Some Aspects of Ecology and Genetics of Chironomidae (Diptera) in Rice Field and the Effect of Selected Herbicides on Its Population
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

ARTHROPOD COMMUNITIES and PASSERINE DIET: EFFECTS of SHRUB EXPANSION in WESTERN ALASKA by Molly Tankersley Mcdermott, B.A./B.S
Arthropod communities and passerine diet: effects of shrub expansion in Western Alaska Item Type Thesis Authors McDermott, Molly Tankersley Download date 26/09/2021 06:13:39 Link to Item http://hdl.handle.net/11122/7893 ARTHROPOD COMMUNITIES AND PASSERINE DIET: EFFECTS OF SHRUB EXPANSION IN WESTERN ALASKA By Molly Tankersley McDermott, B.A./B.S. A Thesis Submitted in Partial Fulfillment of the Requirements for the Degree of Master of Science in Biological Sciences University of Alaska Fairbanks August 2017 APPROVED: Pat Doak, Committee Chair Greg Breed, Committee Member Colleen Handel, Committee Member Christa Mulder, Committee Member Kris Hundertmark, Chair Department o f Biology and Wildlife Paul Layer, Dean College o f Natural Science and Mathematics Michael Castellini, Dean of the Graduate School ABSTRACT Across the Arctic, taller woody shrubs, particularly willow (Salix spp.), birch (Betula spp.), and alder (Alnus spp.), have been expanding rapidly onto tundra. Changes in vegetation structure can alter the physical habitat structure, thermal environment, and food available to arthropods, which play an important role in the structure and functioning of Arctic ecosystems. Not only do they provide key ecosystem services such as pollination and nutrient cycling, they are an essential food source for migratory birds. In this study I examined the relationships between the abundance, diversity, and community composition of arthropods and the height and cover of several shrub species across a tundra-shrub gradient in northwestern Alaska. To characterize nestling diet of common passerines that occupy this gradient, I used next-generation sequencing of fecal matter. Willow cover was strongly and consistently associated with abundance and biomass of arthropods and significant shifts in arthropod community composition and diversity. -

Dear Colleagues
NEW RECORDS OF CHIRONOMIDAE (DIPTERA) FROM CONTINENTAL FRANCE Joel Moubayed-Breil Applied ecology, 10 rue des Fenouils, 34070-Montpellier, France, Email: [email protected] Abstract Material recently collected in Continental France has allowed me to generate a list of 83 taxa of chironomids, including 37 new records to the fauna of France. According to published data on the chironomid fauna of France 718 chironomid species are hitherto known from the French territories. The nomenclature and taxonomy of the species listed are based on the last version of the Chironomidae data in Fauna Europaea, on recent revisions of genera and other recent publications relevant to taxonomy and nomenclature. Introduction French territories represent almost the largest Figure 1. Major biogeographic regions and subregions variety of aquatic ecosystems in Europe with of France respect to both physiographic and hydrographic aspects. According to literature on the chironomid fauna of France, some regions still are better Sites and methodology sampled then others, and the best sampled areas The identification of slide mounted specimens are: The northern and southern parts of the Alps was aided by recent taxonomic revisions and keys (regions 5a and 5b in figure 1); western, central to adults or pupal exuviae (Reiss and Säwedal and eastern parts of the Pyrenees (regions 6, 7, 8), 1981; Tuiskunen 1986; Serra-Tosio 1989; Sæther and South-Central France, including inland and 1990; Soponis 1990; Langton 1991; Sæther and coastal rivers (regions 9a and 9b). The remaining Wang 1995; Kyerematen and Sæther 2000; regions located in the North, the Middle and the Michiels and Spies 2002; Vårdal et al. -

