange of Hong Kong Limited take no responsibility for the contents of this announcement, make no representation as to its accuracy or completeness and expressly disclaim any liability whatsoever for any loss howsoever arising from or in reliance upon the whole or any part of the contents of this announcement. (Stock Code of H Shares: 1216) (Stock Code of Preference Shares: 4617) ANNOUNCEMENT OF ANNUAL RESULTS FOR THE YEAR ENDED DECEMBER 31, 2019 The board of directors (the “Board”) of Zhongyuan Bank Co., Ltd. (the “Bank”) is pleased to announce the audited consolidated annual results (the “Annual Results”) of the Bank and its subsidiaries for the year ended December 31, 2019 (the “Reporting Period”) which were prepared in accordance with the International Financial Reporting Standards (“IFRSs”). The Board and the audit committee of the Board have reviewed and confirmed the Annual Results. This results announcement is published on the websites of The Stock Exchange of Hong Kong Limited (www.hkexnews.hk) and the Bank (www.zybank.com.cn). The annual report for the year ended December 31, 2019 will be despatched to the shareholders of the Bank and will be available on the above websites in due course. On behalf of the Board Zhongyuan Bank Co., Ltd.* DOU Rongxing Chairman Zhengzhou, the People’s Republic of China March 27, 2020 As at the date of this announcement, the Board comprises Mr. DOU Rongxing, Mr. WANG Jiong, Mr. LI Yulin and Mr. WEI Jie as executive directors; Mr. LI Qiaocheng, Mr. LI Xipeng and Mr. -

Newsletter November 2019 2019.12.01
Country Garden Holdings Company Limited 碧 桂 园 控 股 有 限 公 司 (2007.HK) Newsletter November 2019 Country Garden Holdings Company Limited(“Country Garden” or the “Company”) together with its subsidiaries,(collectively, the “Group”)(stock code: 2007) is one of China’s leading integrated property developers and a constituent stock of Hang Seng Index. The Group became a constituent stock of MSCI Global Standard Indices on 1 September 2007, Hang Seng Composite Index and Hang Seng Mainland 100 on 10 September 2007, FTSE China 50 Index on 14 September 2016, Hang Seng China(Hong Kong-listed) 25 Index on 12 June 2017, Hang Seng China 50 Index on 5 March 2018 and Hang Seng China Enterprises Index on 17 June 2019. Contracted Sales For the first 10 months of 2019, the Group, together with its joint ventures and associates, achieved contracted sales and contracted sales GFA attributable to owners of the Company amounted to RMB 479.69 billion and 53.66 million square meters(“sqm”), respectively. Geographical breakdown of contracted sales for Product type breakdown of contracted sales for the first 10 months of 2019 (By Attributable Value) the first 10 months of 2019 (By Attributable Value) Parking space & commercial, 6% Low-rise residential, 4% Guizhou, Others*, Guangdong, 3% 23% 22% Guangxi, 4% Jiangsu, 11% Hubei, High-rise residential, 90% 4% Zhejiang, Hunan, 8% 5% Shandong, Anhui, Henan, 6% 7% 7% Note : Others* including Shaanxi, Sichuan, Hebei, Yunnan, Jiangxi, Fujian, Hainan, Chongqing, Liaoning, Tianjin, Xinjiang, Gansu, Shanxi, Qinghai, Hong Kong, Malaysia, Ningxia, Beijing, United States, Shanghai, Inner Mongolia, Australia, Jilin, Thailand, Tibet, New Zealand, Indonesia , India and Heilongjiang. -

Newsletter August 2019 2019.09.02
Country Garden Holdings Company Limited 碧 桂 园 控 股 有 限 公 司 (2007.HK) Newsletter August 2019 Country Garden Holdings Company Limited(“Country Garden” or the “Company”) together with its subsidiaries,(collectively, the “Group”)(stock code: 2007) is one of China’s leading integrated property developers and a constituent stock of Hang Seng Index. The Group became a constituent stock of MSCI Global Standard Indices on 1 September 2007, Hang Seng Composite Index and Hang Seng Mainland 100 on 10 September 2007, FTSE China 50 Index on 14 September 2016, Hang Seng China(Hong Kong-listed) 25 Index on 12 June 2017, Hang Seng China 50 Index on 5 March 2018 and Hang Seng China Enterprises Index on 17 June 2019. Contracted Sales ◼For the first 7 months of 2019, the Group, together with