Transporting Biotherapeutics Across the Blood-Brain Barrier
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Alzheimer's Disease Protective P522R Variant of PLCG2
bioRxiv preprint doi: https://doi.org/10.1101/2020.04.27.059600; this version posted April 28, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. The Alzheimer’s disease protective P522R variant of PLCG2, consistently enhances stimulus-dependent PLCγ2 activation, depleting substrate and altering cell function. Emily Maguire #1, Georgina E. Menzies#1, Thomas Phillips#1, Michael Sasner2, Harriet M. Williams2, Magdalena A. Czubala3, Neil Evans1, Emma L Cope4, Rebecca Sims5, Gareth R. Howell2, Emyr Lloyd-Evans4, Julie Williams†1,5, Nicholas D. Allen†4 and Philip R. Taylor†*1,3. 1 UK Dementia Research Institute at Cardiff, Hadyn Ellis Building, Maindy Road, Cardiff, CF24 4HQ, Wales, UK. 2 The Jackson Laboratory, Bar Harbor, Maine 04660, USA. 3 Systems Immunity University Research Institute, Tenovus Building, Heath Park, Cardiff CF 14 4XN, Wales, UK. 4 School of Biosciences, Cardiff University, Museum Avenue, Cardiff, CF10 3AX. 5 MRC Centre for Neuropsychiatric Genetics & Genomics, Hadyn Ellis Building, Maindy Road, Cardiff, CF24 4HQ, Wales, UK. #†These authors contributed equally *To whom correspondence should be addressed (lead contact): Prof Philip R. Taylor; Tel: +44(0)2920687328; Email: [email protected]. Abstract: Recent genome-wide association studies of Alzheimer’s disease (AD) have identified variants implicating immune pathways in disease development. A rare coding variant of PLCG2, which encodes PLCγ2, shows a significant protective effect for AD (rs72824905, P522R, P=5.38x10-10, Odds Ratio = 0.68). -

Plasma Lipidome Is Dysregulated in Alzheimer's Disease and Is
Liu et al. Translational Psychiatry (2021) 11:344 https://doi.org/10.1038/s41398-021-01362-2 Translational Psychiatry ARTICLE Open Access Plasma lipidome is dysregulated in Alzheimer’s disease and is associated with disease risk genes Yue Liu1,2, Anbupalam Thalamuthu1, Karen A. Mather1,3, John Crawford1, Marina Ulanova1, Matthew Wai Kin Wong1, Russell Pickford4, Perminder S. Sachdev 1,5 and Nady Braidy 1,6 Abstract Lipidomics research could provide insights of pathobiological mechanisms in Alzheimer’s disease. This study explores a battery of plasma lipids that can differentiate Alzheimer’s disease (AD) patients from healthy controls and determines whether lipid profiles correlate with genetic risk for AD. AD plasma samples were collected from the Sydney Memory and Ageing Study (MAS) Sydney, Australia (aged range 75–97 years; 51.2% male). Untargeted lipidomics analysis was performed by liquid chromatography coupled–mass spectrometry (LC–MS/MS). We found that several lipid species from nine lipid classes, particularly sphingomyelins (SMs), cholesterol esters (ChEs), phosphatidylcholines (PCs), phosphatidylethanolamines (PIs), phosphatidylinositols (PIs), and triglycerides (TGs) are dysregulated in AD patients and may help discriminate them from healthy controls. However, when the lipid species were grouped together into lipid subgroups, only the DG group was significantly higher in AD. ChEs, SMs, and TGs resulted in good classification accuracy using the Glmnet algorithm (elastic net penalization for the generalized linear model [glm]) with more than 80% AUC. In general, group lipids and the lipid subclasses LPC and PE had less classification accuracy compared to the other subclasses. We also found significant increases in SMs, PIs, and the LPE/PE ratio in human U251 astroglioma cell lines exposed to pathophysiological concentrations of oligomeric Aβ42. -

