Genome-Wide Analysis of ATP-Binding
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Genotyping of Breech Flystrike Resource – Update
Project No: ON-00515 Contract No: PO4500010753 AWI Project Manager: Bridget Peachey Contractor Name: CSIRO Agriculture and Food Prepared by: Dr Sonja Dominik Publication Date: July 2019 Genotyping of breech flystrike resource – update Published by Australian Wool Innovation Limited, Level 6, 68 Harrington Street, THE ROCKS, NSW, 2000 This publication should only be used as a general aid and is not a substitute for specific advice. To the extent permitted by law, we exclude all liability for loss or damage arising from the use of the information in this publication. AWI invests in research, development, innovation and marketing activities along the global supply chain for Australian wool. AWI is grateful for its funding, which is primarily provided by Australian woolgrowers through a wool levy and by the Australian Government which provides a matching contribution for eligible R&D activities © 2019 Australian Wool Innovation Ltd. All rights reserved. Contents Executive Summary .................................................................................................................... 3 1 Introduction/Hypothesis .................................................................................................... 5 2 Literature Review ............................................................................................................... 6 3 Project Objectives .............................................................................................................. 8 4 Success in Achieving Objectives ........................................................................................ -

Evolutionary and Functional Studies on Microsporidian ATP-Binding
Infection, Genetics and Evolution 68 (2019) 136–144 Contents lists available at ScienceDirect Infection, Genetics and Evolution journal homepage: www.elsevier.com/locate/meegid Evolutionary and functional studies on microsporidian ATP-binding T cassettes: Insights into the adaptation of microsporidia to obligated intracellular parasitism Qiang Hea, Charles R. Vossbrinckc, Qiong Yangd, Xian-Zhi Menga, Jian Luoa, Guo-Qing Pana, ⁎ ⁎ Ze-Yang Zhoua,b, , Tian Lia, a State Key Laboratory of Silkworm Genome Biology, Southwest University, Chongqing 400715, China b College of Life Science, Chongqing Normal University, Chongqing 400047, China c Department of Soil and Water, The Connecticut Agricultural Experiment Station, 123 Huntington Street, New Haven, CT 06511, USA d Sericulture and Agri-Food Research Institute, Guangdong Academy of Agricultural Sciences, Guangzhou, China ARTICLE INFO ABSTRACT Keywords: ATP-binding cassette (ABC) transporters comprise the largest family of transmembrane proteins and are found in Microsporidian all domains of life. The ABCs are involved in a variety of biological processes and as exporters play important ABC transporter roles in multidrug resistance. However, the ABC transporters have not been addressed in microsporidia, which Genome are a very large group of obligate intracellular parasites that can infect nearly all animals, including humans. Evolution Here, a total of 234 ABC transporters were identified from 18 microsporidian genomes and classified intofive Nosema bombycis subfamilies, including 74 ABCBs, 2 ABCCs, 18 ABCEs, 15 ABCFs, 102 ABCGs and 23 uncategorized members. Two subfamilies, ABCA and ABCD, are found in most organisms, but lost in microsporidia. Phylogenetic analysis indicated that microsporidian ABCB and ABCG subfamilies expanded by recent gene duplications, which re- sulted in the two largest subfamilies in microsporidia. -

Polymorphisms of the Multidrug Pump ABCG2: a Systematic Review of Their Effect on Protein Expression, Function, and Drug Pharmacokinetics
1521-009X/46/12/1886–1899$35.00 https://doi.org/10.1124/dmd.118.083030 DRUG METABOLISM AND DISPOSITION Drug Metab Dispos 46:1886–1899, December 2018 Copyright ª 2018 by The American Society for Pharmacology and Experimental Therapeutics Minireview Polymorphisms of the Multidrug Pump ABCG2: A Systematic Review of Their Effect on Protein Expression, Function, and Drug Pharmacokinetics Niall Heyes, Parth Kapoor, and Ian D. Kerr School of Life Sciences, Queen’s Medical Centre, University of Nottingham, Nottingham, United Kingdom Received June 12, 2018; accepted September 20, 2018 Downloaded from ABSTRACT The widespread expression and polyspecificity of the multidrug to improve outcomes in cancer patients treated with tyrosine ABCG2 efflux transporter make it an important determinant of the kinase inhibitors, but the reasons for this are yet to be estab- pharmacokinetics of a variety of substrate drugs. Null ABCG2 lished, and this residue’s role in the mechanism of the protein is expression has been linked to the Junior blood group. Polymor- unexplored by current biochemical and structural approaches. phisms affecting the expression or function of ABCG2 may have Research into the less-common polymorphisms is confined to dmd.aspetjournals.org clinically important roles in drug disposition and efficacy. The in vitro studies, with several polymorphisms shown to decrease most well-studied single nucleotide polymorphism (SNP), Q141K resistance to anticancer agents such as SN-38 and mitoxantrone. (421C>A), is shown to decrease ABCG2 expression and activity, In this review, we present a systematic analysis of the effects of resulting in increased total drug exposure and decreased re- ABCG2 polymorphisms on ABCG2 function and drug pharmaco- sistance to various substrates. -

