Pharma &Biotech
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

In the United States District Court for the Eastern District of Pennsylvania ______: in Re Suboxone (Buprenorphine : Mdl No
IN THE UNITED STATES DISTRICT COURT FOR THE EASTERN DISTRICT OF PENNSYLVANIA __________________________________________ : IN RE SUBOXONE (BUPRENORPHINE : MDL NO. 2445 HYDROCHLORIDE AND NALOXONE) : 13-MD-2445 ANTITRUST LITIGATION : : THIS DOCUMENT RELATES TO:, : : Wisconsin, et al. v. Indivior Inc. et al. : Case No. 16-cv-5073 : __________________________________________: STATE OF WISCONSIN : By Attorney General Brad D. Schimel, et al. : : CIV. A. NO. 16-5073 Plaintiffs, : v. : : INDIVIOR INC. f/k/a RECKITT BENCKISER : PHARMACEUTICALS, INC., et al. : : Defendants. : __________________________________________: Goldberg, J. November 24, 2020 MEMORANDUM Defendant Reckitt Benckiser, Inc. (“Defendant”) manufactures Suboxone, a drug commonly used to combat opioid addiction.1 Suboxone previously came in tablet form, but in 2010, citing safety concerns, Defendant effectuated a change in the administration of this drug, switching from tablet to sublingual film. Various purchasers/consumers of Suboxone claimed that this switch was anticompetitive and solely designed to maintain Defendant’s market exclusivity—a scheme known as a “product hop.” These claims have resulted in multi-district, antitrust litigation before this Court. 1 Reckitt is currently known as Indivior, Inc. In December 2014, Reckitt Benckiser Pharmaceuticals, Inc. was demerged from its prior parent, the Reckitt Benckiser Group PLC, into Indivior PLC. Although Indivior is technically the named defendant in this case, the pleadings and many of the relevant exhibits use the name “Reckitt.” As discovery and class certification litigation have come to a close, the parties have raised numerous challenges under Daubert v. Merrell Dow Pharmaceuticals, 509 U.S. 579 (1993), seeking exclusion of all or selected portions of nine expert witnesses anticipated opinions. This Opinion explains my reasoning for the resolution of these motions and will hopefully set forth a clearer path towards trial. -

FREDERICK ('ERIC') RANDALL SMITH Bsc, Phd(Edin), FRSC Eric
FREDERICK ('ERIC') RANDALL SMITH BSc, PhD(Edin), FRSC Eric Smith was responsible for research activities in Duncan Flockhart Ltd, T & H Smith Ltd and Edinburgh Pharmaceutical Industries Ltd during the period 1947 to 1971. He and his colleagues made several lasting contributions to medicine and commerce notable amongst which were the creation of a new opiate industry based on large scale poppy growing in Australia, the establishment of dihydrocodeine ('DF118') as a major analgesic, and the discovery of 'Bitrex', an intensely bitter substance, now a widely used denaturant. He was elected a Fellow of the Society in 1962 and served on Council 1969-71. Born in Edinburgh on April 4th, 1911, Eric spent his childhood, his school and university days, and most of his working life and retirement in or near to the City. He died there on November 4th, 1994. Eric's enquiring attitude to life owed much to his father. This unusual man earned his living in the Actuarial Department of the Scottish Provident Institution but his main interests were astronomy and art. He was an FRAS and his personal observations led to his name being given to a minor lunar crater, and a good enough artist for his works to be hung in the Royal Academies in Edinburgh and London. Perhaps this is why Eric chose to be a scientist and his sister an art teacher. Eric was educated at the Edinburgh Institution (later named Melville College) where he was an excellent scholar and a good enough sportsman to play for the 1st XV and the FPs. -

