Atomic Weights
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Package 'Ciaawconsensus'
Package ‘CIAAWconsensus’ September 19, 2018 Type Package Title Isotope Ratio Meta-Analysis Version 1.3 Author Juris Meija and Antonio Possolo Maintainer Juris Meija <[email protected]> Description Calculation of consensus values for atomic weights, isotope amount ratios, and iso- topic abundances with the associated uncertainties using multivariate meta-regression ap- proach for consensus building. License Unlimited LazyData yes Imports mvtnorm, stringr, numDeriv, stats, Matrix NeedsCompilation no Repository CRAN Date/Publication 2018-09-19 13:30:12 UTC R topics documented: abundances2ratios . .2 at.weight . .3 ciaaw.mass.2003 . .4 ciaaw.mass.2012 . .5 ciaaw.mass.2016 . .6 iridium.data . .6 mmm ............................................7 normalize.ratios . .8 platinum.data . .9 Index 10 1 2 abundances2ratios abundances2ratios Isotope ratios of a chemical element from isotopic abundances Description This function calculates the isotope ratios of a chemical element from the given isotopic abundances and their uncertainties. The uncertainty evaluation is done using the propagation of uncertainty and the missing correlations between the isotopic abundances are reconstructed using Monte Carlo methods. Usage abundances2ratios(x, ux, ref=1, iterations=1e4) Arguments x A vector of isotopic abundances of an element ux Standard uncertainties of x ref Index to specify the desired reference isotope for isotope amount ratios iterations Number of iterations for isotopic abundance correlation mapping Details Situations are often encountered where isotopic abundances are reported but not the isotope ratios. In such cases we reconstruct the isotope ratios that are consistent with the abundances and their uncertainties. Given only the abundances and their uncertainties, for elements with four or more isotopes one cannot unambiguously infer the uncertainties of the ratios due to the unknown correla- tions between isotopic abundances. -

Properties of Carbon the Atomic Element Carbon Has Very Diverse
Properties of Carbon The atomic element carbon has very diverse physical and chemical properties due to the nature of its bonding and atomic arrangement. fig. 1 Allotropes of Carbon Some allotropes of carbon: (a) diamond, (b) graphite, (c) lonsdaleite, (d–f) fullerenes (C60, C540, C70), (g) amorphous carbon, and (h) carbon nanotube. Carbon has several allotropes, or different forms in which it can exist. These allotropes include graphite and diamond, whose properties span a range of extremes. Despite carbon's ability to make 4 bonds and its presence in many compounds, it is highly unreactive under normal conditions. Carbon exists in 2 main isotopes: 12C and 13C. There are many other known isotopes, but they tend to be short-lived and have extremely short half-lives. Allotropes The different forms of a chemical element. Cabon is the chemical element with the symbol C and atomic number 6. As a member of group 14 on the periodic table, it is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon has 6 protons and 6 Source URL: https://www.boundless.com/chemistry/nonmetallic-elements/carbon/properties-carbon/ Saylor URL: http://www.saylor.org/courses/chem102#6.1 Attributed to: Boundless www.saylor.org Page 1 of 2 neutrons, and has a standard atomic weight of 12.0107 amu. Its electron configuration is denoted as 1s22s22p2. It is a solid, and sublimes at 3,642 °C. It's oxidation state ranges from 4 to -4, and it has an electronegativity rating of 2.55 on the Pauling scale. Carbon has several allotropes, or different forms in which it exists. -
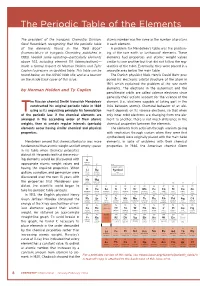
The Periodic Table of the Elements
The Periodic Table of the Elements The president of the Inorganic Chemistry Division, atomic number was the same as the number of protons Gerd Rosenblatt, recognizing that the periodic table in each element. of the elements found in the “Red Book” A problem for Mendeleev’s table was the position- (Nomenclature of Inorganic Chemistry, published in ing of the rare earth or lanthanoid* elements. These 1985) needed some updating—particularly elements elements had properties and atomic weight values above 103, including element 110 (darmstadtium)— similar to one another but that did not follow the reg- made a formal request to Norman Holden and Tyler ularities of the table. Eventually, they were placed in a Coplen to prepare an updated table. This table can be separate area below the main table. found below, on the IUPAC Web site, and as a tear-off The Danish physicist Niels Henrik David Bohr pro- on the inside back cover of this issue. posed his electronic orbital structure of the atom in 1921, which explained the problem of the rare earth by Norman Holden and Ty Coplen elements. The electrons in the outermost and the penultimate orbits are called valence electrons since generally their actions account for the valence of the he Russian chemist Dmitri Ivanovich Mendeleev element (i.e., electrons capable of taking part in the constructed his original periodic table in 1869 links between atoms). Chemical behavior of an ele- Tusing as its organizing principle his formulation ment depends on its valence electrons, so that when of the periodic law: if the chemical elements are only inner orbit electrons are changing from one ele- arranged in the ascending order of their atomic ment to another, there is not much difference in the weights, then at certain regular intervals (periods) chemical properties between the elements. -