5' Untranslated Region Elements Show High Abundance and Great
International Journal of Molecular Sciences Article 0 5 Untranslated Region Elements Show High Abundance and Great Variability in Homologous ABCA Subfamily Genes Pavel Dvorak 1,2,* , Viktor Hlavac 2,3 and Pavel Soucek 2,3 1 Department of Biology, Faculty of Medicine in Pilsen, Charles University, 32300 Pilsen, Czech Republic 2 Biomedical Center, Faculty of Medicine in Pilsen, Charles University, 32300 Pilsen, Czech Republic; [email protected] (V.H.); [email protected] (P.S.) 3 Toxicogenomics Unit, National Institute of Public Health, 100 42 Prague, Czech Republic * Correspondence: [email protected]; Tel.: +420-377593263 Received: 7 October 2020; Accepted: 20 November 2020; Published: 23 November 2020 Abstract: The 12 members of the ABCA subfamily in humans are known for their ability to transport cholesterol and its derivatives, vitamins, and xenobiotics across biomembranes. Several ABCA genes are causatively linked to inborn diseases, and the role in cancer progression and metastasis is studied intensively. The regulation of translation initiation is implicated as the major mechanism in the processes of post-transcriptional modifications determining final protein levels. In the current bioinformatics study, we mapped the features of the 50 untranslated regions (50UTR) known to have the potential to regulate translation, such as the length of 50UTRs, upstream ATG codons, upstream open-reading frames, introns, RNA G-quadruplex-forming sequences, stem loops, and Kozak consensus motifs, in the DNA sequences of all members of the subfamily. Subsequently, the conservation of the features, correlations among them, ribosome profiling data as well as protein levels in normal human tissues were examined. The 50UTRs of ABCA genes contain above-average numbers of upstream ATGs, open-reading frames and introns, as well as conserved ones, and these elements probably play important biological roles in this subfamily, unlike RG4s. -

Identification of Candidate ATP-Binding Cassette Transporter
www.nature.com/scientificreports OPEN Identifcation of candidate ATP- binding cassette transporter gene family members in Diaphorina citri (Hemiptera: Psyllidae) via adult tissues transcriptome analysis Zhengbing Wang 1, Fajun Tian1, Lijun Cai2, Jie Zhang2, Jiali Liu1 & Xinnian Zeng1* The ATP-binding cassette (ABC) transporters exist in all living organisms and play major roles in various biological functions by transporting a wide variety of substrates across membranes. The functions of ABC transporters in drug resistance have been extensively studied in vertebrates; however, they are rarely characterized in agricultural pests. The Asian citrus psyllid, Diaphorina citri, is one of the most damaging pests of the Citrus genus because of its transmission of Huanglongbing, also known as Yellow Dragon disease. In this study, the next-generation sequencing technique was applied to research the ABC transporters of D. citri. Fifty-three ABC transporter genes were found in the RNA-Seq data, and among these ABC transporters, 4, 4, 5, 2, 1, 4, 18 and 15 ABC proteins belonged to the ABCA-ABCH subfamilies, respectively. Diferent expression profles of 52 genes between imidacloprid-resistant and imidacloprid-susceptible strains were studied by qRT-PCR; 5 ABCGs and 4 ABCHs were signifcantly upregulated in the imidacloprid-resistant strain. In addition, fve of the nine upregulated genes were widely expressed in adult tissues in spatial expression analysis. The results suggest that these genes may play key roles in this phenotype. In general, this study contributed to our current understanding of D. citri resistance to insecticides. Te ATP-binding cassette (ABC) transporter family is one of the largest families of membrane proteins and uni- versally exists in all living organisms on Earth1. -

