PLCG2 Gene Phospholipase C Gamma 2
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Four Diseases, PLAID, APLAID, FCAS3 and CVID and One Gene
Four diseases, PLAID, APLAID, FCAS3 and CVID and one gene (PHOSPHOLIPASE C, GAMMA-2; PLCG2 ) : striking clinical phenotypic overlap and difference Necil Kutukculer1, Ezgi Yilmaz1, Afig Berdeli1, Raziye Burcu G¨uven Bilgin1, Ayca Aykut1, Asude Durmaz1, Ozgur Cogulu1, G¨uzideAksu1, and Neslihan Karaca1 1Ege University Faculty of Medicine May 15, 2020 Abstract We suggest PLAID,APLAID and FCAS3 have to be considered as same diseases,because of our long-term clinical experiences and genetic results in six patients.Small proportion of CVID patients are also PLAID/APLAID/FCAS3 patients and all these have disease-causing-mutations in PLCG2-genes,so it may be better to define all of them as “PLCG2 deficiency”. Key Clinical Message: Germline mutations in PLCG2 gene cause PLAID,APLAID,FCAS3, and CVID.Clinical experiences in patients with PLCG2 mutations led us to consider that PLAID, APLAID and FCAS3 are same diseases.It may be better to define all of them as “PLCG2 deficiency”. INTRODUCTION The PLCG2 gene which is located on the 16th chromosome (16q23.3) encodes phospholipase Cg2 (PLCG2), a transmembrane signaling enzyme that catalyzes the production of second messenger molecules utilizing calcium as a cofactor and propagates downstream signals in several hematopoietic cells (1). Recently, het- erozygous germline mutations in human PLCG2 were linked to some clinical phenotypes with some overlap- ping features|PLCg2-associated antibody deficiency and immune dysregulation syndrome (PLAID) (OMIM 614878) and autoinflammation, antibody deficiency, and immune dysregulation syndrome (APLAID) (OMIM 614878) (2-4) and familial cold autoinflammatory syndrome (FCAS3) (OMIM 614468) (5). All of them are autosomal dominant inherited diseases. -

The Alzheimer's Disease Protective P522R Variant of PLCG2
bioRxiv preprint doi: https://doi.org/10.1101/2020.04.27.059600; this version posted April 28, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. The Alzheimer’s disease protective P522R variant of PLCG2, consistently enhances stimulus-dependent PLCγ2 activation, depleting substrate and altering cell function. Emily Maguire #1, Georgina E. Menzies#1, Thomas Phillips#1, Michael Sasner2, Harriet M. Williams2, Magdalena A. Czubala3, Neil Evans1, Emma L Cope4, Rebecca Sims5, Gareth R. Howell2, Emyr Lloyd-Evans4, Julie Williams†1,5, Nicholas D. Allen†4 and Philip R. Taylor†*1,3. 1 UK Dementia Research Institute at Cardiff, Hadyn Ellis Building, Maindy Road, Cardiff, CF24 4HQ, Wales, UK. 2 The Jackson Laboratory, Bar Harbor, Maine 04660, USA. 3 Systems Immunity University Research Institute, Tenovus Building, Heath Park, Cardiff CF 14 4XN, Wales, UK. 4 School of Biosciences, Cardiff University, Museum Avenue, Cardiff, CF10 3AX. 5 MRC Centre for Neuropsychiatric Genetics & Genomics, Hadyn Ellis Building, Maindy Road, Cardiff, CF24 4HQ, Wales, UK. #†These authors contributed equally *To whom correspondence should be addressed (lead contact): Prof Philip R. Taylor; Tel: +44(0)2920687328; Email: [email protected]. Abstract: Recent genome-wide association studies of Alzheimer’s disease (AD) have identified variants implicating immune pathways in disease development. A rare coding variant of PLCG2, which encodes PLCγ2, shows a significant protective effect for AD (rs72824905, P522R, P=5.38x10-10, Odds Ratio = 0.68). -

