Disposable Electrochemical Biosensor for Organophosphorous Nerve
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Carbamate Pesticides Aldicarb Aldicarb Sulfoxide Aldicarb Sulfone
Connecticut General Statutes Sec 19a-29a requires the Commissioner of Public Health to annually publish a list setting forth all analytes and matrices for which certification for testing is required. Connecticut ELCP Drinking Water Analytes Revised 05/31/2018 Microbiology Total Coliforms Fecal Coliforms/ E. Coli Carbamate Pesticides Legionella Aldicarb Cryptosporidium Aldicarb Sulfoxide Giardia Aldicarb Sulfone Carbaryl Physicals Carbofuran Turbidity 3-Hydroxycarbofuran pH Methomyl Conductivity Oxamyl (Vydate) Minerals Chlorinated Herbicides Alkalinity, as CaCO3 2,4-D Bromide Dalapon Chloride Dicamba Chlorine, free residual Dinoseb Chlorine, total residual Endothall Fluoride Picloram Hardness, Calcium as Pentachlorophenol CaCO3 Hardness, Total as CaCO3 Silica Chlorinated Pesticides/PCB's Sulfate Aldrin Chlordane (Technical) Nutrients Dieldrin Endrin Ammonia Heptachlor Nitrate Heptachlor Epoxide Nitrite Lindane (gamma-BHC) o-Phosphate Metolachlor Total Phosphorus Methoxychlor PCB's (individual aroclors) Note 1 PCB's (as decachlorobiphenyl) Note 1 Demands Toxaphene TOC Nitrogen-Phosphorus Compounds Alachlor Metals Atrazine Aluminum Butachlor Antimony Diquat Arsenic Glyphosate Barium Metribuzin Beryllium Paraquat Boron Propachlor Cadmium Simazine Calcium Chromium Copper SVOC's Iron Benzo(a)pyrene Lead bis-(2-ethylhexyl)phthalate Magnesium bis-(ethylhexyl)adipate Manganese Hexachlorobenzene Mercury Hexachlorocyclopentadiene Molybdenum Nickel Potassium Miscellaneous Organics Selenium Dibromochloropropane (DBCP) Silver Ethylene Dibromide (EDB) -

Neurotoxicity in Preclinical Models of Occupational Exposure to Organophosphorus Compounds
CORE Metadata, citation and similar papers at core.ac.uk Provided by Frontiers - Publisher Connector REVIEW published: 18 January 2017 doi: 10.3389/fnins.2016.00590 Neurotoxicity in Preclinical Models of Occupational Exposure to Organophosphorus Compounds Jaymie R. Voorhees 1, 2*, Diane S. Rohlman 2, 3, Pamela J. Lein 4 and Andrew A. Pieper 1, 2, 5, 6, 7, 8, 9* 1 Department of Psychiatry, University of Iowa Carver College of Medicine, Iowa City, IA, USA, 2 Interdisciplinary Graduate Program in Human Toxicology, University of Iowa Carver College of Medicine, Iowa City, IA, USA, 3 Department of Occupational and Environmental Health, University of Iowa College of Public Health, Iowa City, IA, USA, 4 Department of Molecular Biosciences, School of Veterinary Medicine, University of California, Davis, Davis, CA, USA, 5 Department of Neurology, University of Iowa Carver College of Medicine, Iowa City, IA, USA, 6 Department of Free Radical and Radiation Biology Program, University of Iowa Carver College of Medicine, Iowa City, IA, USA, 7 Department of Radiation Oncology Holden Comprehensive Cancer Center, University of Iowa Carver College of Medicine, Iowa City, IA, USA, 8 Department of Veteran Affairs, University of Iowa Carver College of Medicine, Iowa City, IA, USA, 9 Weill Cornell Autism Research Program, Weill Cornell Medical College, New York, NY, USA Organophosphorus (OPs) compounds are widely used as insecticides, plasticizers, and fuel additives. These compounds potently inhibit acetylcholinesterase (AChE), the enzyme that inactivates acetylcholine at neuronal synapses, and acute exposure to high Edited by: OP levels can cause cholinergic crisis in humans and animals. Evidence further suggests Stefano L. -