ABCG4: a Novel Human White Family ABC-Transporter Expressed in the Brain and Eye
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Biochimica et Biophysica Acta 1591 (2002) 175–179 www.bba-direct.com Short sequence-paper ABCG4: a novel human white family ABC-transporter expressed in the brain and eye Susan Oldfield *, Christopher A. Lowry, Jon Ruddick, Stafford L. Lightman University Research Centre for Neuroendocrinology, BRI, University of Bristol, Marlborough Street, Bristol BS2 8HW, UK Received 24 July 2001; received in revised form 11 April 2002; accepted 24 May 2002 Abstract White family transporters are a group of ATP-binding cassette (ABC) proteins that show sequence homology to the Drosophila white gene product. The Drosophila protein is a subunit of heterodimeric transporters of precursors for eye-pigment synthesis. A novel, human member of this family (ABCG4) has been identified. Northern blotting shows that ABCG4 is expressed specifically in the brain and the eye. D 2002 Elsevier Science B.V. All rights reserved. Keywords: White gene; ABC-transporter; ABCG1; Sterol ATP-binding cassette (ABC) transporters form a large and its murine counterpart is known as ABCG1 or ABC8. superfamily of proteins that mediate the transport of a large Northern blotting has shown that the mRNAs encoding variety of diverse molecules across biological membranes in ABCG1 are present in several tissues, predominantly brain, an ATP-dependent manner [1]. The functional transporters spleen and lung [10]. contain two ATP-binding folds and two transmembrane The function of ABCG1 is unknown, although there is domains each with, usually, five or six membrane spanning evidence that suggests that it may have a role in cholesterol helices. -

Transcriptional and Post-Transcriptional Regulation of ATP-Binding Cassette Transporter Expression
Transcriptional and Post-transcriptional Regulation of ATP-binding Cassette Transporter Expression by Aparna Chhibber DISSERTATION Submitted in partial satisfaction of the requirements for the degree of DOCTOR OF PHILOSOPHY in Pharmaceutical Sciences and Pbarmacogenomies in the Copyright 2014 by Aparna Chhibber ii Acknowledgements First and foremost, I would like to thank my advisor, Dr. Deanna Kroetz. More than just a research advisor, Deanna has clearly made it a priority to guide her students to become better scientists, and I am grateful for the countless hours she has spent editing papers, developing presentations, discussing research, and so much more. I would not have made it this far without her support and guidance. My thesis committee has provided valuable advice through the years. Dr. Nadav Ahituv in particular has been a source of support from my first year in the graduate program as my academic advisor, qualifying exam committee chair, and finally thesis committee member. Dr. Kathy Giacomini graciously stepped in as a member of my thesis committee in my 3rd year, and Dr. Steven Brenner provided valuable input as thesis committee member in my 2nd year. My labmates over the past five years have been incredible colleagues and friends. Dr. Svetlana Markova first welcomed me into the lab and taught me numerous laboratory techniques, and has always been willing to act as a sounding board. Michael Martin has been my partner-in-crime in the lab from the beginning, and has made my days in lab fly by. Dr. Yingmei Lui has made the lab run smoothly, and has always been willing to jump in to help me at a moment’s notice. -
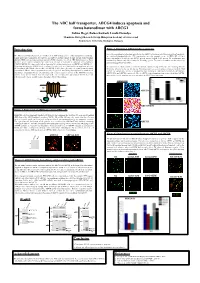
The ABC Half Transporter, ABCG4 Induces Apoptosis and Forms Heterodimer with ABCG1
The ABC half transporter, ABCG4 induces apoptosis and forms heterodimer with ABCG1 Zoltán Hegyi, Balázs Sarkadi, László Homolya Membrane Biology Research Group, Hungarian Academy of Sciences and Semmelweis University, Budapest, Hungary Introduction Figure 3. Functional ABCG4 induces apoptosis Since the morphological alterations observed in the ABCG4-expressing cells were indicative of apoptosis, The ABCG1 and ABCG4 proteins are members of the ATP binding cassette (ABC) transporter G subfamily. we examined phosphatidyl-serine (PS) translocation, an early apoptotic event in HEK293H cell lines Unlike most ABC transporters, the ABCG1 and ABCG4 proteins consist of only one nucleotide binding transiently transfected with the wt ABCG4 and its inactive mutant (KM) variant. PS translocation was domain (NBD) and one transmembrane domain (TMD), therefore are called ABC half-transporters. Some monitored by fluorescently labeled Annexin V binding (green). The total cell number was determined by members of the ABCG subfamily have proven to function as homodimers (ABCG2) or heterodimers nuclear staining by Hoechst (blue). (ABCG5/ABCG8). Previous results indicated potential heterodimerization between ABCG1 and ABCG4 (1). Regarding their function, ABCG1 has been proposed to play a role in cellular lipid/sterol regulation, whereas We found that the cultures transfected with wt ABCG4 contained a large number of cells exhibiting Annexin the function of ABCG4, the closest relative of ABCG1, is still elusive. Recently, we reported that functional V binding, whereas hardly any labeling for PS translocation was seen in cultures transfected with the KM expression of ABCG1 induces apoptosis in several cell types (2). Our finding was supported by rounded cell mutant. For comparison, Annexin V binding was also examined in cells transfected with the wt ABCG1, morphology, phosphatidyl-serine externalization, and elevated caspase 3 activity in the ABCG1-expressing ABCG1-KM, and ABCG2, respectively. -