its joint ventures and associates, achieved contracted sales and contracted sales GFA attributable to owners of the Company amounted to RMB 324.52 billion and 36.06 million square meters(“sqm”), respectively. Geographical breakdown of contracted sales for Product type breakdown of contracted sales for the first 7 months of 2019 (By Attributable Value) the first 7 months of 2019 (By Attributable Value) Parking space & commercial, 7% Low-rise residential, 4% Guizhou, Others*, Guangdong, 3% 22% 22% Guangxi, 4% Jiangsu, Hubei, 12% High-rise residential, 89% 4% Hunan, Zhejiang, 5% 8% Shandong, Anhui, Henan, 6% 7% 7% Note : Others* including Shaanxi, Sichuan, Yunnan, Hebei, Jiangxi, Fujian, Hainan, Chongqing, Liaoning, Tianjin, Shanxi, Xinjiang, Gansu, Qinghai, Hong Kong, Malaysia, Ningxia, Beijing, United States, Shanghai, Inner Mongolia, Australia, Jilin, Thailand, New Zealand, Tibet, Indonesia and Heilongjiang. -

Annual Report 2019
(Incorporated in the Cayman Islands with limited liability) Stock Code: 1638 Annual Report 2019 For identifi cation purposes only CORPORATE OVERVIEW Established in 1999, Kaisa Group Holdings Limited (the “Company” or “Kaisa”, which together with its subsidiaries, is referred to as the “Group”), is a large-scale integrated property developer. The shares of the Company began trading on the Main Board of The Stock Exchange of Hong Kong Limited on 9 December 2009. As a pioneer in the property market of the Guangdong-Hong Kong-Macao Greater Bay Area (the “Greater Bay Area”), Kaisa has anticipated China’s national development strategy and proactively undertaken comprehensive property development, urban redevelopment, operation of commercial properties, hotel management and property management. Its products comprise of residential properties, villas, offices, serviced apartments, integrated commercial buildings and mega urban complexes. Having established its home base in Shenzhen, Kaisa has been expanding its presence in the Greater Bay Area over the years. With footholds in Shenzhen, Guangzhou, Huizhou, Zhuhai, Foshan, Dongguan and Zhongshan, the Group’s businesses also cover the economically vibrant cities in such regions as the Yangtze River Delta, western and central parts of China and the pan-Bohai Rim. As at 31 December 2019, the Group’s land bank in the Greater Bay Area totaled approximately 13.6 million sq.m., or approximately 51% of its total land bank. By virtue of its rich experience in urban redevelopment, the Group has secured many high-quality urban redevelopment projects in the Greater Bay Area, mainly in such cities as Shenzhen, Guangzhou and Zhongshan, which will lay a solid foundation for the Group’s future development. -

Newsletter July 2019 2019.08.02
Country Garden Holdings Company Limited 碧桂园控股有限公司(2007.HK) Newsletter July 2019 Country Garden Holdings Company Limited(“Country Garden” or the “Company”) together with its subsidiaries,(collectively, the “Group”)(stock code: 2007) is one of China’s leading integrated property developers and a constituent stock of Hang Seng Index. The Group became a constituent stock of MSCI Global Standard Indices on 1 September 2007, Hang Seng Composite Index and Hang Seng Mainland 100 on 10 September 2007, FTSE China 50 Index on 14 September 2016, Hang Seng China(Hong Kong-listed) 25 Index on 12 June 2017, and Hang Seng China 50 Index on 5 March 2018 and Hang Seng China Enterprises Index on 17 June 2019. Contracted Sales For the first 6 months of 2019, the Group, together with its joint ventures and associates, achieved contracted sales and contracted sales GFA attributable to owners of the Company amounted to RMB 281.95 billion and 31.29million square meters(“sqm”), respectively. Geographical breakdown of contracted sales for Product type breakdown of contracted sales for the first 6 months of 2019 (By Attributable Value) the first 6 months of 2019 (By Attributable Value) Parking space & commercial, 7% Low-rise residential, 4% Guizhou, Others*, Guangdong, 3% 23% 22% Hubei, Jiangsu, 4% 12% Guangxi, High-rise residential, 89% 4% Hunan, Anhui, 8% 5% Shandong, Henan, Zhejiang, 7% 6% 6% Note : Others* including Shaanxi, Sichuan, Yunnan, Hebei, Jiangxi, Fujian, Chongqing, Hainan, Liaoning, Tianjin, Shanxi, Hong Kong, Gansu, Qinghai, Xinjiang, Malaysia, Ningxia, America, Shanghai, Beijing, Inner Mongolia, Australia, Jilin, Thailand, New Zealand, Indonesia, Tibet and Heilongjiang. -