The Nestling Diet of Svalbard Snow Buntings Identified by DNA Metabarcoding
Faculty of Biosciences, Fisheries and Economics, Department of Arctic and Marine Biology The nestling diet of Svalbard snow buntings identified by DNA metabarcoding — Christian Stolz BIO-3950 Master thesis in Biology, Northern Populations and Ecosystems, May 2019 Faculty of Biosciences, Fisheries and Economics, Department of Arctic and Marine Biology The nestling diet of Svalbard snow buntings identified by DNA metabarcoding Christian Stolz, UiT The Arctic University of Norway, Tromsø, Norway and The University Centre in Svalbard (UNIS), Longyearbyen, Norway BIO-3950 Master Thesis in Biology, Northern Populations and Ecosystems, May 2018 Supervisors: Frode Fossøy, Norwegian Institute for Nature Research (NINA), Trondheim, Norway Øystein Varpe, The University Centre in Svalbard (UNIS), Longyearbyen, Norway Rolf Anker Ims, UiT The Arctic University of Norway, Tromsø, Norway i Abstract Tundra arthropods have considerable ecological importance as a food source for several bird species that are reproducing in the Arctic. The actual arthropod taxa comprising the chick diet are however rarely known, complicating assessments of ecological interactions. In this study, I identified the nestling diet of Svalbard snow bunting (Plectrophenax nivalis) for the first time. Faecal samples of snow bunting chicks were collected in Adventdalen, Svalbard in the breeding season 2018 and analysed via DNA metabarcoding. Simultaneously, the availability of prey arthropods was measured via pitfall trapping. The occurrence of 32 identified prey taxa in the nestling diet changed according to varying abundances and emergence patterns within the tun- dra arthropod community: Snow buntings provisioned their offspring mainly with the most abundant prey items which were in the early season different Chironomidae (Diptera) taxa and Scathophaga furcata (Diptera: Scathophagidae), followed by Spilogona dorsata (Diptera: Mus- cidae). -

Austroconops Wirth and Lee, a Lower Cretaceous Genus of Biting Midges
PUBLISHED BY THE AMERICAN MUSEUM OF NATURAL HISTORY CENTRAL PARK WEST AT 79TH STREET, NEW YORK, NY 10024 Number 3449, 67 pp., 26 ®gures, 6 tables August 23, 2004 Austroconops Wirth and Lee, a Lower Cretaceous Genus of Biting Midges Yet Living in Western Australia: a New Species, First Description of the Immatures and Discussion of Their Biology and Phylogeny (Diptera: Ceratopogonidae) ART BORKENT1 AND DOUGLAS A. CRAIG2 ABSTRACT The eggs and all four larval instars of Austroconops mcmillani Wirth and Lee and A. annettae Borkent, new species, are described. The pupa of A. mcmillani is also described. Life cycles and details of behavior of each life stage are reported, including feeding by the aquatic larvae on microscopic organisms in very wet soil/detritus, larval locomotion, female adult biting habits on humans and kangaroos, and male adult swarming. Austroconops an- nettae Borkent, new species, is attributed to the ®rst author. Cladistic analysis shows that the two extant Austroconops Wirth and Lee species are sister species. Increasingly older fossil species of Austroconops represent increasingly earlier line- ages. Among extant lineages, Austroconops is the sister group of Leptoconops Skuse, and together they form the sister group of all other Ceratopogonidae. Dasyhelea Kieffer is the sister group of Forcipomyia Meigen 1 Atrichopogon Kieffer, and together they form the sister group of the Ceratopogoninae. Forcipomyia has no synapomorphies and may be paraphyletic in relation to Atrichopogon. Austroconops is morphologically conservative (possesses many plesiomorphic features) in each life stage and this allows for interpretation of a number of features within Ceratopogonidae and other Culicomorpha. A new interpretation of Cretaceous fossil lineages shows that Austroconops, Leptoconops, Minyohelea Borkent, Jordanoconops 1 Royal British Columbia Museum, American Museum of Natural History, and Instituto Nacional de Biodiversidad. -