Antibody Response Cell Antigen Receptor Signaling And
Lysophosphatidic Acid Receptor 5 Inhibits B Cell Antigen Receptor Signaling and Antibody Response This information is current as Jiancheng Hu, Shannon K. Oda, Kristin Shotts, Erin E. of September 24, 2021. Donovan, Pamela Strauch, Lindsey M. Pujanauski, Francisco Victorino, Amin Al-Shami, Yuko Fujiwara, Gabor Tigyi, Tamas Oravecz, Roberta Pelanda and Raul M. Torres J Immunol 2014; 193:85-95; Prepublished online 2 June 2014; Downloaded from doi: 10.4049/jimmunol.1300429 http://www.jimmunol.org/content/193/1/85 Supplementary http://www.jimmunol.org/content/suppl/2014/05/31/jimmunol.130042 http://www.jimmunol.org/ Material 9.DCSupplemental References This article cites 63 articles, 17 of which you can access for free at: http://www.jimmunol.org/content/193/1/85.full#ref-list-1 Why The JI? Submit online. by guest on September 24, 2021 • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2014 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Lysophosphatidic Acid Receptor 5 Inhibits B Cell Antigen Receptor Signaling and Antibody Response Jiancheng Hu,*,1,2 Shannon K. -

Survival-Associated Metabolic Genes in Colon and Rectal Cancers
Survival-associated Metabolic Genes in Colon and Rectal Cancers Yanfen Cui ( [email protected] ) Tianjin Cancer Institute: Tianjin Tumor Hospital https://orcid.org/0000-0001-7760-7503 Baoai Han tianjin tumor hospital He Zhang tianjin tumor hospital Zhiyong Wang tianjin tumor hospital Hui Liu tianjin tumor hospital Fei Zhang tianjin tumor hospital Ruifang Niu tianjin tumor hospital Research Keywords: colon cancer, rectal cancer, prognosis, metabolism Posted Date: December 4th, 2020 DOI: https://doi.org/10.21203/rs.3.rs-117478/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/42 Abstract Background Uncontrolled proliferation is the most prominent biological feature of tumors. To rapidly proliferate and maximize the use of available nutrients, tumor cells regulate their metabolic behavior and the expression of metabolism-related genes (MRGs). In this study, we aimed to construct prognosis models for colon and rectal cancers, using MRGs to indicate the prognoses of patients. Methods We rst acquired the gene expression proles of colon and rectal cancers from the TCGA and GEO database, and utilized univariate Cox analysis, lasso regression, and multivariable cox analysis to identify MRGs for risk models. Then GSEA and KEGG functional enrichment analysis were utilized to identify the metabolism pathway of MRGs in the risk models and analyzed these genes comprehensively using GSCALite. Results Eight genes (CPT1C, PLCB2, PLA2G2D, GAMT, ENPP2, PIP4K2B, GPX3, and GSR) in the colon cancer risk model and six genes (TDO2, PKLR, GAMT, EARS2, ACO1, and WAS) in the rectal cancer risk model were identied successfully. Multivariate Cox analysis indicated that the models predicted overall survival accurately and independently for patients with colon or rectal cancer. -

