Time Resolved Quantitative Phosphoproteomics Reveals Distinct Patterns of SHP2
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Deregulated Gene Expression Pathways in Myelodysplastic Syndrome Hematopoietic Stem Cells
Leukemia (2010) 24, 756–764 & 2010 Macmillan Publishers Limited All rights reserved 0887-6924/10 $32.00 www.nature.com/leu ORIGINAL ARTICLE Deregulated gene expression pathways in myelodysplastic syndrome hematopoietic stem cells A Pellagatti1, M Cazzola2, A Giagounidis3, J Perry1, L Malcovati2, MG Della Porta2,MJa¨dersten4, S Killick5, A Verma6, CJ Norbury7, E Hellstro¨m-Lindberg4, JS Wainscoat1 and J Boultwood1 1LRF Molecular Haematology Unit, NDCLS, John Radcliffe Hospital, Oxford, UK; 2Department of Hematology Oncology, University of Pavia Medical School, Fondazione IRCCS Policlinico San Matteo, Pavia, Italy; 3Medizinische Klinik II, St Johannes Hospital, Duisburg, Germany; 4Division of Hematology, Department of Medicine, Karolinska Institutet, Stockholm, Sweden; 5Department of Haematology, Royal Bournemouth Hospital, Bournemouth, UK; 6Albert Einstein College of Medicine, Bronx, NY, USA and 7Sir William Dunn School of Pathology, University of Oxford, Oxford, UK To gain insight into the molecular pathogenesis of the the World Health Organization.6,7 Patients with refractory myelodysplastic syndromes (MDS), we performed global gene anemia (RA) with or without ringed sideroblasts, according to expression profiling and pathway analysis on the hemato- poietic stem cells (HSC) of 183 MDS patients as compared with the the French–American–British classification, were subdivided HSC of 17 healthy controls. The most significantly deregulated based on the presence or absence of multilineage dysplasia. In pathways in MDS include interferon signaling, thrombopoietin addition, patients with RA with excess blasts (RAEB) were signaling and the Wnt pathways. Among the most signifi- subdivided into two categories, RAEB1 and RAEB2, based on the cantly deregulated gene pathways in early MDS are immuno- percentage of bone marrow blasts. -

The Alzheimer's Disease Protective P522R Variant of PLCG2
bioRxiv preprint doi: https://doi.org/10.1101/2020.04.27.059600; this version posted April 28, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. The Alzheimer’s disease protective P522R variant of PLCG2, consistently enhances stimulus-dependent PLCγ2 activation, depleting substrate and altering cell function. Emily Maguire #1, Georgina E. Menzies#1, Thomas Phillips#1, Michael Sasner2, Harriet M. Williams2, Magdalena A. Czubala3, Neil Evans1, Emma L Cope4, Rebecca Sims5, Gareth R. Howell2, Emyr Lloyd-Evans4, Julie Williams†1,5, Nicholas D. Allen†4 and Philip R. Taylor†*1,3. 1 UK Dementia Research Institute at Cardiff, Hadyn Ellis Building, Maindy Road, Cardiff, CF24 4HQ, Wales, UK. 2 The Jackson Laboratory, Bar Harbor, Maine 04660, USA. 3 Systems Immunity University Research Institute, Tenovus Building, Heath Park, Cardiff CF 14 4XN, Wales, UK. 4 School of Biosciences, Cardiff University, Museum Avenue, Cardiff, CF10 3AX. 5 MRC Centre for Neuropsychiatric Genetics & Genomics, Hadyn Ellis Building, Maindy Road, Cardiff, CF24 4HQ, Wales, UK. #†These authors contributed equally *To whom correspondence should be addressed (lead contact): Prof Philip R. Taylor; Tel: +44(0)2920687328; Email: [email protected]. Abstract: Recent genome-wide association studies of Alzheimer’s disease (AD) have identified variants implicating immune pathways in disease development. A rare coding variant of PLCG2, which encodes PLCγ2, shows a significant protective effect for AD (rs72824905, P522R, P=5.38x10-10, Odds Ratio = 0.68). -