(Insecta: Diptera) Assemblages Along an Andean Altitudinal Gradient
Vol. 20: 169–184, 2014 AQUATIC BIOLOGY Published online April 9 doi: 10.3354/ab00554 Aquat Biol Chironomid (Insecta: Diptera) assemblages along an Andean altitudinal gradient Erica E. Scheibler1,*, Sergio A. Roig-Juñent1, M. Cristina Claps2 1IADIZA, CCT CONICET Mendoza. Avda. Ruiz Leal s/n. Parque Gral. San Martín, CC 507, 5500 Mendoza, Argentina 2ILPLA, CCT CONICET La Plata, Boulevard 120 y 62 1900 La Plata, Argentina ABSTRACT: Chironomidae are an abundant, diverse and ecologically important group common in mountain streams worldwide, but their patterns of distribution have been poorly described for the Andes region in western-central Argentina. Here we examine chironomid assemblages along an altitudinal gradient in the Mendoza River basin to study how spatial and seasonal variations affect the abundance patterns of genera across a gradient of elevation, assess the effects of envi- ronmental variables on the chironomid community and to describe its diversity using rarefaction and Shannon’s indices. Three replicate samples and physicochemical parameters were measured seasonally at 11 sites in 2000 and 2001. Twelve genera of chironomid larvae were identified, which belonged to 5 subfamilies. Chironomid composition changed from the headwaters to the outlet and was associated with changes in altitude, water temperature, substrate size and conduc- tivity. We found a pronounced seasonal and spatial variation in the macroinvertebrate community and in physicochemical parameters. Environmental conditions such as elevated conductivity levels and increased river discharge occurring during the summer produced low chironomid density values at the sampling sites. The rarefaction index revealed that the sampling sites with highest richness were LU (middle section of the river) and PO (lower section). -