Isotopes Ions Electronic Structure Relative Atomic Mass (Ar) Chemical
Chemistry 1: Atomic structure and the periodic table Electronic Structure Atoms are ny, too small to see. They have a radius of 0.1 nanometres ( 1 x 10 –10 m) Atoms 1st shell– Lowest energy level and can hold 2 electrons Atoms have no charge because they have the same number of protons and electrons. 2nd shell– Energy level can hold up to 8 electrons Electron Proton 3rd shell onwards– Can hold up to 8 electrons. Nucleus Neutron Electron structure and the periodic table Elements in the same group have the same number of electrons on their outer shell. Electron Orbit around nucleus in Mass Number : shells protons + neutrons Proton number = Electron Number Proton Found in the nucleus Atomic number: Number of neutrons= Neutron Found in the nucleus Protons Mass number—Atomic number An ion is an atom that has lost or gained electrons. An isotope is an atom that has the same number Ions Isotopes of protons but a different number of neutrons. In an ion the number of protons is not equal to the number of electrons so the atom has an overall They have the same atomic number but different atomic mass numbers. charge. This can either be posive or negave. Relative atomic mass (Ar) Relave atomic = sum of (isotope abundance x isotope mass number) Mass (Ar) sum of abundance of all the isotopes An average mass of an element that has a number of different isotopes. Chemical Equations Balancing equaons: There must always be the same number of atoms on Chemical reacons are shown using: both sides of a symbol equaon. -

Guidelines for the Use of Atomic Weights 5 10 11 12 DOI: ..., Received ...; Accepted
IUPAC Guidelines for the us e of atomic weights For Peer Review Only Journal: Pure and Applied Chemistry Manuscript ID PAC-REC-16-04-01 Manuscript Type: Recommendation Date Submitted by the Author: 01-Apr-2016 Complete List of Authors: van der Veen, Adriaan; VSL Meija, Juris Possolo, Antonio; National Institute of Standards and Technology Hibbert, David; University of New South Wales, School of Chemistry atomic weights, atomic-weight intervals, molecular weight, standard Keywords: atomic weight, measurement uncertainty, uncertainty propagation Author-Supplied Keywords: P.O. 13757, Research Triangle Park, NC (919) 485-8700 Page 1 of 13 IUPAC Pure Appl. Chem. 2016; aop 1 2 3 4 Sponsoring body: IUPAC Inorganic Chemistry Division Committee: see more details on page XXX. 5 IUPAC Recommendation 6 7 Adriaan M. H. van der Veen*, Juris Meija, Antonio Possolo, and D. Brynn Hibbert 8 9 Guidelines for the use of atomic weights 5 10 11 12 DOI: ..., Received ...; accepted ... 13 14 Abstract: Standard atomicFor weights Peer are widely used Review in science, yet the uncertainties Only associated with these 15 values are not well-understood. This recommendation provides guidance on the use of standard atomic 16 weights and their uncertainties. Furthermore, methods are provided for calculating standard uncertainties 17 of molecular weights of substances. Methods are also outlined to compute material-specific atomic weights 10 18 whose associated uncertainty may be smaller than the uncertainty associated with the standard atomic 19 weights. 20 21 Keywords: atomic weights; atomic-weight intervals; molecular weight; standard atomic weight; uncertainty; 22 uncertainty propagation 23 24 25 1 Introduction 15 26 27 Atomic weights provide a practical link the SI base units kilogram and mole. -