Morphinone Reductase for the Preparation Of
Europäisches Patentamt *EP000649465B1* (19) European Patent Office Office européen des brevets (11) EP 0 649 465 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.7: C12N 9/02, C12P 17/18, of the grant of the patent: C12Q 1/32 24.07.2002 Bulletin 2002/30 (86) International application number: (21) Application number: 93913236.1 PCT/GB93/01129 (22) Date of filing: 28.05.1993 (87) International publication number: WO 94/00565 (06.01.1994 Gazette 1994/02) (54) MORPHINONE REDUCTASE FOR THE PREPARATION OF HYDROMORPHONE AND HYDROCODONE MORPHINONE REDUKTASE ZUR HERSTELLUNG VON HYDROMORPHONE UND HYDROCODONE MORPHINONE REDUCTASE DESTINEE A LA PREPARATION D’HYDROMORPHONE ET D’HYDROCODONE (84) Designated Contracting States: (74) Representative: Perry, Robert Edward AT BE CH DE DK ES FR GB IE IT LI NL PT SE GILL JENNINGS & EVERY Broadgate House (30) Priority: 25.06.1992 GB 9213524 7 Eldon Street London EC2M 7LH (GB) (43) Date of publication of application: 26.04.1995 Bulletin 1995/17 (56) References cited: WO-A-90/13634 (73) Proprietor: MACFARLAN SMITH LIMITED Edinburgh EH11 2QA (GB) • THE BIOCHEMICAL JOURNAL vol. 274, no. 3, 15 March 1991, pages 875 - 880 NEIL C. BRUCE ET (72) Inventors: AL. ’Microbial degradation of the morphine • HAILES, Anne, Maria 59 Lower Road alkaloids. Purification and characterization of Kent DA8 1AY (GB) morphine dehydrogenase from Pseudomonas • FRENCH, Christopher, Edward putida M10’ Palmerston North (NZ) • BRUCE, Neil, Charles Cambridge CB2 1ND (GB) Note: Within nine months from the publication of the mention of the grant of the European patent, any person may give notice to the European Patent Office of opposition to the European patent granted. -

PHARMACEUTICALS + Biovian Oy Jemedic AB Bayer Oy + Shetland Bergen Helse Bergen HF, Haukeland 3
BARENTS SEA Jan Mayen (Norway) Tromsø NORWAY Murmansk DENMARK STRAIT NORWEGIAN SEA Raufarhöfn Bolungarvik Kalix Aromtech Oy Mosjøen SWEDEN Kemi Akureyri Luleå WHITE SEA Piteå Eskifjördur Oy Woikoski Ab + Pharmatory Ltd + Bioactive Bone Substitutes Oolu Fermion Oy Olafsvik ICELAND Skellefteå IA Hornafjördur Reykjavik 1-5 Grindavik Apotek Produktion & Laboratorier AB Oy Woikoski Ab Umeå Norrlands Universitetssjukhus Frøya Vik Trondheim Kristiansund GULF OF BOTHN Vaasa Averøya Galena Pharma Oy + Orion Oyj + Finvector Oy Sandøy Härnösand Ålesund Sundsvall Unimedic AB + MAP Medical Technologies Oy Woikoski Ab Kristinestad Tórshavn Apoteksverkiö, Framleiösluapotekiö Måløy EWOS AS Santen Oy Cytomed Oy SWEDEN - Not Shown Vitabalans Oy + Umeå + Curida AS Apotek Produktion & Laboratorier AB Oy Aga Ab Norrlands Universitetssjukhus Jemedic AB FINLAND OSLO Gävle Alby Vyborg + Nouryon Pulp & Performance Chemicals AB Hankintatukku Oy 1. Smerud Medical Research Norway AS Pcas Finland Oy Finex Oy Hamina Orion Corporation Oy Woikoski Ab 2. Catapult Life Science AS Gävle + + EUROPE - PHARMACEUTICALS + Biovian Oy Jemedic AB Bayer Oy + Shetland Bergen Helse Bergen HF, Haukeland 3. PhotoCure ASA Turku Orion Corporation A.Vogel Oy universitetssjukehus 4. GE Healthcare AS Vitabalans Oy MAP Medicals Technologies Oy + Nanoform Finland Oy Islands 5. Oncoinvent AS Fermion Oy AGA Gas AB Lumene Oy Lerwick 6. The Cyclotron Center Pharmaq AS + Scan Aqua AS Orion Corporation + 7. Oslo Universitetssykehus HF HELSINKI 8. Diatec Monoclonals AS Institute for Energy Technology -