Diptera: Chironomidae) with Terrestrial and Semiaqutic Larvae from Ukraine
Українська ентомофауністика 2011, 2(5) : 13–15 Дата публікації: 31.10.2011 New and additional records of the non-biting midges (Diptera: Chironomidae) with terrestrial and semiaqutic larvae from Ukraine. V. A. Baranov Dept. of Zoology and Animal Ecology V.N. Karazin Kharkiv National University 4 Svoboda Sq. 61077, Kharkiv, Ukraine Ukrainian Science Research Institute of Ecological Problems Bakulina St. 6, 61166, Kharkiv, Ukraine E-mail: [email protected] Baranov V. A. New records of the non-biting midges (Diptera: Chironomidae) with terrestrial and semiaqutic larvae from Ukraine. Summary. Seven species of chironomid midges, Bryophaenocladius furcatus (Kieffer, 1916), Hydrosmittia oxoniana (Edwards, 1922), Smittia edwardsi Goethgebuer, 1932 , Smittia foliacea (Kieffer, 1921), Paraphaenocladius penerasus (Edwards, 1929), Paraphaenocladius impensus (Walker, 1856), Psedosmittia forcipata (Goetghebuer, 1921), are recorded for the first time from Ukraine based on adult specimens and larvae. Camptocladius stercorarius (De Geer, 1776) is recorded for the second time. Keywords : Diptera, Chironomidae, non-biting midges, terrestrial larvae, adults, first record, Ukraine,. Баранов В. О. Нові знахідки комарів -дзвінців (Diptera: Chironomidae) з наземними та напівводними личинками з України. Ре - зюме. Вперше у фауні України відмічено сім видів комарів-дзвінців: Bryophaenocladius furcatus (Kieffer, 1916), Hydrosmittia oxoniana (Edwards, 1922), Smittia edwardsi Goethgebuer, 1932 , Smittia foliacea (Kieffer, 1921), Paraphaenocladius penerasus (Edwards, -
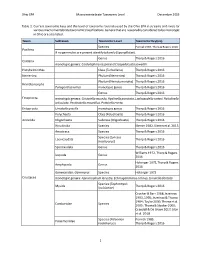
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 1 Table 1. Current Taxonomic Keys and the Level of Taxonomy Routinely U
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Table 1. Current taxonomic keys and the level of taxonomy routinely used by the Ohio EPA in streams and rivers for various macroinvertebrate taxonomic classifications. Genera that are reasonably considered to be monotypic in Ohio are also listed. Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Species Pennak 1989, Thorp & Rogers 2016 Porifera If no gemmules are present identify to family (Spongillidae). Genus Thorp & Rogers 2016 Cnidaria monotypic genera: Cordylophora caspia and Craspedacusta sowerbii Platyhelminthes Class (Turbellaria) Thorp & Rogers 2016 Nemertea Phylum (Nemertea) Thorp & Rogers 2016 Phylum (Nematomorpha) Thorp & Rogers 2016 Nematomorpha Paragordius varius monotypic genus Thorp & Rogers 2016 Genus Thorp & Rogers 2016 Ectoprocta monotypic genera: Cristatella mucedo, Hyalinella punctata, Lophopodella carteri, Paludicella articulata, Pectinatella magnifica, Pottsiella erecta Entoprocta Urnatella gracilis monotypic genus Thorp & Rogers 2016 Polychaeta Class (Polychaeta) Thorp & Rogers 2016 Annelida Oligochaeta Subclass (Oligochaeta) Thorp & Rogers 2016 Hirudinida Species Klemm 1982, Klemm et al. 2015 Anostraca Species Thorp & Rogers 2016 Species (Lynceus Laevicaudata Thorp & Rogers 2016 brachyurus) Spinicaudata Genus Thorp & Rogers 2016 Williams 1972, Thorp & Rogers Isopoda Genus 2016 Holsinger 1972, Thorp & Rogers Amphipoda Genus 2016 Gammaridae: Gammarus Species Holsinger 1972 Crustacea monotypic genera: Apocorophium lacustre, Echinogammarus ischnus, Synurella dentata Species (Taphromysis Mysida Thorp & Rogers 2016 louisianae) Crocker & Barr 1968; Jezerinac 1993, 1995; Jezerinac & Thoma 1984; Taylor 2000; Thoma et al. Cambaridae Species 2005; Thoma & Stocker 2009; Crandall & De Grave 2017; Glon et al. 2018 Species (Palaemon Pennak 1989, Palaemonidae kadiakensis) Thorp & Rogers 2016 1 Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Informal grouping of the Arachnida Hydrachnidia Smith 2001 water mites Genus Morse et al. -

Checklist of the Family Chironomidae (Diptera) of Finland
A peer-reviewed open-access journal ZooKeys 441: 63–90 (2014)Checklist of the family Chironomidae (Diptera) of Finland 63 doi: 10.3897/zookeys.441.7461 CHECKLIST www.zookeys.org Launched to accelerate biodiversity research Checklist of the family Chironomidae (Diptera) of Finland Lauri Paasivirta1 1 Ruuhikoskenkatu 17 B 5, FI-24240 Salo, Finland Corresponding author: Lauri Paasivirta ([email protected]) Academic editor: J. Kahanpää | Received 10 March 2014 | Accepted 26 August 2014 | Published 19 September 2014 http://zoobank.org/F3343ED1-AE2C-43B4-9BA1-029B5EC32763 Citation: Paasivirta L (2014) Checklist of the family Chironomidae (Diptera) of Finland. In: Kahanpää J, Salmela J (Eds) Checklist of the Diptera of Finland. ZooKeys 441: 63–90. doi: 10.3897/zookeys.441.7461 Abstract A checklist of the family Chironomidae (Diptera) recorded from Finland is presented. Keywords Finland, Chironomidae, species list, biodiversity, faunistics Introduction There are supposedly at least 15 000 species of chironomid midges in the world (Armitage et al. 1995, but see Pape et al. 2011) making it the largest family among the aquatic insects. The European chironomid fauna consists of 1262 species (Sæther and Spies 2013). In Finland, 780 species can be found, of which 37 are still undescribed (Paasivirta 2012). The species checklist written by B. Lindeberg on 23.10.1979 (Hackman 1980) included 409 chironomid species. Twenty of those species have been removed from the checklist due to various reasons. The total number of species increased in the 1980s to 570, mainly due to the identification work by me and J. Tuiskunen (Bergman and Jansson 1983, Tuiskunen and Lindeberg 1986). -