Role and Regulation of the P53-Homolog P73 in the Transformation of Normal Human Fibroblasts
Role and regulation of the p53-homolog p73 in the transformation of normal human fibroblasts Dissertation zur Erlangung des naturwissenschaftlichen Doktorgrades der Bayerischen Julius-Maximilians-Universität Würzburg vorgelegt von Lars Hofmann aus Aschaffenburg Würzburg 2007 Eingereicht am Mitglieder der Promotionskommission: Vorsitzender: Prof. Dr. Dr. Martin J. Müller Gutachter: Prof. Dr. Michael P. Schön Gutachter : Prof. Dr. Georg Krohne Tag des Promotionskolloquiums: Doktorurkunde ausgehändigt am Erklärung Hiermit erkläre ich, dass ich die vorliegende Arbeit selbständig angefertigt und keine anderen als die angegebenen Hilfsmittel und Quellen verwendet habe. Diese Arbeit wurde weder in gleicher noch in ähnlicher Form in einem anderen Prüfungsverfahren vorgelegt. Ich habe früher, außer den mit dem Zulassungsgesuch urkundlichen Graden, keine weiteren akademischen Grade erworben und zu erwerben gesucht. Würzburg, Lars Hofmann Content SUMMARY ................................................................................................................ IV ZUSAMMENFASSUNG ............................................................................................. V 1. INTRODUCTION ................................................................................................. 1 1.1. Molecular basics of cancer .......................................................................................... 1 1.2. Early research on tumorigenesis ................................................................................. 3 1.3. Developing -

ABCA5 (NM 018672) Human Tagged ORF Clone – RC213865 | Origene
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for RC213865 ABCA5 (NM_018672) Human Tagged ORF Clone Product data: Product Type: Expression Plasmids Product Name: ABCA5 (NM_018672) Human Tagged ORF Clone Tag: Myc-DDK Symbol: ABCA5 Synonyms: ABC13; EST90625; HTC3 Vector: pCMV6-Entry (PS100001) E. coli Selection: Kanamycin (25 ug/mL) Cell Selection: Neomycin ORF Nucleotide >RC213865 representing NM_018672 Sequence: Red=Cloning site Blue=ORF Green=Tags(s) TTTTGTAATACGACTCACTATAGGGCGGCCGGGAATTCGTCGACTGGATCCGGTACCGAGGAGATCTGCC GCCGCGATCGCC ATGTCCACTGCAATTAGGGAGGTAGGAGTTTGGAGACAGACCAGAACACTTCTACTGAAGAATTACTTAA TTAAATGCAGAACCAAAAAGAGTAGTGTTCAGGAAATTCTTTTTCCACTATTTTTTTTATTTTGGTTAAT ATTAATTAGCATGATGCATCCAAATAAGAAATATGAAGAAGTGCCTAATATAGAACTCAATCCTATGGAC AAGTTTACTCTTTCTAATCTAATTCTTGGATATACTCCAGTGACTAATATTACAAGCAGCATCATGCAGA AAGTGTCTACTGATCATCTACCTGATGTCATAATTACTGAAGAATATACAAATGAAAAAGAAATGTTAAC ATCCAGTCTCTCTAAGCCGAGCAACTTTGTAGGTGTGGTTTTCAAAGACTCCATGTCCTATGAACTTCGT TTTTTTCCTGATATGATTCCAGTATCTTCTATTTATATGGATTCAAGAGCTGGCTGTTCAAAATCATGTG AGGCTGCTCAGTACTGGTCCTCAGGTTTCACAGTTTTACAAGCATCCATAGATGCTGCCATTATACAGTT GAAGACCAATGTTTCTCTTTGGAAGGAGCTGGAGTCAACTAAAGCTGTTATTATGGGAGAAACTGCTGTT GTAGAAATAGATACCTTTCCCCGAGGAGTAATTTTAATATACCTAGTTATAGCATTTTCACCTTTTGGAT ACTTTTTGGCAATTCATATCGTAGCAGAAAAAGAAAAAAAAATAAAAGAATTTTTAAAGATAATGGGACT TCATGATACTGCCTTTTGGCTTTCCTGGGTTCTTCTATATACAAGTTTAATTTTTCTTATGTCCCTTCTT ATGGCAGTCATTGCGACAGCTTCTTTGTTATTTCCTCAAAGTAGCAGCATTGTGATATTTCTGCTTTTTT -