Introducing the IMED Biotech Unit What Science Can Do Introduction What Science Can Do
Introducing the IMED Biotech Unit What science can do Introduction What science can do At AstraZeneca, our purpose is to push the Our IMED Biotech Unit applies its research and Our approach to R&D development capabilities and technologies to The IMED Biotech Unit plays a critical boundaries of science to deliver life-changing accelerate the progress of our pipeline. Through role in driving AstraZeneca’s success. Working together with MedImmune, medicines. We achieve this by placing science great collaboration across our three science units, our global biologics arm and Global we are confident that we can deliver the next wave Medicines Development (GMD), our at the centre of everything we do. late-stage development organisation, of innovative medicines to transform the lives of we are ensuring we deliver an innovative patients around the world. and sustainable pipeline. Pancreatic beta cells at different Eosinophil prior to Minute pieces of circulating tumour DNA stages of regeneration apoptosis (ctDNA) in the bloodstream IMED Biotech Unit MedImmune Global Medicines Development Focuses on driving scientific advances Focuses on biologics research and Focuses on late-stage development in small molecules, oligonucleotides and development in therapeutic proteins, of our innovative pipeline, transforming other emerging platforms to push the monoclonal antibodies and other next- exciting science into valued new boundaries of medical science. generation molecules to attack a range medicines and ensuring patients of diseases. around the world can access them. It’s science that compels us to push the boundaries of what is possible. We trust in the potential of ideas and pursue them, alone and with others, until we have transformed the treatment of disease. -

United States Securities and Exchange Commission Form 10-K Shire Pharmaceuticals Group
UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, D.C. 20549 FORM 10-K (Mark One) ፤ ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 For the fiscal year ended December 31, 1999 អ TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 Commission file number 0-29630 SHIRE PHARMACEUTICALS GROUP PLC (Exact name of registrant as specified in its charter) England and Wales (State or other jurisdiction (I.R.S. Employer of incorporation or organization) Identification No.) N.A. East Anton, Andover, Hampshire SP10 5RG England (Address of principal executive offices) (Zip Code) 44 1264 333455 (Registrant's telephone number, including area code) Securities registered pursuant to Section 12(b) of the Act: Title of each class Name of exchange on which registered American Depository Shares, each representing Nasdaq National Market 3 Ordinary Shares, 5 pence nominal value per share Securities registered pursuant to Section 12(g) of the Act: None (Title of class) Indicate by check mark whether the Registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the Registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ፤ No អ Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of the Registrant's knowledge, in definitive proxy or information statements incorporated by reference to Part III of this Form 10-K or any amendment to this Form 10-K. -

August/September 2016
INTERNATIONAL PHARMACEUTICAL QUALITY IPQ Inside the Global Regulatory Dialogue IPQPUBS.COM VOL. 8, NO. 5 MONTHLY UPDATE - AUGUST/SEPTEMBER 2016 UNITED STATES ● Review/Inspection Control Strategy Communications Key to Advancing Biotech p. 4 Regulation, FDA Biotech Official Stresses ● More Clarity Sought in FDA’s Inactive Ingredient Database Regarding Drug Delivery Devices p. 13 EUROPE ● Protein Sequence Variants, Process Development and Spec Setting Are Generating Queries p. 20 from EU Biopharmaceutical CMC Assessors ● Procedures, Eligibilities and Goals of EMA’s Accelerated Access Efforts Are Taking More p. 32 Concrete Shape INTERNATIONAL ● Goal of FDA/EU Mutual Inspection Reliance is Nearing Realization p. 36 UPDATES IN BRIEF p. 47 U.S. CMC: ● GDUFA II Agreement ● ANDA Impurity Refusals ● Capsule Supplier Changes ● Co-Crystal Classification ● ANDA Impurity Refusals U.S. GMP: ● Insanitary Compounding ● NIH Clinical Manufacturing ● Establishment Registration ● Generics Facility ID'ing ● USP on Analytical Lifecycles EUROPE CMC: ● EMA Streamlining ● EMA Adaptive Pathways Workshop ● EDQM on ICH Q3D EUROPE GMP: ● EMA WFI Q&A ● EC on Plastics INTERNATIONAL CMC: ● Antimicrobial Resistance Roadmap ● WHO on Variations ● Canada on ICH Q3D INTERNATIONAL GMP: ● China on Drug Manufacturing ● India Manufacturing Self-Assessment FDA WARNING LETTERS, EMA NON-COMPLIANCE REPORTS, AND FDA RECALLS POSTED IN AUGUST/SEPTEMBER p. 51 MONTHLY UPDATE - AUGUST / SEPTEMBER 2016 INTERNATIONAL PHARMACEUTICAL QUALITY provides in-depth coverage of emerging drug, biologic and combination product CMC and GMP issues and developments with a mission of helping advance and harmonize the quality regulatory process globally. Headquartered in Washington, D.C., IPQ is read by regulatory agencies, manufacturers, suppliers, consultants, law firms, and universities around the world. -