Plasma Lipidome Is Dysregulated in Alzheimer's Disease and Is
Liu et al. Translational Psychiatry (2021) 11:344 https://doi.org/10.1038/s41398-021-01362-2 Translational Psychiatry ARTICLE Open Access Plasma lipidome is dysregulated in Alzheimer’s disease and is associated with disease risk genes Yue Liu1,2, Anbupalam Thalamuthu1, Karen A. Mather1,3, John Crawford1, Marina Ulanova1, Matthew Wai Kin Wong1, Russell Pickford4, Perminder S. Sachdev 1,5 and Nady Braidy 1,6 Abstract Lipidomics research could provide insights of pathobiological mechanisms in Alzheimer’s disease. This study explores a battery of plasma lipids that can differentiate Alzheimer’s disease (AD) patients from healthy controls and determines whether lipid profiles correlate with genetic risk for AD. AD plasma samples were collected from the Sydney Memory and Ageing Study (MAS) Sydney, Australia (aged range 75–97 years; 51.2% male). Untargeted lipidomics analysis was performed by liquid chromatography coupled–mass spectrometry (LC–MS/MS). We found that several lipid species from nine lipid classes, particularly sphingomyelins (SMs), cholesterol esters (ChEs), phosphatidylcholines (PCs), phosphatidylethanolamines (PIs), phosphatidylinositols (PIs), and triglycerides (TGs) are dysregulated in AD patients and may help discriminate them from healthy controls. However, when the lipid species were grouped together into lipid subgroups, only the DG group was significantly higher in AD. ChEs, SMs, and TGs resulted in good classification accuracy using the Glmnet algorithm (elastic net penalization for the generalized linear model [glm]) with more than 80% AUC. In general, group lipids and the lipid subclasses LPC and PE had less classification accuracy compared to the other subclasses. We also found significant increases in SMs, PIs, and the LPE/PE ratio in human U251 astroglioma cell lines exposed to pathophysiological concentrations of oligomeric Aβ42. -

Antibody Response Cell Antigen Receptor Signaling And
Lysophosphatidic Acid Receptor 5 Inhibits B Cell Antigen Receptor Signaling and Antibody Response This information is current as Jiancheng Hu, Shannon K. Oda, Kristin Shotts, Erin E. of September 24, 2021. Donovan, Pamela Strauch, Lindsey M. Pujanauski, Francisco Victorino, Amin Al-Shami, Yuko Fujiwara, Gabor Tigyi, Tamas Oravecz, Roberta Pelanda and Raul M. Torres J Immunol 2014; 193:85-95; Prepublished online 2 June 2014; Downloaded from doi: 10.4049/jimmunol.1300429 http://www.jimmunol.org/content/193/1/85 Supplementary http://www.jimmunol.org/content/suppl/2014/05/31/jimmunol.130042 http://www.jimmunol.org/ Material 9.DCSupplemental References This article cites 63 articles, 17 of which you can access for free at: http://www.jimmunol.org/content/193/1/85.full#ref-list-1 Why The JI? Submit online. by guest on September 24, 2021 • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2014 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Lysophosphatidic Acid Receptor 5 Inhibits B Cell Antigen Receptor Signaling and Antibody Response Jiancheng Hu,*,1,2 Shannon K. -