Project Note Weston Solutions, Inc
PROJECT NOTE WESTON SOLUTIONS, INC. To: Canadian Radium & Uranium Corp. Site File Date: June 5, 2014 W.O. No.: 20405.012.013.2222.00 From: Denise Breen, Weston Solutions, Inc. Subject: Determination of Significant Lead Concentrations in Sediment Samples References 1. New York State Department of Environmental Conservation. Technical Guidance for Screening Contaminated Sediments. March 1998. [45 pages] 2. U.S. Environmental Protection Agency (EPA) Office of Emergency Response. Establishing an Observed Release – Quick Reference Fact Sheet. Federal Register, Volume 55, No. 241. September 1995. [7 pages] 3. International Union of Pure and Applied Chemistry, Inorganic Chemistry Division Commission on Atomic Weights and Isotopic Abundances. Atomic Weights of Elements: Review 2000. 2003. [120 pages] WESTON personnel collected six sediment samples (including one environmental duplicate sample) from five locations along the surface water pathway of the Canadian Radium & Uranium Corp. (CRU) site in May 2014. The sediment samples were analyzed for Target Analyte List (TAL) Metals and Stable Lead Isotopes. 1. TAL Lead Interpretation: In order to quantify the significance for Lead, Thallium and Mercury the following was performed: 1. WESTON personnel tabulated all available TAL Metal data from the May 2014 Sediment Sampling event. 2. For each analyte of concern (Lead, Thallium, and Mercury), the highest background concentration was selected and then multiplied by three. This is the criteria to find the significance of site attributable release as per Hazard Ranking System guidelines. 3. One analytical lead result (2222-SD04) of 520 mg/kg (J) was qualified with an unknown bias. In accordance with US EPA document “Using Data to Document an Observed Release and Observed Contamination”, 2222-SD03 lead concentration was adjusted by dividing by the factor value for lead of 1.44 to equal 361 mg/kg. -

C H E M I S T
Post-16 Induction C GCSE to A Level Chemistry - Bridging work - H Name : _______________________________ E M I ALL sections should be completed Bring this booklet to your first chemistry lesson in September. S T R Y 1 | P a g e INTRODUCTION Welcome to chemistry. In choosing to study chemistry you will develop many useful lifelong transferable skills, in addition to developing a deeper understanding of the subject. This booklet is designed to prepare you for your first year of study and hopefully answer some of the questions you may have at this stage. The first section gives some useful information about the subject, the details of your course, and gives you the opportunity to see course overview. You will typically have two chemistry teachers; (covering organic and inorganic modules), and lessons will take place in the Lycett building (GL0.02 to GL0.05). Contact me (subject leader) by e-mailing [email protected] over the summer if you have any questions. The second section (from page 6) is designed to review some of the work you will have covered at GCSE and make links with the new material you will cover in your first year of A-level chemistry. A lot of the material we cover in year 12 chemistry you will recognise from GCSE, however, we will build on, and extend your knowledge and understanding in each area. It would be advantageous for you to complete this booklet over the summer (use the answers at the back to self-mark) so that your subject knowledge is fresh when you start in the autumn. -