United States Patent Office Patented Aug
3,830,819 United States Patent Office Patented Aug. 20, 1974 2 The invention provides a process for the purification of l-dihydrocodeine containing as impurities 1-dihydrothebai 3,830,819 MANUFACTURE OF l-DHYDROCODENE none and or 1-dihydrothebainol, which comprises treating Edward Leon Grew and David Jackson Powles, Edin the impure 1-dihydrocodeine to remove said impurities burgh, Scotland, assignors to MacFarlan Smith Limited, therefrom by either extraction with an aqueous alkali or Edinburgh, Scotland 5 by passage of the material through an anion exchange No Drawing. Fied Feb. 22, 1972, Ser. No. 228,367 resin of a quaternary ammonium type in the hydroxide Claims priority, application Great Britain, Feb. 22, 1971, form whereby the proportion of said impurities is reduced. 5,081/71 The crude 1-dihydrocodeine may be one prepared by Int, C. C07d 43/28 the catalytic hydrogenation of 1-dihydrocodeinone and ac U.S. C. 260-285 3 Claims 0. cording to a feature of the invention, there is therefore provided a process for the preparation of a purified 1-di ABSTRACT OF THE DESCLOSURE hydrocodeine which comprises the steps of A process for the preparation of a purified 1-dihydro (a) catalytically hydrogenating 1-dihydrocodeinone in a codeine which comprises the steps of: 5 liquid medium using a platinum oxide or supported (a) catalytically hydrogenating 1-dihydrocodeinone in a platinum metal catalyst to produce a solution of crude liquid medium using a catalyst selected from the group l-dihydrocodeine, consisting of platinum oxide and supported platinum (b) removing the catalyst from the solution, and metal to produce a solution of crude 1-dihydrocodeine, (c) treating the solution for the recovery of a purified (b) removing the catalyst from the solution, and 20 1-dihydrocodeine having a reduced content of l-dihy (c) treating the solution for recovery of a purified 1-di drothebainone and/or 1-dihydrothebainol. -
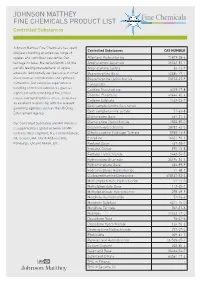
JOHNSON MATTHEY FINE CHEMICALS PRODUCT LIST Controlled Substances
JOHNSON MATTHEY FINE CHEMICALS PRODUCT LIST Controlled Substances Johnson Matthey Fine Chemicals has spent Controlled Substances CAS NUMBER 200 years building an extensive range of opiates and controlled substance. Our Alfentanil Hydrochloride 70879-28-6 heritage includes Macfarlan Smith Ltd, the Amphetamine Aspartate 25333-81-7 world’s leading manufacturer of opiate Amphetamine Sulfate 60-13-9 alkaloids. Additionally we specialise in other Buprenorphine Base 52485-79-7 areas such as cannabinoids and synthetic Buprenorphine Hydrochloride 53152-21-9 stimulants. Our extensive experience in Cannabidiol - handling controlled substances gives us Codeine Monohydrate 6059-47-8 significant understanding of the critical Codeine Phosphate 41444-62-6 issues surrounding these areas, as well as Codeine Sulphate 1420-53-7 an excellent relationship with the relevant Dextroamphetamine Saccharate - governing agencies such as the US Drug Dextroamphetamine Sulfate 51-63-8 Enforcement Agency. Diamorphine Base 561-27-3 Our Controlled Substance and API Portfolio Diamorphine Hydrochloride 1502-95-0 is supported by a global network of GMP Difenoxin Hydrochloride 28782-42-5 facilities: West Deptford, NJ; Conshohocken, Dihydrocodeine Hydrogen Tartrate 5965-13-9 PA; Devens, MA; North Andover, MA; Etorphine 14521-96-1 Edinburgh, UK and Annan, UK. Fentanyl Base 437-38-7 Fentanyl Citrate 990-73-8 Fentanyl Hydrochloride 1443-54-5 Hydrocodone Bitartrate 34195-34-1 Hydromorphone Base 466-99-9 Hydromorphone Hydrochloride 71-68-1 Lisdexamfetamine Dimesylate 608137-33-3 -