Plasma Lipidome Is Dysregulated in Alzheimer's Disease and Is
Liu et al. Translational Psychiatry (2021) 11:344 https://doi.org/10.1038/s41398-021-01362-2 Translational Psychiatry ARTICLE Open Access Plasma lipidome is dysregulated in Alzheimer’s disease and is associated with disease risk genes Yue Liu1,2, Anbupalam Thalamuthu1, Karen A. Mather1,3, John Crawford1, Marina Ulanova1, Matthew Wai Kin Wong1, Russell Pickford4, Perminder S. Sachdev 1,5 and Nady Braidy 1,6 Abstract Lipidomics research could provide insights of pathobiological mechanisms in Alzheimer’s disease. This study explores a battery of plasma lipids that can differentiate Alzheimer’s disease (AD) patients from healthy controls and determines whether lipid profiles correlate with genetic risk for AD. AD plasma samples were collected from the Sydney Memory and Ageing Study (MAS) Sydney, Australia (aged range 75–97 years; 51.2% male). Untargeted lipidomics analysis was performed by liquid chromatography coupled–mass spectrometry (LC–MS/MS). We found that several lipid species from nine lipid classes, particularly sphingomyelins (SMs), cholesterol esters (ChEs), phosphatidylcholines (PCs), phosphatidylethanolamines (PIs), phosphatidylinositols (PIs), and triglycerides (TGs) are dysregulated in AD patients and may help discriminate them from healthy controls. However, when the lipid species were grouped together into lipid subgroups, only the DG group was significantly higher in AD. ChEs, SMs, and TGs resulted in good classification accuracy using the Glmnet algorithm (elastic net penalization for the generalized linear model [glm]) with more than 80% AUC. In general, group lipids and the lipid subclasses LPC and PE had less classification accuracy compared to the other subclasses. We also found significant increases in SMs, PIs, and the LPE/PE ratio in human U251 astroglioma cell lines exposed to pathophysiological concentrations of oligomeric Aβ42. -

To Study Mutant P53 Gain of Function, Various Tumor-Derived P53 Mutants
Differential effects of mutant TAp63γ on transactivation of p53 and/or p63 responsive genes and their effects on global gene expression. A thesis submitted in partial fulfillment of the requirements for the degree of Master of Science By Shama K Khokhar M.Sc., Bilaspur University, 2004 B.Sc., Bhopal University, 2002 2007 1 COPYRIGHT SHAMA K KHOKHAR 2007 2 WRIGHT STATE UNIVERSITY SCHOOL OF GRADUATE STUDIES Date of Defense: 12-03-07 I HEREBY RECOMMEND THAT THE THESIS PREPARED UNDER MY SUPERVISION BY SHAMA KHAN KHOKHAR ENTITLED Differential effects of mutant TAp63γ on transactivation of p53 and/or p63 responsive genes and their effects on global gene expression BE ACCEPTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF Master of Science Madhavi P. Kadakia, Ph.D. Thesis Director Daniel Organisciak , Ph.D. Department Chair Committee on Final Examination Madhavi P. Kadakia, Ph.D. Steven J. Berberich, Ph.D. Michael Leffak, Ph.D. Joseph F. Thomas, Jr., Ph.D. Dean, School of Graduate Studies 3 Abstract Khokhar, Shama K. M.S., Department of Biochemistry and Molecular Biology, Wright State University, 2007 Differential effect of TAp63γ mutants on transactivation of p53 and/or p63 responsive genes and their effects on global gene expression. p63, a member of the p53 gene family, known to play a role in development, has more recently also been implicated in cancer progression. Mice lacking p63 exhibit severe developmental defects such as limb truncations, abnormal skin, and absence of hair follicles, teeth, and mammary glands. Germline missense mutations of p63 have been shown to be responsible for several human developmental syndromes including SHFM, EEC and ADULT syndromes and are associated with anomalies in the development of organs of epithelial origin. -

Antibody Response Cell Antigen Receptor Signaling And
Lysophosphatidic Acid Receptor 5 Inhibits B Cell Antigen Receptor Signaling and Antibody Response This information is current as Jiancheng Hu, Shannon K. Oda, Kristin Shotts, Erin E. of September 24, 2021. Donovan, Pamela Strauch, Lindsey M. Pujanauski, Francisco Victorino, Amin Al-Shami, Yuko Fujiwara, Gabor Tigyi, Tamas Oravecz, Roberta Pelanda and Raul M. Torres J Immunol 2014; 193:85-95; Prepublished online 2 June 2014; Downloaded from doi: 10.4049/jimmunol.1300429 http://www.jimmunol.org/content/193/1/85 Supplementary http://www.jimmunol.org/content/suppl/2014/05/31/jimmunol.130042 http://www.jimmunol.org/ Material 9.DCSupplemental References This article cites 63 articles, 17 of which you can access for free at: http://www.jimmunol.org/content/193/1/85.full#ref-list-1 Why The JI? Submit online. by guest on September 24, 2021 • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2014 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Lysophosphatidic Acid Receptor 5 Inhibits B Cell Antigen Receptor Signaling and Antibody Response Jiancheng Hu,*,1,2 Shannon K. -