Description Cy5 LG Cy3 MI Ratio(Cy3/Cy5) C9orf135
Supplementary Table S2. DNA microarray dataset of top 30 differentially expressed genes and housekeeping genes Up-regulated in SI cancer cells Symbol Description Cy5_LG Cy3_MI Ratio(Cy3/Cy5) C9orf135 Uncharacterized protein C9orf135 1.37 307.8 224.3 KIAA1245 Notch homolog 2 N-terminal like protein 1.82 406.8 223.6 APITD1 Centromere protein S (CENP-S) 2.56 560.3 218.7 PPIL6 Peptidyl-prolyl cis-trans isomerase-like 6 1.85 343.0 185.7 MYCBP2 Probable E3 ubiquitin-protein ligase MYCBP2 2.01 332.1 165.6 ANGPTL4 Angiopoietin-related protein 4 precursor 10.26 1500.1 146.3 C10orf79 Novel protein (Fragment) 2.86 415.5 145.5 NP_653323.1 KPL2 protein isoform 2 1.82 257.5 141.6 ZNF345 Zinc finger protein 345 1.58 215.7 136.8 SIX1 Homeobox protein SIX1 1.59 216.1 136.0 KLHL7 Kelch-like protein 7 1.91 254.4 133.3 TBX1 T-box transcription factor TBX1 1.57 209.0 133.1 PAG1 Phosphoprotein associated with glycosphingolipid-enriched microdomains 1 2.19 290.9 133.0 NOL12 Nucleolar protein 12 1.61 203.7 126.5 ZNF606 Zinc finger protein 606 2.12 267.7 126.0 NFKBIE NF-kappa-B inhibitor epsilon 5.28 658.3 124.7 ZMYND10 Zinc finger MYND domain-containing protein 10 6.85 835.5 121.9 hCG_23177 - 14.94 1758.9 117.7 KIF3A Kinesin-like protein KIF3A 1.94 224.9 116.1 Q9C0K3_HUMAN Actin-related protein Arp11 3.38 368.0 108.9 NP_056263.1 DPCD protein 2.61 270.5 103.7 GBP1 Interferon-induced guanylate-binding protein 1 1.46 149.1 102.3 NP_660151.2 NAD(P) dependent steroid dehydrogenase-like 1.35 137.4 101.5 NP_689672.2 CDNA FLJ90761 fis, clone THYRO1000099 3.68 372.5 -
Technical Note, Appendix: an Analysis of Blood Processing Methods to Prepare Samples for Genechip® Expression Profiling (Pdf, 1
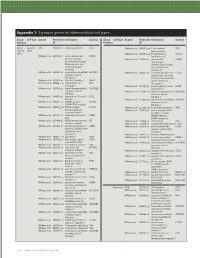
Appendix 1: Signature genes for different blood cell types. Blood Cell Type Source Probe Set Description Symbol Blood Cell Type Source Probe Set Description Symbol Fraction ID Fraction ID Mono- Lympho- GSK 203547_at CD4 antigen (p55) CD4 Whitney et al. 209813_x_at T cell receptor TRG nuclear cytes gamma locus cells Whitney et al. 209995_s_at T-cell leukemia/ TCL1A Whitney et al. 203104_at colony stimulating CSF1R lymphoma 1A factor 1 receptor, Whitney et al. 210164_at granzyme B GZMB formerly McDonough (granzyme 2, feline sarcoma viral cytotoxic T-lymphocyte- (v-fms) oncogene associated serine homolog esterase 1) Whitney et al. 203290_at major histocompatibility HLA-DQA1 Whitney et al. 210321_at similar to granzyme B CTLA1 complex, class II, (granzyme 2, cytotoxic DQ alpha 1 T-lymphocyte-associated Whitney et al. 203413_at NEL-like 2 (chicken) NELL2 serine esterase 1) Whitney et al. 203828_s_at natural killer cell NK4 (H. sapiens) transcript 4 Whitney et al. 212827_at immunoglobulin heavy IGHM Whitney et al. 203932_at major histocompatibility HLA-DMB constant mu complex, class II, Whitney et al. 212998_x_at major histocompatibility HLA-DQB1 DM beta complex, class II, Whitney et al. 204655_at chemokine (C-C motif) CCL5 DQ beta 1 ligand 5 Whitney et al. 212999_x_at major histocompatibility HLA-DQB Whitney et al. 204661_at CDW52 antigen CDW52 complex, class II, (CAMPATH-1 antigen) DQ beta 1 Whitney et al. 205049_s_at CD79A antigen CD79A Whitney et al. 213193_x_at T cell receptor beta locus TRB (immunoglobulin- Whitney et al. 213425_at Homo sapiens cDNA associated alpha) FLJ11441 fis, clone Whitney et al. 205291_at interleukin 2 receptor, IL2RB HEMBA1001323, beta mRNA sequence Whitney et al. -

Time Resolved Quantitative Phosphoproteomics Reveals Distinct Patterns of SHP2
bioRxiv preprint doi: https://doi.org/10.1101/598664; this version posted April 12, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Time resolved quantitative phosphoproteomics reveals distinct patterns of SHP2 dependence in EGFR signaling Vidyasiri Vemulapalli1,2, Lily Chylek3, Alison Erickson4, Jonathan LaRochelle1,2, Kartik Subramanian3, Morvarid Mohseni5, Matthew LaMarche5, Michael G. Acker5, Peter K. Sorger3, Steven P. Gygi4, and Stephen C. Blacklow1,2* 1Department of Cancer Biology, Dana-Farber Cancer Institute Boston, MA 02115, USA 2Department of Biological Chemistry & Molecular Pharmacology, Blavatnik Institute, Harvard Medical School, Boston, MA 02115, USA 3Laboratory of Systems Pharmacology, Harvard Medical School, Boston, MA 02115, USA 4Department of Cell Biology, Harvard Medical School, Boston, MA 02115, USA 5Novartis Institutes for Biomedical Research, Cambridge, MA, 02139, USA *To whom correspondence should be addressed: [email protected] bioRxiv preprint doi: https://doi.org/10.1101/598664; this version posted April 12, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Abstract SHP2 is a protein tyrosine phosphatase that normally potentiates intracellular signaling by growth factors, antigen receptors, and some cytokines; it is frequently mutated in childhood leukemias and other cancers. Here, we examine the role of SHP2 in the responses of breast cancer cells to EGF by monitoring phosphoproteome dynamics when SHP2 is allosterically inhibited by the small molecule SHP099. -