Table of Contents 2
Southwest Association of Freshwater Invertebrate Taxonomists (SAFIT) List of Freshwater Macroinvertebrate Taxa from California and Adjacent States including Standard Taxonomic Effort Levels 1 March 2011 Austin Brady Richards and D. Christopher Rogers Table of Contents 2 1.0 Introduction 4 1.1 Acknowledgments 5 2.0 Standard Taxonomic Effort 5 2.1 Rules for Developing a Standard Taxonomic Effort Document 5 2.2 Changes from the Previous Version 6 2.3 The SAFIT Standard Taxonomic List 6 3.0 Methods and Materials 7 3.1 Habitat information 7 3.2 Geographic Scope 7 3.3 Abbreviations used in the STE List 8 3.4 Life Stage Terminology 8 4.0 Rare, Threatened and Endangered Species 8 5.0 Literature Cited 9 Appendix I. The SAFIT Standard Taxonomic Effort List 10 Phylum Silicea 11 Phylum Cnidaria 12 Phylum Platyhelminthes 14 Phylum Nemertea 15 Phylum Nemata 16 Phylum Nematomorpha 17 Phylum Entoprocta 18 Phylum Ectoprocta 19 Phylum Mollusca 20 Phylum Annelida 32 Class Hirudinea Class Branchiobdella Class Polychaeta Class Oligochaeta Phylum Arthropoda Subphylum Chelicerata, Subclass Acari 35 Subphylum Crustacea 47 Subphylum Hexapoda Class Collembola 69 Class Insecta Order Ephemeroptera 71 Order Odonata 95 Order Plecoptera 112 Order Hemiptera 126 Order Megaloptera 139 Order Neuroptera 141 Order Trichoptera 143 Order Lepidoptera 165 2 Order Coleoptera 167 Order Diptera 219 3 1.0 Introduction The Southwest Association of Freshwater Invertebrate Taxonomists (SAFIT) is charged through its charter to develop standardized levels for the taxonomic identification of aquatic macroinvertebrates in support of bioassessment. This document defines the standard levels of taxonomic effort (STE) for bioassessment data compatible with the Surface Water Ambient Monitoring Program (SWAMP) bioassessment protocols (Ode, 2007) or similar procedures. -

Insecta Diptera) in Freshwater (Excluding Simulidae, Culicidae, Chironomidae, Tipulidae and Tabanidae) Rüdiger Wagner University of Kassel
Entomology Publications Entomology 2008 Global diversity of dipteran families (Insecta Diptera) in freshwater (excluding Simulidae, Culicidae, Chironomidae, Tipulidae and Tabanidae) Rüdiger Wagner University of Kassel Miroslav Barták Czech University of Agriculture Art Borkent Salmon Arm Gregory W. Courtney Iowa State University, [email protected] Follow this and additional works at: http://lib.dr.iastate.edu/ent_pubs BoudewPart ofijn the GoBddeeiodivrisersity Commons, Biology Commons, Entomology Commons, and the TRoyerarle Bestrlgiialan a Indnstit Aquaute of Nticat uErcaol Scienlogyce Cs ommons TheSee nex tompc page forle addte bitioniblaiol agruthorapshic information for this item can be found at http://lib.dr.iastate.edu/ ent_pubs/41. For information on how to cite this item, please visit http://lib.dr.iastate.edu/ howtocite.html. This Book Chapter is brought to you for free and open access by the Entomology at Iowa State University Digital Repository. It has been accepted for inclusion in Entomology Publications by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. Global diversity of dipteran families (Insecta Diptera) in freshwater (excluding Simulidae, Culicidae, Chironomidae, Tipulidae and Tabanidae) Abstract Today’s knowledge of worldwide species diversity of 19 families of aquatic Diptera in Continental Waters is presented. Nevertheless, we have to face for certain in most groups a restricted knowledge about distribution, ecology and systematic, -