Atomic Weights and Isotopic Abundances*
Pure&App/. Chem., Vol. 64, No. 10, pp. 1535-1543, 1992. Printed in Great Britain. @ 1992 IUPAC INTERNATIONAL UNION OF PURE AND APPLIED CHEMISTRY INORGANIC CHEMISTRY DIVISION COMMISSION ON ATOMIC WEIGHTS AND ISOTOPIC ABUNDANCES* 'ATOMIC WEIGHT' -THE NAME, ITS HISTORY, DEFINITION, AND UNITS Prepared for publication by P. DE BIEVRE' and H. S. PEISER2 'Central Bureau for Nuclear Measurements (CBNM), Commission of the European Communities-JRC, B-2440 Geel, Belgium 2638 Blossom Drive, Rockville, MD 20850, USA *Membership of the Commission for the period 1989-1991 was as follows: J. R. De Laeter (Australia, Chairman); K. G. Heumann (FRG, Secretary); R. C. Barber (Canada, Associate); J. CCsario (France, Titular); T. B. Coplen (USA, Titular); H. J. Dietze (FRG, Associate); J. W. Gramlich (USA, Associate); H. S. Hertz (USA, Associate); H. R. Krouse (Canada, Titular); A. Lamberty (Belgium, Associate); T. J. Murphy (USA, Associate); K. J. R. Rosman (Australia, Titular); M. P. Seyfried (FRG, Associate); M. Shima (Japan, Titular); K. Wade (UK, Associate); P. De Bi&vre(Belgium, National Representative); N. N. Greenwood (UK, National Representative); H. S. Peiser (USA, National Representative); N. K. Rao (India, National Representative). Republication of this report is permitted without the need for formal IUPAC permission on condition that an acknowledgement, with full reference together with IUPAC copyright symbol (01992 IUPAC), is printed. Publication of a translation into another language is subject to the additional condition of prior approval from the relevant IUPAC National Adhering Organization. ’Atomic weight‘: The name, its history, definition, and units Abstract-The widely used term “atomic weight” and its acceptance within the international system for measurements has been the subject of debate. -

Mendeleev's Periodic Table of the Elements the Periodic Table Is Thus
An Introduction to General / Inorganic Chemistry Mendeleev’s Periodic Table of the Elements Dmitri Mendeleev born 1834 in the Soviet Union. In 1869 he organised the 63 known elements into a periodic table based on atomic masses. He predicted the existence and properties of unknown elements and pointed out accepted atomic weights that were in error. The periodic table is thus an arrangement of the elements in order of increasing atomic number. Elements are arranged in rows called periods. Elements with similar properties are placed in the same column. These columns are called groups. An Introduction to General / Inorganic Chemistry The modern day periodic table can be further divided into blocks. http://www.chemsoc.org/viselements/pages/periodic_table.html The s, p, d and f blocks This course only deals with the s and p blocks. The s block is concerned only with the filling of s orbitals and contains groups I and II which have recently been named 1 and 2. The p block is concerned only with the filling of p orbitals and contains groups III to VIII which have recently been named 13 to 18. An Introduction to General / Inorganic Chemistry Groups exist because the electronic configurations of the elements within each group are the same. Group Valence Electronic configuration 1 s1 2 s2 13 s2p1 14 s2p2 15 s2p3 16 s2p4 17 s2p5 18 s2p6 The type of chemistry exhibited by an element is reliant on the number of valence electrons, thus the chemistry displayed by elements within a given group is similar. Physical properties Elemental physical properties can also be related to electronic configuration as illustrated in the following four examples: An Introduction to General / Inorganic Chemistry 1. -

Atomic Structure the Periodic Table Amount of Substance Bonding And
1 | C h e m i s t r y Contents 2(a) Atomic Structure 3 2(b) The Periodic Table 14 2(c) Amount of Substance 18 2(d) Bonding and Structure 28 2(e) Enthalpy Changes 39 2 | C h e m i s t r y 2(a) – Atomic Structure Scientists working in any area of chemical industry or research require a firm understanding of atomic structure and electron configurations and their use in providing the fundamental basis for chemical structures and reactions. Radiographers, environmental chemists and archaeologists all make use of specific isotopes in their work. Analytical chemists use UV/visible spectra and flame emission spectra to help characterise substances and colorimetry as a quantitative analytical technique. The origin of colour in compounds is of great importance in the dye-, pigment-, and paint-based industries and to development chemists researching new products. The Atom All elements are made of atoms. Atoms are made up of 3 types of particle. These are protons, neutrons and electrons. Electrons have -1 charge. They move around the nucleus in orbitals, which take up most of the volume of the atom. Most of the mass of the atom is concentrated in the nucleus. The nucleus is very small in comparison to the whole atom. The nucleus is where you find the protons and neutrons. The mass and charge of these subatomic particles is really small, so relative mass and relative charge are used instead. The table shows the relative masses and charges of protons, neutrons and electrons. Subatomic particle Relative mass Relative Charge Proton 1 +1 Neutron 1 0 Electron 1/2000 -1 3 | C h e m i s t r y Atomic Number and Mass Number You can figure out the number of protons, neutrons and electrons in an atom from the nuclear symbol. -