Mutational Landscape and Clinical Outcome of Patients with De Novo Acute Myeloid Leukemia and Rearrangements Involving 11Q23/KMT2A
Mutational landscape and clinical outcome of patients with de novo acute myeloid leukemia and rearrangements involving 11q23/KMT2A Marius Billa,1,2, Krzysztof Mrózeka,1,2, Jessica Kohlschmidta,b, Ann-Kathrin Eisfelda,c, Christopher J. Walkera, Deedra Nicoleta,b, Dimitrios Papaioannoua, James S. Blachlya,c, Shelley Orwicka,c, Andrew J. Carrolld, Jonathan E. Kolitze, Bayard L. Powellf, Richard M. Stoneg, Albert de la Chapelleh,i,2, John C. Byrda,c, and Clara D. Bloomfielda,c aThe Ohio State University Comprehensive Cancer Center, Columbus, OH 43210; bAlliance for Clinical Trials in Oncology Statistics and Data Center, The Ohio State University Comprehensive Cancer Center, Columbus, OH 43210; cDivision of Hematology, Department of Internal Medicine, The Ohio State University Comprehensive Cancer Center, Columbus, OH 43210; dDepartment of Genetics, University of Alabama at Birmingham, Birmingham, AL 35294; eNorthwell Health Cancer Institute, Zucker School of Medicine at Hofstra/Northwell, Lake Success, NY 11042; fDepartment of Internal Medicine, Section on Hematology & Oncology, Wake Forest Baptist Comprehensive Cancer Center, Winston-Salem, NC 27157; gDepartment of Medical Oncology, Dana-Farber/Partners Cancer Care, Boston, MA 02215; hHuman Cancer Genetics Program, Comprehensive Cancer Center, The Ohio State University, Columbus, OH 43210; and iDepartment of Cancer Biology and Genetics, Comprehensive Cancer Center, The Ohio State University, Columbus, OH 43210 Contributed by Albert de la Chapelle, August 28, 2020 (sent for review July 17, 2020; reviewed by Anne Hagemeijer and Stefan Klaus Bohlander) Balanced rearrangements involving the KMT2A gene, located at patterns that include high expression of HOXA genes and thereby 11q23, are among the most frequent chromosome aberrations in contribute to leukemogenesis (14–16). -

The Alzheimer's Disease-Associated
Takalo et al. Molecular Neurodegeneration (2020) 15:52 https://doi.org/10.1186/s13024-020-00402-7 SHORT REPORT Open Access The Alzheimer’s disease-associated protective Plcγ2-P522R variant promotes immune functions Mari Takalo1, Rebekka Wittrahm1, Benedikt Wefers2,3, Samira Parhizkar4, Kimmo Jokivarsi5, Teemu Kuulasmaa1, Petra Mäkinen1, Henna Martiskainen1, Wolfgang Wurst2,3,6, Xianyuan Xiang4, Mikael Marttinen1,7, Pekka Poutiainen8, Annakaisa Haapasalo5, Mikko Hiltunen1*† and Christian Haass2,4,6*† Abstract Background: Microglia-specific genetic variants are enriched in several neurodegenerative diseases, including Alzheimer’s disease (AD), implicating a central role for alterations of the innate immune system in the disease etiology. A rare coding variant in the PLCG2 gene (rs72824905, p.P522R) expressed in myeloid lineage cells was recently identified and shown to reduce the risk for AD. Methods: To assess the role of the protective variant in the context of immune cell functions, we generated a Plcγ2-P522R knock-in (KI) mouse model using CRISPR/Cas9 gene editing. Results: Functional analyses of macrophages derived from homozygous KI mice and wild type (WT) littermates revealed that the P522R variant potentiates the primary function of Plcγ2 as a Pip2-metabolizing enzyme. This was associated with improved survival and increased acute inflammatory response of the KI macrophages. Enhanced phagocytosis was observed in mouse BV2 microglia-like cells overexpressing human PLCγ2-P522R, but not in PLCγ2-WT expressing cells. Immunohistochemical analyses did not reveal changes in the number or morphology of microglia in the cortex of Plcγ2-P522R KI mice. However, the brain mRNA signature together with microglia-related PET imaging suggested enhanced microglial functions in Plcγ2-P522R KI mice. -