Whole-Exome Sequencing Identifies Novel Mutations in ABC Transporter
Liu et al. BMC Pregnancy and Childbirth (2021) 21:110 https://doi.org/10.1186/s12884-021-03595-x RESEARCH ARTICLE Open Access Whole-exome sequencing identifies novel mutations in ABC transporter genes associated with intrahepatic cholestasis of pregnancy disease: a case-control study Xianxian Liu1,2†, Hua Lai1,3†, Siming Xin1,3, Zengming Li1, Xiaoming Zeng1,3, Liju Nie1,3, Zhengyi Liang1,3, Meiling Wu1,3, Jiusheng Zheng1,3* and Yang Zou1,2* Abstract Background: Intrahepatic cholestasis of pregnancy (ICP) can cause premature delivery and stillbirth. Previous studies have reported that mutations in ABC transporter genes strongly influence the transport of bile salts. However, to date, their effects are still largely elusive. Methods: A whole-exome sequencing (WES) approach was used to detect novel variants. Rare novel exonic variants (minor allele frequencies: MAF < 1%) were analyzed. Three web-available tools, namely, SIFT, Mutation Taster and FATHMM, were used to predict protein damage. Protein structure modeling and comparisons between reference and modified protein structures were performed by SWISS-MODEL and Chimera 1.14rc, respectively. Results: We detected a total of 2953 mutations in 44 ABC family transporter genes. When the MAF of loci was controlled in all databases at less than 0.01, 320 mutations were reserved for further analysis. Among these mutations, 42 were novel. We classified these loci into four groups (the damaging, probably damaging, possibly damaging, and neutral groups) according to the prediction results, of which 7 novel possible pathogenic mutations were identified that were located in known functional genes, including ABCB4 (Trp708Ter, Gly527Glu and Lys386Glu), ABCB11 (Gln1194Ter, Gln605Pro and Leu589Met) and ABCC2 (Ser1342Tyr), in the damaging group. -

Cholesterol Loading Suppresses the Atheroinflammatory Gene
www.nature.com/scientificreports OPEN Cholesterol loading suppresses the atheroinfammatory gene polarization of human macrophages induced by colony stimulating factors Jani Lappalainen1, Nicolas Yeung1, Su D. Nguyen1, Matti Jauhiainen2, Petri T. Kovanen1* & Miriam Lee‑Rueckert1 In atherosclerotic lesions, blood‑derived monocytes diferentiate into distinct macrophage subpopulations, and further into cholesterol‑flled foam cells under a complex milieu of cytokines, which also contains macrophage‑colony stimulating factor (M‑CSF) and granulocyte–macrophage‑ colony stimulating factor (GM‑CSF). Here we generated human macrophages in the presence of either M‑CSF or GM‑CSF to obtain M‑MØ and GM‑MØ, respectively. The macrophages were converted into cholesterol‑loaded foam cells by incubating them with acetyl‑LDL, and their atheroinfammatory gene expression profles were then assessed. Compared with GM‑MØ, the M‑MØ expressed higher levels of CD36, SRA1, and ACAT1, and also exhibited a greater ability to take up acetyl‑LDL, esterify cholesterol, and become converted to foam cells. M‑MØ foam cells expressed higher levels of ABCA1 and ABCG1, and, correspondingly, exhibited higher rates of cholesterol efux to apoA‑I and HDL2. Cholesterol loading of M‑MØ strongly suppressed the high baseline expression of CCL2, whereas in GM‑MØ the low baseline expression CCL2 remained unchanged during cholesterol loading. The expression of TNFA, IL1B, and CXCL8 were reduced in LPS‑activated macrophage foam cells of either subtype. In summary, cholesterol loading converged the CSF‑dependent expression of key genes related to intracellular cholesterol balance and infammation. These fndings suggest that transformation of CSF‑polarized macrophages into foam cells may reduce their atheroinfammatory potential in atherogenesis.