Government's Response to OFT's Review of Undertakings by Macfarlan Smith Limited
Government’s Response to OFT’s Review of Undertakings by MacFarlan Smith Limited (MSL) Market Analysis Opium in the UK – Analysis to date March 08 Sections in this report I) Background II) Home Office Undertaking III) Market Structure IV) Conclusions from Previous Market Analysis V) Taking the Analysis Forwards VI) Conclusions To Date Annex A: Copy of text from letter written by the EU Commission to EU Parliament re Opiates Annex B: Summary of responses to Home Office data request. I) Background a) OFT Review In 2006 the OFT published a report: Opium Derivatives, A Review of the Undertakings Given by Macfarlan Smith (MSL)1. This report recommended that the Government should consider competition when setting licensing policy for opium derivatives. b) Government Response The Department for Business, Enterprise and Regulatory Reform (BERR) has a general interest in OFT's market studies and co-ordinates Government responses where those studies make regulatory recommendations. Consideration of regulatory and policy issues is a matter for the relevant Department. In its response to the OFT2, the Government agreed to: Undertake to analyse the effects of MSL’s position as sole supplier on customers and competition, contrasting this with the positions of customers in other countries (both those who impose significant restrictions and those who do not). c) Work Undertaken by ERA & Drugs Liaison & Enforcement Unit (DLEU) BERR commissioned the Home Office to take forwards this market analysis. It was agreed that, at this stage, rather than repeat the OFT’s work a few years on (which was largely based on a survey of MSL’s UK customers) it made more sense to try and take the analysis forwards. -

Astrazeneca Plc
6/15/2020 AstraZeneca - Wikipedia AstraZeneca AstraZeneca plc[3] is a British-Swedish multinational pharmaceutical and biopharmaceutical company with its global AstraZeneca plc headquarters in Cambridge, England.[4] Its R&D is concentrated in Cambridge, Gaithersburg, Maryland, and Mölndal in Sweden.[5] AstraZeneca has a portfolio of products for major disease areas including cancer, cardiovascular, gastrointestinal, infection, Type Public limited neuroscience, respiratory and inflammation.[6] company Traded as LSE: AZN (https:// The company was founded in 1999 through the merger of the www.londonstocke [7][8] Swedish Astra AB and the British Zeneca Group (itself formed xchange.com/exch by the demerger of the pharmaceutical operations of Imperial ange/searchengin Chemical Industries in 1993). Since the merger it has been among e/search.html?lang the world's largest pharmaceutical companies and has made =en&x=0&y=0&q= numerous corporate acquisitions, including Cambridge Antibody AZN) Technology (in 2006), MedImmune (in 2007), Spirogen (in 2013) NYSE: AZN (http and Definiens (by MedImmune in 2014). s://www.nyse.com/ quote/XNYS:AZN) AstraZeneca has a primary listing on the London Stock Exchange Nasdaq and is a constituent of the FTSE 100 Index. It has secondary listings Stockholm: AZN (ht on the New York Stock Exchange and the OMX exchange. tp://www.nasdaqom xnordic.com/aktier/ microsite?language Contents Id=1&Instrument=S SE3524) History FTSE 100 2000–06 Component 2007–12: The patent cliff and subsequent acquisitions ISIN GB0009895292 2013 -

Pharmaceuticals in the Environment
Pharmaceuticals in the Environment As a responsible healthcare company, we are committed to the health and safety of our society and planet. We make it a priority to effectively manage the risks associated with pharmaceuticals in the environment (PIE). 1 How do Pharmaceutical Products get into the Environment? Pharmaceuticals enter the environmental mainly as AstraZeneca supports actions based on scientific a result of patient use, where they can pass through evidence to address the challenges presented by PIE. our bodies and into waterways. Drug manufacture and These include: the improper disposal of unused medicines also add • Assessing the environmental risk of our medicines to the trace levels of pharmaceuticals in rivers, lakes, to support drug marketing authorisations soils, and, sometimes, drinking water. AstraZeneca recognises that, even in such low concentrations, • Actively managing the environmental risks resulting the risks associated with Pharmaceuticals in the from our manufacturing Environment (PIE) should be determined, minimised • Supporting industry and government efforts to and managed. improve medicine disposal programs and education • Co-sponsoring research to fill scientific gaps 88% to understand and mitigate the risks of PIE. as a result of patient use To learn more about the risks associated with PIE and what we are doing to manage them please click here. 2% attributed to waste Pharmaceuticals from production in the environment 10% from unused medicines that people don’t dispose of properly 2 Societal Concerns about PIE Trace amounts of pharmaceuticals have been detected in the environment for more than 20 years. As environmental monitoring expands and the methods for measuring pharmaceuticals in the environment become more sophisticated and detection limits get better, the geographical scope and number of pharmaceuticals measured in the environment will grow.