Florida State Emergency Response Commission
Florida State Emergency Response Commission Sub-Committee on Training (SOT) HAZARDOUS MATERIALS MEDICAL TREATMENT PROTOCOLS Version 3.3 TOXIDROMES Toxidromes are clinical syndromes that the patient presents with. These patterns of signs and symptoms are essential for the successful recognition of chemical exposure. The toxidromes identified in this protocol are chemical exposure based while others such as the opioids are found within general medical protocol. These chemical toxidromes are identified clinically into five syndromes: Irritant Gas Toxidrome Asphyxiant Toxidrome Corrosive Toxidrome Hydrocarbon and Halogenated Hydrocarbons Toxidrome Cholinergic Toxidrome Each can present as a clinical manifestation of the chemical/poisoning involved with some cross-over between toxidromes. This list combines the toxic syndromes found within NFPA 473 (A.5.4.1(2) and traditional syndromes. Toxidrome Correlation to NFPA Standard 473 and Traditional Syndromes Toxidrome NFPA 473 A.5.4.1(2) Hazardous Materials Protocol Correlation Irritant Gas (j) Irritants Bronchospasm OC Pepper spray & lacrimants Asphyxiant (c) Chemical asphyxiants Carbon Monoxide (d) Simple asphyxiants Aniline dyes, Nitriles, Nitrares (h) Blood Agents Cyanide & Hydrogen Sulfide (n) Nitrogen Compounds Closed Space Fires Simple Asphyxants Corrosive (a) Corrosives Hydrofluroic Acid (g) Vesicants Chemical burns to the eye Choramine and Chlorine Hydrocarbon (e) Organic solvents Phenol and (q) Phenolic Compounds Halogenated Hydrocarbons Halogenated Hydrocarbons Cholinergic (b) Pesticides -

Chemical Name Federal P Code CAS Registry Number Acutely
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extremely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extremely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extremely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extremely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extremely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extremely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extremely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extremely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extremely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -
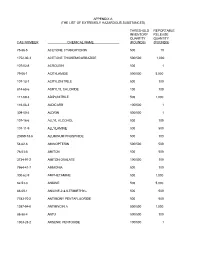
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

Malathion Human Health and Ecological Risk Assessment Final Report
SERA TR-052-02-02c Malathion Human Health and Ecological Risk Assessment Final Report Submitted to: Paul Mistretta, COR USDA/Forest Service, Southern Region 1720 Peachtree RD, NW Atlanta, Georgia 30309 USDA Forest Service Contract: AG-3187-C-06-0010 USDA Forest Order Number: AG-43ZP-D-06-0012 SERA Internal Task No. 52-02 Submitted by: Patrick R. Durkin Syracuse Environmental Research Associates, Inc. 5100 Highbridge St., 42C Fayetteville, New York 13066-0950 Fax: (315) 637-0445 E-Mail: [email protected] Home Page: www.sera-inc.com May 12, 2008 Table of Contents Table of Contents............................................................................................................................ ii List of Figures................................................................................................................................. v List of Tables ................................................................................................................................. vi List of Appendices ......................................................................................................................... vi List of Attachments........................................................................................................................ vi ACRONYMS, ABBREVIATIONS, AND SYMBOLS ............................................................... vii COMMON UNIT CONVERSIONS AND ABBREVIATIONS.................................................... x CONVERSION OF SCIENTIFIC NOTATION .......................................................................... -

Table II. EPCRA Section 313 Chemical List for Reporting Year 2017 (Including Toxic Chemical Categories)
Table II. EPCRA Section 313 Chemical List For Reporting Year 2017 (including Toxic Chemical Categories) Individually listed EPCRA Section 313 chemicals with CAS numbers are arranged alphabetically starting on page II-3. Following the alphabetical list, the EPCRA Section 313 chemicals are arranged in CAS number order. Covered chemical categories follow. Note: Chemicals may be added to or deleted from the list. The Emergency Planning and Community Right-to-Know Call Center or the TRI-Listed Chemicals website will provide up-to-date information on the status of these changes. See section B.3.c of the instructions for more information on the de minimis % limits listed below. There are no de minimis levels for PBT chemicals since the de minimis exemption is not available for these chemicals (an asterisk appears where a de minimis limit would otherwise appear in Table II). However, for purposes of the supplier notification requirement only, such limits are provided in Appendix C. Chemical Qualifiers Certain EPCRA Section 313 chemicals listed in Table II have parenthetic “qualifiers.” These qualifiers indicate that these EPCRA Section 313 chemicals are subject to the section 313 reporting requirements if manufactured, processed, or otherwise used in a specific form or when a certain activity is performed. An EPCRA Section 313 chemical that is listed without a qualifier is subject to reporting in all forms in which it is manufactured, processed, and otherwise used. The following chemicals are reportable only if they are manufactured, processed, or otherwise used in the specific form(s) listed below: Chemical/ Chemical Category CAS Number Qualifier Aluminum (fume or dust) 7429-90-5 Only if it is a fume or dust form. -