Historical Review on the Spread of Chinese Martial Arts in Northern Europe
2018 4th International Conference on Innovative Development of E-commerce and Logistics (ICIDEL 2018) Historical Review on the Spread of Chinese Martial Arts in Northern Europe Xiaoxue Liu1, Yanfen Zhang2, and Xuezhi Ma3 1Department of Physical Education, China University of Geosciences, Xueyuan Road, Haidian District, Beijing, P. R. China 2Department of Life Sciences, Xinxiang University, Xinxiang Henan Province, Eastern Section of Hua Lan Road, Hongqi District, Xinxiang City, Henan, China 3Beijing Sport University Wushu School, information road, Haidian District, Beijing, China [email protected], [email protected], [email protected] Keywords: Chinese Martial Arts, Northern Europe, Cultural Integration, Acceptance, Spread Abstract: With literature data method and survey method, this paper studies the historical review on the spread of Chinese martial arts in northern Europe. Chinese martial arts is a national traditional sport. Therefore, the spread of Chinese martial arts in northern Europe is a continuation of the development of other traditional projects in the Nordic region which shows pluralism on introduction and development. The process of the spread of Chinese martial arts in northern Europe contains a process in which Chinese martial arts like other traditional sports have gradually been accepted and recognized by Nordic society. 1. Introduction The history of spread of Chinese martial arts in northern Europe has a close relationship with globalization as well as social politics and economic environment of China. Besides, it is closely associated with the influence of China as a main communication subject on northern Europe (Xu Zhiwei, 1991). Therefore, if we want to analyze and explain the historical evolution of the spread of Chinese martial arts in northern Europe, we must have a review of the development process of modern Chinese martial arts and its spreading history in northern Europe. -
Sanctioned Entities Name of Firm & Address Date of Imposition of Sanction Sanction Imposed Grounds China Railway Constructio
Sanctioned Entities Name of Firm & Address Date of Imposition of Sanction Sanction Imposed Grounds China Railway Construction Corporation Limited Procurement Guidelines, (中国铁建股份有限公司)*38 March 4, 2020 - March 3, 2022 Conditional Non-debarment 1.16(a)(ii) No. 40, Fuxing Road, Beijing 100855, China China Railway 23rd Bureau Group Co., Ltd. Procurement Guidelines, (中铁二十三局集团有限公司)*38 March 4, 2020 - March 3, 2022 Conditional Non-debarment 1.16(a)(ii) No. 40, Fuxing Road, Beijing 100855, China China Railway Construction Corporation (International) Limited Procurement Guidelines, March 4, 2020 - March 3, 2022 Conditional Non-debarment (中国铁建国际集团有限公司)*38 1.16(a)(ii) No. 40, Fuxing Road, Beijing 100855, China *38 This sanction is the result of a Settlement Agreement. China Railway Construction Corporation Ltd. (“CRCC”) and its wholly-owned subsidiaries, China Railway 23rd Bureau Group Co., Ltd. (“CR23”) and China Railway Construction Corporation (International) Limited (“CRCC International”), are debarred for 9 months, to be followed by a 24- month period of conditional non-debarment. This period of sanction extends to all affiliates that CRCC, CR23, and/or CRCC International directly or indirectly control, with the exception of China Railway 20th Bureau Group Co. and its controlled affiliates, which are exempted. If, at the end of the period of sanction, CRCC, CR23, CRCC International, and their affiliates have (a) met the corporate compliance conditions to the satisfaction of the Bank’s Integrity Compliance Officer (ICO); (b) fully cooperated with the Bank; and (c) otherwise complied fully with the terms and conditions of the Settlement Agreement, then they will be released from conditional non-debarment. If they do not meet these obligations by the end of the period of sanction, their conditional non-debarment will automatically convert to debarment with conditional release until the obligations are met. -