Survival-Associated Metabolic Genes in Colon and Rectal Cancers
Survival-associated Metabolic Genes in Colon and Rectal Cancers Yanfen Cui ( [email protected] ) Tianjin Cancer Institute: Tianjin Tumor Hospital https://orcid.org/0000-0001-7760-7503 Baoai Han tianjin tumor hospital He Zhang tianjin tumor hospital Zhiyong Wang tianjin tumor hospital Hui Liu tianjin tumor hospital Fei Zhang tianjin tumor hospital Ruifang Niu tianjin tumor hospital Research Keywords: colon cancer, rectal cancer, prognosis, metabolism Posted Date: December 4th, 2020 DOI: https://doi.org/10.21203/rs.3.rs-117478/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/42 Abstract Background Uncontrolled proliferation is the most prominent biological feature of tumors. To rapidly proliferate and maximize the use of available nutrients, tumor cells regulate their metabolic behavior and the expression of metabolism-related genes (MRGs). In this study, we aimed to construct prognosis models for colon and rectal cancers, using MRGs to indicate the prognoses of patients. Methods We rst acquired the gene expression proles of colon and rectal cancers from the TCGA and GEO database, and utilized univariate Cox analysis, lasso regression, and multivariable cox analysis to identify MRGs for risk models. Then GSEA and KEGG functional enrichment analysis were utilized to identify the metabolism pathway of MRGs in the risk models and analyzed these genes comprehensively using GSCALite. Results Eight genes (CPT1C, PLCB2, PLA2G2D, GAMT, ENPP2, PIP4K2B, GPX3, and GSR) in the colon cancer risk model and six genes (TDO2, PKLR, GAMT, EARS2, ACO1, and WAS) in the rectal cancer risk model were identied successfully. Multivariate Cox analysis indicated that the models predicted overall survival accurately and independently for patients with colon or rectal cancer. -

Domain Requires the Gab2 Pleckstrin Homology Negative Regulation Of
Ligation of CD28 Stimulates the Formation of a Multimeric Signaling Complex Involving Grb-2-Associated Binder 2 (Gab2), Src Homology Phosphatase-2, and This information is current as Phosphatidylinositol 3-Kinase: Evidence That of October 1, 2021. Negative Regulation of CD28 Signaling Requires the Gab2 Pleckstrin Homology Domain Richard V. Parry, Gillian C. Whittaker, Martin Sims, Downloaded from Christine E. Edmead, Melanie J. Welham and Stephen G. Ward J Immunol 2006; 176:594-602; ; doi: 10.4049/jimmunol.176.1.594 http://www.jimmunol.org/content/176/1/594 http://www.jimmunol.org/ References This article cites 59 articles, 38 of which you can access for free at: http://www.jimmunol.org/content/176/1/594.full#ref-list-1 Why The JI? Submit online. by guest on October 1, 2021 • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2006 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Ligation of CD28 Stimulates the Formation of a Multimeric Signaling Complex Involving Grb-2-Associated Binder 2 (Gab2), Src Homology Phosphatase-2, and Phosphatidylinositol 3-Kinase: Evidence That Negative Regulation of CD28 Signaling Requires the Gab2 Pleckstrin Homology Domain1 Richard V. -