Genetic Testing Policy Number: PG0041 ADVANTAGE | ELITE | HMO Last Review: 04/11/2021
Genetic Testing Policy Number: PG0041 ADVANTAGE | ELITE | HMO Last Review: 04/11/2021 INDIVIDUAL MARKETPLACE | PROMEDICA MEDICARE PLAN | PPO GUIDELINES This policy does not certify benefits or authorization of benefits, which is designated by each individual policyholder terms, conditions, exclusions and limitations contract. It does not constitute a contract or guarantee regarding coverage or reimbursement/payment. Paramount applies coding edits to all medical claims through coding logic software to evaluate the accuracy and adherence to accepted national standards. This medical policy is solely for guiding medical necessity and explaining correct procedure reporting used to assist in making coverage decisions and administering benefits. SCOPE X Professional X Facility DESCRIPTION A genetic test is the analysis of human DNA, RNA, chromosomes, proteins, or certain metabolites in order to detect alterations related to a heritable or acquired disorder. This can be accomplished by directly examining the DNA or RNA that makes up a gene (direct testing), looking at markers co-inherited with a disease-causing gene (linkage testing), assaying certain metabolites (biochemical testing), or examining the chromosomes (cytogenetic testing). Clinical genetic tests are those in which specimens are examined and results reported to the provider or patient for the purpose of diagnosis, prevention or treatment in the care of individual patients. Genetic testing is performed for a variety of intended uses: Diagnostic testing (to diagnose disease) Predictive -

Molecular Mechanisms Regulating Phospholipase C-Γ2 Activity
Molecular mechanisms regulating phospholipase C‐2 activity Dissertation zur Erlangung des Doktorgrades Dr. rer. nat. der Fakultät für Naturwissenschaften der Universität Ulm vorgelegt von Anja Jasmin Bühler aus Ulm Universität Ulm Institut für Pharmakologie and Toxikologie Ulm 2013 Current Dean of the Faculty of Natural Sciences: Prof. Dr. Joachim Ankerhold First Supervisor: Prof. Dr. Peter Gierschik Second Supervisor: Prof. Dr. Ralf Marienfeld Day Doctorate Awarded: 24th March 2014 Table of contents Table of Contents List of Figures............................................................................................................................... v Abbreviations and Units............................................................................................................ vii 1 Introduction .................................................................................................................. 1 1.1 Signal transduction......................................................................................................... 1 1.2 Phospholipases C............................................................................................................. 2 1.3 Regulation of Phospholipases C..................................................................................... 5 1.4 Role of PLC2 in inflammatory and autoimmune diseases ....................................... 14 1.5 Anaplastic lymphoma kinase ....................................................................................... 17 1.6 Aim of the work............................................................................................................ -

Human Induced Pluripotent Stem Cell–Derived Podocytes Mature Into Vascularized Glomeruli Upon Experimental Transplantation
BASIC RESEARCH www.jasn.org Human Induced Pluripotent Stem Cell–Derived Podocytes Mature into Vascularized Glomeruli upon Experimental Transplantation † Sazia Sharmin,* Atsuhiro Taguchi,* Yusuke Kaku,* Yasuhiro Yoshimura,* Tomoko Ohmori,* ‡ † ‡ Tetsushi Sakuma, Masashi Mukoyama, Takashi Yamamoto, Hidetake Kurihara,§ and | Ryuichi Nishinakamura* *Department of Kidney Development, Institute of Molecular Embryology and Genetics, and †Department of Nephrology, Faculty of Life Sciences, Kumamoto University, Kumamoto, Japan; ‡Department of Mathematical and Life Sciences, Graduate School of Science, Hiroshima University, Hiroshima, Japan; §Division of Anatomy, Juntendo University School of Medicine, Tokyo, Japan; and |Japan Science and Technology Agency, CREST, Kumamoto, Japan ABSTRACT Glomerular podocytes express proteins, such as nephrin, that constitute the slit diaphragm, thereby contributing to the filtration process in the kidney. Glomerular development has been analyzed mainly in mice, whereas analysis of human kidney development has been minimal because of limited access to embryonic kidneys. We previously reported the induction of three-dimensional primordial glomeruli from human induced pluripotent stem (iPS) cells. Here, using transcription activator–like effector nuclease-mediated homologous recombination, we generated human iPS cell lines that express green fluorescent protein (GFP) in the NPHS1 locus, which encodes nephrin, and we show that GFP expression facilitated accurate visualization of nephrin-positive podocyte formation in