2019 ANNUAL REPORT Chapter 1 Corporate Information
Content Corporate Information 2 Financial Summary 8 Chairman’s Statement 11 President’s Statement 13 Management Discussion and Analysis 16 Changes in Share Capital and Information on Shareholders 79 Directors, Supervisors, Senior Management, Employees and Institutions 89 Corporate Governance Report 114 Report of the Board of Directors 149 Report of the Board of Supervisors 161 Important Events 165 Independent Auditor’s Report 169 Unaudited Supplementary Financial Information 354 Definitions 358 Chapter 1 Corporate Information 1. Basic Corporate Profile Legal Name of the Company 中原銀行股份有限公司1 (abbreviated as 中原銀行) English Name of the Company ZHONGYUAN BANK CO., LTD.1 (abbreviated as ZYBANK) Registered Office Zhongke Golden Tower, No. 23 Shangwu Waihuan Road, Zhengdong New District CBD, Zhengzhou, Henan Province, the PRC Headquarters in China Zhongke Golden Tower, No. 23 Shangwu Waihuan Road, Zhengdong New District CBD, Zhengzhou, Henan Province, the PRC Principal Business Place in Hong Kong 40/F, Sunlight Tower, 248 Queen’s Road East, Wanchai, Hong Kong Board of Directors Executive Directors: Mr. DOU Rongxing (Chairman) Mr. WANG Jiong Mr. LI Yulin Mr. WEI Jie Non-Executive Directors: Mr. LI Qiaocheng Mr. LI Xipeng Mr. MI Hongjun 1 Zhongyuan Bank Co., Ltd. is not an authorized institution within the meaning of the Banking Ordinance (Chapter 155 of the Laws of Hong Kong), not subject to the supervision of the Hong Kong Monetary Authority, and not authorized to carry on banking/deposit-taking business in Hong Kong 2 ZHONGYUAN BANK CO., LTD. 2019 ANNUAL REPORT Chapter 1 Corporate Information Independent non-executive Directors: Ms. PANG Hong Mr. LI Hongchang Mr. JIA Tingyu Mr. -
Relationship Between Emotional Intelligence and Job Well-Being In
Li et al. BMC Nursing (2021) 20:144 https://doi.org/10.1186/s12912-021-00658-4 RESEARCH Open Access Relationship between emotional intelligence and job well-being in Chinese clinical nurses: multiple mediating effects of empathy and communication satisfaction Xue Li1, Hongjuan Chang1*, Quanying Zhang2, Jianli Yang2, Rui Liu1 and Yajie Song1 Abstract Background: Nursing work is associated with great pressure, and nurses are often overwhelmed. Therefore, correct emotional regulation is essential to improve nurses’ job well-being and promote better engagement in nursing work. The purpose of this study was to establish a structural model to estimate the impact of Chinese clinical nurses’ emotional intelligence on job well-being, using multiple intermediaries to explain the internal mechanisms underlying the relationship. Methods: This was a cross-sectional study of 1475 registered nurses from a Chinese hospital who provided responses to emotional intelligence, empathy, communication satisfaction, and job well-being scales. Path analysis using a multiple mediation model was performed using AMOS 23.0. Results: Among all clinical nurses who participated in the survey, 1475 (98.33 %) completed the questionnaire. The nurses’ job well-being score was 83.61 ± 12.63. There was a significant positive correlation between job well-being and communication satisfaction, emotional intelligence, and empathy ability (r = 0.346–0.570, P < 0.001). Empathy and communication satisfaction partially mediated the relationship between emotional intelligence and job well- being, with effect sizes of 0.047 and 0.227, respectively. The chain mediating effect of empathy and communication satisfaction had a value of 0.045. Conclusions: It is recommended that hospital managers take actions to improve nurses’ emotional intelligence level, and conduct professional psychological training to improve nurses’ empathy and communication satisfaction, and ultimately improve their job well-being.