Make an Atom Vocabulary Grade Levels
MAKE AN ATOM Fundamental to physical science is a basic understanding of the atom. Atoms are comprised of protons, neutrons, and electrons. Protons and neutrons are at the center of the atom while electrons live in lobe-shaped clouds outside the nucleus. The number of electrons usually matches the number of protons, yielding a net neutral charge for the atom. Sometimes an atom has less neutrons or more neutrons than protons. This is called an isotope. If an atom has different numbers of electrons than protons, then it is an ion. If an atom has different numbers of protons, it is a different element all together. Scientists at Idaho National Laboratory study, create, and use radioactive isotopes like Uranium 234. The 234 means this isotope has an atomic mass of 234 Atomic Mass Units (AMU). GRADE LEVELS: 3-8 VOCABULARY Atom – The basic unit of a chemical element. Proton – A stable subatomic particle occurring in all atomic nuclei, with a positive electric charge equal in magnitude to that of an electron, but of opposite sign. Neutron – A subatomic particle of about the same mass as a proton but without an electric charge, present in all atomic nuclei except those of ordinary hydrogen. Electron – A stable subatomic particle with a charge of negative electricity, found in all atoms and acting as the primary carrier of electricity in solids. Orbital – Each of the actual or potential patterns of electron density that may be formed on an atom or molecule by one or more electrons. Ion – An atom or molecule with a net electric charge due to the loss or gain of one or more electrons. -

Atomic Weights of the Elements 1989
Atomic Weights of the Elements 1989 Cite as: Journal of Physical and Chemical Reference Data 20, 1313 (1991); https://doi.org/10.1063/1.555902 Submitted: 03 June 1991 . Published Online: 15 October 2009 J. R. De Laeter, and K. G. Heumann ARTICLES YOU MAY BE INTERESTED IN The Solubility of Carbon Dioxide in Water at Low Pressure Journal of Physical and Chemical Reference Data 20, 1201 (1991); https:// doi.org/10.1063/1.555900 Chemical Kinetic Data Sheets for High-Temperature Reactions. Part II Journal of Physical and Chemical Reference Data 20, 1211 (1991); https:// doi.org/10.1063/1.555901 Atomic Weights of the Elements 1991 Journal of Physical and Chemical Reference Data 22, 1571 (1993); https:// doi.org/10.1063/1.555933 Journal of Physical and Chemical Reference Data 20, 1313 (1991); https://doi.org/10.1063/1.555902 20, 1313 © 1991 American Institute of Physics for the National Institute of Standards and Technology. a Atomic Weights of the Elements 1989 ) J. R. De Laeter Curtin UlliversityofTechnology, Perth, Western Australia, 6001, Australia K. G. Heumann University ofRegensburg, Regensburg, Germany Received June 3, 1991 The biennial review of atomic weight, A r (E), determinations, and other cognate data has resulted in changes for nickel from 58.69 ± 0.01 to 58.6934 ± 0.0002 and for antimo ny from 121.75 ± 0.03 to 121.757 ± 0.003 due to new calibrated measurements. Because the measurement of the isotopic composition of mercury has also been improved during the last two years, the Commission was able to reduce the uncertainty of the atomic weight of this eJement from 200.59 ± 0.03 to 200.59 ± 0.02.