Jackson & Nelson R2 Figs
Fine tuning of vision-based prey-choice decisions by a predator that targets malaria vectors Running Head: PREY PREFERENCE OF A SPIDER THAT TARGETS MALARIA VECTORS Ximena J. NELSON1*, Robert R. JACKSON1,2 1School of Biological Sciences, University of Canterbury, Private Bag 4800, Christchurch, New Zealand. 2International Centre of Insect Physiology and Ecology, Thomas Odhiambo Campus, P.0. Box 30 Mbita Point 40350, Kenya. *Email: [email protected] Phone: 64-3-3642987 extn. 4050 Fax: 64-3-3642590 Key words: foraging, mosquito, predation, prey preference, salticid, specialization ABSTRACT. Evarcha culicivora is a jumping spider (Salticidae) that has the distinction of being the only predator known to express an active preference for the vectors of human malaria (i.e., the mosquito genus Anopheles) and to feed indirectly on blood by choosing blood-carrying female mosquitoes as prey. Here we examine this predator’s preference profile in greater detail than has been achieved before. Lures (dead prey mounted in life-like posture) were made from two mosquitoes (Anopheles gambiae and Culex quinquefasciatus) and a non-biting midge (Clinotanypus claripennis). Testing protocols were simultaneous-presentation (two prey presented simultaneously), alternate-day (two prey, each presented singly but on alternate days) and alternative-prey (second prey presented while test spider feeding on first prey). Pre-trial fasts were 1, 7, 15 and 21 days. Findings from this combination of variables were used to estimate strengths of preferences. Besides confirming the preference of E. culicivora for blood meals and for Anopheles in particular, we provide the first evidence of a preference, independent of blood meals, for female instead of male mosquitoes. -

Structure of the Coxa and Homeosis of Legs in Nematocera (Insecta: Diptera)
Acta Zoologica (Stockholm) 85: 131–148 (April 2004) StructureBlackwell Publishing, Ltd. of the coxa and homeosis of legs in Nematocera (Insecta: Diptera) Leonid Frantsevich Abstract Schmalhausen-Institute of Zoology, Frantsevich L. 2004. Structure of the coxa and homeosis of legs in Nematocera Kiev-30, Ukraine 01601 (Insecta: Diptera). — Acta Zoologica (Stockholm) 85: 131–148 Construction of the middle and hind coxae was investigated in 95 species of Keywords: 30 nematoceran families. As a rule, the middle coxa contains a separate coxite, Insect locomotion – Homeotic mutations the mediocoxite, articulated to the sternal process. In most families, this coxite – Diptera – Nematocera is movably articulated to the eucoxite and to the distocoxite area; the coxa is Accepted for publication: radially split twice. Some groups are characterized by a single split. 1 July 2004 The coxa in flies is restricted in its rotation owing to a partial junction either between the meron and the pleurite or between the eucoxite and the meropleurite. Hence the coxa is fastened to the thorax not only by two pivots (to the pleural ridge and the sternal process), but at the junction named above. Rotation is impossible without deformations; the role of hinges between coxites is to absorb deformations. This adaptive principle is confirmed by physical modelling. Middle coxae of limoniid tribes Eriopterini and Molophilini are compact, constructed by the template of hind coxae. On the contrary, hind coxae in all families of Mycetophiloidea and in Psychodidae s.l. are constructed like middle ones, with the separate mediocoxite, centrally suspended at the sternal process. These cases are considered as homeotic mutations, substituting one structure with a no less efficient one.