Description Cy5 LG Cy3 MI Ratio(Cy3/Cy5) C9orf135
Supplementary Table S2. DNA microarray dataset of top 30 differentially expressed genes and housekeeping genes Up-regulated in SI cancer cells Symbol Description Cy5_LG Cy3_MI Ratio(Cy3/Cy5) C9orf135 Uncharacterized protein C9orf135 1.37 307.8 224.3 KIAA1245 Notch homolog 2 N-terminal like protein 1.82 406.8 223.6 APITD1 Centromere protein S (CENP-S) 2.56 560.3 218.7 PPIL6 Peptidyl-prolyl cis-trans isomerase-like 6 1.85 343.0 185.7 MYCBP2 Probable E3 ubiquitin-protein ligase MYCBP2 2.01 332.1 165.6 ANGPTL4 Angiopoietin-related protein 4 precursor 10.26 1500.1 146.3 C10orf79 Novel protein (Fragment) 2.86 415.5 145.5 NP_653323.1 KPL2 protein isoform 2 1.82 257.5 141.6 ZNF345 Zinc finger protein 345 1.58 215.7 136.8 SIX1 Homeobox protein SIX1 1.59 216.1 136.0 KLHL7 Kelch-like protein 7 1.91 254.4 133.3 TBX1 T-box transcription factor TBX1 1.57 209.0 133.1 PAG1 Phosphoprotein associated with glycosphingolipid-enriched microdomains 1 2.19 290.9 133.0 NOL12 Nucleolar protein 12 1.61 203.7 126.5 ZNF606 Zinc finger protein 606 2.12 267.7 126.0 NFKBIE NF-kappa-B inhibitor epsilon 5.28 658.3 124.7 ZMYND10 Zinc finger MYND domain-containing protein 10 6.85 835.5 121.9 hCG_23177 - 14.94 1758.9 117.7 KIF3A Kinesin-like protein KIF3A 1.94 224.9 116.1 Q9C0K3_HUMAN Actin-related protein Arp11 3.38 368.0 108.9 NP_056263.1 DPCD protein 2.61 270.5 103.7 GBP1 Interferon-induced guanylate-binding protein 1 1.46 149.1 102.3 NP_660151.2 NAD(P) dependent steroid dehydrogenase-like 1.35 137.4 101.5 NP_689672.2 CDNA FLJ90761 fis, clone THYRO1000099 3.68 372.5 -
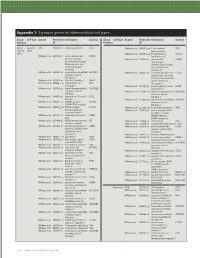
Technical Note, Appendix: an Analysis of Blood Processing Methods to Prepare Samples for Genechip® Expression Profiling (Pdf, 1
Appendix 1: Signature genes for different blood cell types. Blood Cell Type Source Probe Set Description Symbol Blood Cell Type Source Probe Set Description Symbol Fraction ID Fraction ID Mono- Lympho- GSK 203547_at CD4 antigen (p55) CD4 Whitney et al. 209813_x_at T cell receptor TRG nuclear cytes gamma locus cells Whitney et al. 209995_s_at T-cell leukemia/ TCL1A Whitney et al. 203104_at colony stimulating CSF1R lymphoma 1A factor 1 receptor, Whitney et al. 210164_at granzyme B GZMB formerly McDonough (granzyme 2, feline sarcoma viral cytotoxic T-lymphocyte- (v-fms) oncogene associated serine homolog esterase 1) Whitney et al. 203290_at major histocompatibility HLA-DQA1 Whitney et al. 210321_at similar to granzyme B CTLA1 complex, class II, (granzyme 2, cytotoxic DQ alpha 1 T-lymphocyte-associated Whitney et al. 203413_at NEL-like 2 (chicken) NELL2 serine esterase 1) Whitney et al. 203828_s_at natural killer cell NK4 (H. sapiens) transcript 4 Whitney et al. 212827_at immunoglobulin heavy IGHM Whitney et al. 203932_at major histocompatibility HLA-DMB constant mu complex, class II, Whitney et al. 212998_x_at major histocompatibility HLA-DQB1 DM beta complex, class II, Whitney et al. 204655_at chemokine (C-C motif) CCL5 DQ beta 1 ligand 5 Whitney et al. 212999_x_at major histocompatibility HLA-DQB Whitney et al. 204661_at CDW52 antigen CDW52 complex, class II, (CAMPATH-1 antigen) DQ beta 1 Whitney et al. 205049_s_at CD79A antigen CD79A Whitney et al. 213193_x_at T cell receptor beta locus TRB (immunoglobulin- Whitney et al. 213425_at Homo sapiens cDNA associated alpha) FLJ11441 fis, clone Whitney et al. 205291_at interleukin 2 receptor, IL2RB HEMBA1001323, beta mRNA sequence Whitney et al. -

The Regulatory Roles of Phosphatases in Cancer
Oncogene (2014) 33, 939–953 & 2014 Macmillan Publishers Limited All rights reserved 0950-9232/14 www.nature.com/onc REVIEW The regulatory roles of phosphatases in cancer J Stebbing1, LC Lit1, H Zhang, RS Darrington, O Melaiu, B Rudraraju and G Giamas The relevance of potentially reversible post-translational modifications required for controlling cellular processes in cancer is one of the most thriving arenas of cellular and molecular biology. Any alteration in the balanced equilibrium between kinases and phosphatases may result in development and progression of various diseases, including different types of cancer, though phosphatases are relatively under-studied. Loss of phosphatases such as PTEN (phosphatase and tensin homologue deleted on chromosome 10), a known tumour suppressor, across tumour types lends credence to the development of phosphatidylinositol 3--kinase inhibitors alongside the use of phosphatase expression as a biomarker, though phase 3 trial data are lacking. In this review, we give an updated report on phosphatase dysregulation linked to organ-specific malignancies. Oncogene (2014) 33, 939–953; doi:10.1038/onc.2013.80; published online 18 March 2013 Keywords: cancer; phosphatases; solid tumours GASTROINTESTINAL MALIGNANCIES abs in sera were significantly associated with poor survival in Oesophageal cancer advanced ESCC, suggesting that they may have a clinical utility in Loss of PTEN (phosphatase and tensin homologue deleted on ESCC screening and diagnosis.5 chromosome 10) expression in oesophageal cancer is frequent, Cao et al.6 investigated the role of protein tyrosine phosphatase, among other gene alterations characterizing this disease. Zhou non-receptor type 12 (PTPN12) in ESCC and showed that PTPN12 et al.1 found that overexpression of PTEN suppresses growth and protein expression is higher in normal para-cancerous tissues than induces apoptosis in oesophageal cancer cell lines, through in 20 ESCC tissues. -