Probabilistic Assessment of the Cumulative Acute Exposure to Organophosphorus and Carbamate Insecticides in the Brazilian Diet E.D
Toxicology 222 (2006) 132–142 Probabilistic assessment of the cumulative acute exposure to organophosphorus and carbamate insecticides in the Brazilian diet E.D. Caldas a,∗, P.E. Boon b, J. Tressou c a Department of Pharmaceutical Sciences, College of Health Sciences, University of Bras´ılia, 70919-970 Bras´ılia, DF, Brazil b RIKILT, Institute of Food Safety, Wageningen University and Research Centre, 6708 PD Wageningen, The Netherlands c INRA, Unit´eM´et@risk, Methodologies d’analyse de risque alimentaire INA-PG, 75231 Paris, France Received 10 October 2005; received in revised form 1 February 2006; accepted 13 February 2006 Available online 6 March 2006 Abstract In the present study, the cumulative exposure of 25 acetylcholinesterase (AChE) inhibiting pesticides through the consumption of nine fruits and vegetables by the Brazilian population was assessed. Food consumption data were obtained from a household budget survey conducted in all Brazilian states from July 2002 to June 2003. Residue data from 4001 samples were obtained from the Brazilian national monitoring program on pesticide residues. Relative potency factors (RPF) were calculated with methamidophos or acephate as index compounds (IC), using BMD10 or NOAEL for AChE inhibition, mostly in rat brain, obtained from national and international pesticide evaluations. Monocrotophos and triazophos, in addition to aldicarb, had the highest calculated RPF in any scenario. The exposure to AChE inhibiting pesticides for the general population at P99.9, represented 33.6% of the ARfD as methamidophos and 70.2% ARfD as acephate. The exposure calculated as acephate could exceed the ARfD at the upper bound of the 95% confidence interval for this percentile. -

The Dietary Supplement Specialists
ADVANCED LABORATORIES STYLE GUIDE: LOGO CMYK COLORS CMYK C 100% M o% Y o% K o% THE DIETARY SUPPLEMENT SPECIALISTS GRAY SCALE C 0% M o% Y o% K 50% BLACK/WHITE C 75% M 68% Y 67% K 90% THE DIETARY SUPPLEMENT SPECIALISTS Price and Capabilites list ADVANCED LABORATORIES STYLE GUIDE: LOGO CMYK COLORS CMYK C 100% M o% Y o% K o% THE DIETARY SUPPLEMENT SPECIALISTS ADVANCED LABORATORIES CAN PROVIDE ALL OF YOUR ANALYTICAL TESTING NEEDS: GRAY SCALE • Microbiological Analysis • One-stop Sample Shipping C 0% M o% • Chemical Analysis (HPLC, GC, FTIR, etc.) • Competitive pricing, with value-added at no additional charge. Y o% • Metals Analysis (ICP, ICP/MS) • Quick turn-around time, typically 3-5 business days K 50% • Raw Materials Analysis • Open 6 Days / Week includes receiving deliveries and • Finished Products Analysis reporting results on Saturday. Closed major holidays. • Food Products Analysis • Trend Check™ Online Sample Data • Nutritional Labeling Amino Acids | Botanicals | Microbiological | Metals/Minerals | Nutritional Supplements Nutritional Labeling | Pesticide Screens | Shelf-Life Testing | Vitamins cGMP Compliant - Please come audit us! ISO/IEC 17025:2005 BLACK/WHITE C 75% M 68% Our Trend CheckTM online program allows customers to view Y 67% K 90% all pertinent sample data including final test certificates, up to date test results, the ability to export to Excel for easy data entry or trend analysis, as well as access invoices quickly, efficiently, and securely at any time. All this at no charge! One Day Rush = 100% Surcharge | Two Day Rush = 50% Surcharge | Three Day Rush = 25% Surcharge The information provided by Advanced Laboratories® is based on the most popular testing. -