PTPN11 Is the First Identified Proto-Oncogene That Encodes a Tyrosine Phosphatase
From www.bloodjournal.org by guest on July 4, 2016. For personal use only. Review article PTPN11 is the first identified proto-oncogene that encodes a tyrosine phosphatase Rebecca J. Chan1 and Gen-Sheng Feng2,3 1Department of Pediatrics, the Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis; 2Programs in Signal Transduction and Stem Cells & Regeneration, Burnham Institute for Medical Research, La Jolla, CA; 3Institute for Biomedical Research, Xiamen University, Xiamen, China Elucidation of the molecular mechanisms 2 Src-homology 2 (SH2) domains (Shp2). vation of the Ras-Erk pathway. This underlying carcinogenesis has benefited This tyrosine phosphatase was previ- progress represents another milestone in tremendously from the identification and ously shown to play an essential role in the leukemia/cancer research field and characterization of oncogenes and tumor normal hematopoiesis. More recently, so- provides a fresh view on the molecular suppressor genes. One new advance in matic missense PTPN11 gain-of-function mechanisms underlying cell transforma- this field is the identification of PTPN11 mutations have been detected in leuke- tion. (Blood. 2007;109:862-867) as the first proto-oncogene that encodes mias and rarely in solid tumors, and have a cytoplasmic tyrosine phosphatase with been found to induce aberrant hyperacti- © 2007 by The American Society of Hematology Introduction Leukemia and other types of cancer continue to be a leading cause tumor suppressor activity when overexpressed in vitro, and Ptprj of death in the United States, and biomedical scientists sorely note maps to the mouse colon cancer susceptibility locus,3 implicating that victories against cancer remain unacceptably rare. -

Digital Gene Expression Profiling of Primary Acute Lymphoblastic
Leukemia (2012) 26, 1218 --1227 & 2012 Macmillan Publishers Limited All rights reserved 0887-6924/12 www.nature.com/leu ORIGINAL ARTICLE Digital gene expression profiling of primary acute lymphoblastic leukemia cells J Nordlund1, A Kiialainen1,9, O Karlberg1, EC Berglund1,HGo¨ ransson-Kultima2, M Sønderkær3, KL Nielsen3, MG Gustafsson2, M Behrendtz4, E Forestier5, M Perkkio¨ 6,SSo¨ derha¨ll7,GLo¨ nnerholm8 and A-C Syva¨nen1 We determined the genome-wide digital gene expression (DGE) profiles of primary acute lymphoblastic leukemia (ALL) cells from 21 patients taking advantage of ‘second-generation’ sequencing technology. Patients included in this study represent four cytogenetically distinct subtypes of B-cell precursor (BCP) ALL and T-cell lineage ALL (T-ALL). The robustness of DGE combined with supervised classification by nearest shrunken centroids (NSC) was validated experimentally and by comparison with published expression data for large sets of ALL samples. Genes that were differentially expressed between BCP ALL subtypes were enriched to distinct signaling pathways with dic(9;20) enriched to TP53 signaling, t(9;22) to interferon signaling, as well as high hyperdiploidy and t(12;21) to apoptosis signaling. We also observed antisense tags expressed from the non-coding strand of B50% of annotated genes, many of which were expressed in a subtype-specific pattern. Antisense tags from 17 gene regions unambiguously discriminated between the BCP ALL and T-ALL subtypes, and antisense tags from 76 gene regions discriminated between the 4 BCP subtypes. We observed a significant overlap of gene regions with alternative polyadenylation and antisense transcription (Po1 Â 10À15). Our study using DGE profiling provided new insights into the RNA expression patterns in ALL cells.