Code Chemical P026 1-(O-Chlorophenyl)Thiourea P081 1
Code Chemical P026 1-(o-Chlorophenyl)thiourea P081 1,2,3-Propanetriol, trinitrate (R) P042 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, (R)- P067 1,2-Propylenimine P185 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)- carbonyl]oxime 1,4,5,8-Dimethanonaphthalene, 1,2,3,4,10,10-hexa- chloro-1,4,4a,5,8,8a,-hexahydro-, P004 (1alpha,4alpha, 4abeta,5alpha,8alpha,8abeta)- 1,4,5,8-Dimethanonaphthalene, 1,2,3,4,10,10-hexa- chloro-1,4,4a,5,8,8a-hexahydro-, P060 (1alpha,4alpha, 4abeta,5beta,8beta,8abeta)- P002 1-Acetyl-2-thiourea P048 2,4-Dinitrophenol P051 2,7:3,6-Dimethanonaphth [2,3-b]oxirene, 3,4,5,6,9,9 -hexachloro-1a,2,2a,3,6,6a,7,7a- octahydro-, (1aalpha,2beta,2abeta,3alpha,6alpha,6abeta,7 beta, 7aalpha)-, & metabolites 2,7:3,6-Dimethanonaphth[2,3-b]oxirene, 3,4,5,6,9,9- hexachloro-1a,2,2a,3,6,6a,7,7a- P037 octahydro-, (1aalpha,2beta,2aalpha,3beta,6beta,6aalpha,7 beta, 7aalpha)- P045 2-Butanone, 3,3-dimethyl-1-(methylthio)-, O-[methylamino)carbonyl] oxime P034 2-Cyclohexyl-4,6-dinitrophenol 2H-1-Benzopyran-2-one, 4-hydroxy-3-(3-oxo-1- phenylbutyl)-, & salts, when present at P001 concentrations greater than 0.3% P069 2-Methyllactonitrile P017 2-Propanone, 1-bromo- P005 2-Propen-1-ol P003 2-Propenal P102 2-Propyn-1-ol P007 3(2H)-Isoxazolone, 5-(aminomethyl)- P027 3-Chloropropionitrile P047 4,6-Dinitro-o-cresol, & salts P059 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro- 3a,4,7,7a-tetrahydro- P008 4-Aminopyridine P008 4-Pyridinamine P007 5-(Aminomethyl)-3-isoxazolol 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- -

Aldicarb by Caroline Cox Pulses Across the Junctions Between Nerves
Aldicarb By Caroline Cox pulses across the junctions between nerves. (given over a one-year period). The high- This causes loss of muscular coordination, est dose at which no adverse effects were In 1966, four years before the insecticide convulsions, and ultimately death.5 The observed (called the NOEL) was 0.02 mg/ 11 aldicarb was registered for use in the United AChE inhibition is said to be reversible be- kg/day, about one-tenth of the LD50. States, researchers were using the chemical cause the aldicarb disassociates from the There is evidence that people may suf- on an experimental basis.1 One researcher AChE within several hours. This occurs even fer acute symptoms when exposed to even brought a small amount home and his wife if death has occurred. Organophosphate in- lower levels of aldicarb. In humans who applied it to the soil under a backyard rose secticides (malathion and diazinon, for ex- consumed aldicarb-contaminated water- bush. Over three weeks later she ate a sprig ample) have the same mode of action ex- melons, clinical signs of aldicarb poison- of mint from a plant growing nearby. Within cept that the AChE inhibition is not as ing were found in individuals consuming half an hour she was suffering from vomit- readily reversible.7 as little as 0.002 mg/kg of the aldicarb ing, diarrhea, and involuntary urination. Her Aldicarb is a systemic insecticide. It is metabolite aldicarb sulfoxide. This pupils closed to pinpoints, her muscles applied as granules below the soil surface amount is one-tenth the NOEL in the twitched, and her breathing was difficult. -

Investigation on the Behavior of Pesticides in Atmosphere
Aerosol and Air Quality Research, 11: 783–790, 2011 Copyright © Taiwan Association for Aerosol Research ISSN: 1680-8584 print / 2071-1409 online doi: 10.4209/aaqr.2010.10.0085 Investigation on the Behavior of Pesticides in Atmosphere Pasquale Avino1, Giuseppe Cinelli2, Ivan Notardonato2, Mario V. Russo2* 1 DIPIA, INAIL (ex-ISPESL), via Urbana 167, 00184 Rome, Italy 2 Faculty of Agriculture, University of Molise, via De Sanctis, Campobasso, Italy ABSTRACT Although pesticides are widely used in agriculture, they and in particular the relative residues in foodstuffs, water and atmosphere, may cause remarkable sanitary problems due to the harmful effects (carcinogenic and mutagenic effects) on the human health. In fact, their spread in waters and atmosphere can produce undesired effects on various organisms and/or water contamination. This paper shows an analytical approach based on XAD-2 adsorbent and GC analysis for evaluating the pesticide trend in atmosphere: in particular, the pesticides investigated are omethoate, dicrotofos, disulfoton, dimethoate, parathion methyl, formothion, paraoxon ethyl, malaoxon, parathion ethyl, iodofenfos and triazofos. For the analytical methodology a linearity response was obtained (r2 = 0.9988) in GC-NPD whereas the limits of detection range between 2 and 5 pg/μL in GC-NPD with a Relative Standard Deviation below 9.5. Finally, this approach has been successfully applied to real samples: the results show that dimethoate concentration decreases with increasing distance from the sampling site but it is still persistent in atmosphere after few days from the pesticide spraying. Keywords: Pesticides; XAD-2 adsorbent; GC analysis; Atmosphere; Air quality. INTRODUCTION 2009). One of the most important OP reactions is water hydrolysis.