Survival-Associated Metabolic Genes in Colon and Rectal Cancers
Survival-associated Metabolic Genes in Colon and Rectal Cancers Yanfen Cui ( [email protected] ) Tianjin Cancer Institute: Tianjin Tumor Hospital https://orcid.org/0000-0001-7760-7503 Baoai Han tianjin tumor hospital He Zhang tianjin tumor hospital Zhiyong Wang tianjin tumor hospital Hui Liu tianjin tumor hospital Fei Zhang tianjin tumor hospital Ruifang Niu tianjin tumor hospital Research Keywords: colon cancer, rectal cancer, prognosis, metabolism Posted Date: December 4th, 2020 DOI: https://doi.org/10.21203/rs.3.rs-117478/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/42 Abstract Background Uncontrolled proliferation is the most prominent biological feature of tumors. To rapidly proliferate and maximize the use of available nutrients, tumor cells regulate their metabolic behavior and the expression of metabolism-related genes (MRGs). In this study, we aimed to construct prognosis models for colon and rectal cancers, using MRGs to indicate the prognoses of patients. Methods We rst acquired the gene expression proles of colon and rectal cancers from the TCGA and GEO database, and utilized univariate Cox analysis, lasso regression, and multivariable cox analysis to identify MRGs for risk models. Then GSEA and KEGG functional enrichment analysis were utilized to identify the metabolism pathway of MRGs in the risk models and analyzed these genes comprehensively using GSCALite. Results Eight genes (CPT1C, PLCB2, PLA2G2D, GAMT, ENPP2, PIP4K2B, GPX3, and GSR) in the colon cancer risk model and six genes (TDO2, PKLR, GAMT, EARS2, ACO1, and WAS) in the rectal cancer risk model were identied successfully. Multivariate Cox analysis indicated that the models predicted overall survival accurately and independently for patients with colon or rectal cancer. -

Abstract: Age-Related Immune Dysregulation and Increases In
Abstract: Age-related immune dysregulation and increases in inflammation, termed inflammaging, have been consistently implicated in most common age-related diseases, but the precise etiology of inter- individual differences in inflammaging are unknown. Changes in immunity and inflammation occur throughout the life course, but research on these processes among non-elderly populations has been limited. This is important because identifying sources of biological aging and inflammation before individuals reach older age may help identify points for intervention. The composition of the gut microbiota has been shown in animal models to have profound influence over, and interactions with, the immune system. Findings from germ-free mice suggest that commensal gut microbes are a key cause of inflammaging, but this hypothesis has not been well explored in humans. There are currently very few data examining how the microbiome relates to the fundamental aspects of aging biology, specifically inflammatory phenotypes and genomic markers of biological age. We propose to fill gaps in current microbiome research on aging, through the collection and analysis of oral and gut microbiome data in The National Longitudinal Study of Adolescent to Adult Health (Add Health), a nationally representative longitudinal cohort of adults with extensive social environment data and existing or ongoing analyses of genomic and phenotypic markers of inflammation and aging. The specific aims include the: 1) Collection of tongue and stool specimens with which to characterize the oral and gut microbiome in a nationally-representative sample (N ~10,155) of Add Health participants (mean age ~40); 2) Testing the association between the microbiome and biomarkers of aging and inflammation, and the creation of a novel “microbiome age clock”; 3) Examination of the relationships between life course exposures and microbiome species related to biomarkers of aging and inflammation as an adult; 4) Documentation and dissemination of data generated from this project. -

Description Cy5 LG Cy3 MI Ratio(Cy3/Cy5) C9orf135
Supplementary Table S2. DNA microarray dataset of top 30 differentially expressed genes and housekeeping genes Up-regulated in SI cancer cells Symbol Description Cy5_LG Cy3_MI Ratio(Cy3/Cy5) C9orf135 Uncharacterized protein C9orf135 1.37 307.8 224.3 KIAA1245 Notch homolog 2 N-terminal like protein 1.82 406.8 223.6 APITD1 Centromere protein S (CENP-S) 2.56 560.3 218.7 PPIL6 Peptidyl-prolyl cis-trans isomerase-like 6 1.85 343.0 185.7 MYCBP2 Probable E3 ubiquitin-protein ligase MYCBP2 2.01 332.1 165.6 ANGPTL4 Angiopoietin-related protein 4 precursor 10.26 1500.1 146.3 C10orf79 Novel protein (Fragment) 2.86 415.5 145.5 NP_653323.1 KPL2 protein isoform 2 1.82 257.5 141.6 ZNF345 Zinc finger protein 345 1.58 215.7 136.8 SIX1 Homeobox protein SIX1 1.59 216.1 136.0 KLHL7 Kelch-like protein 7 1.91 254.4 133.3 TBX1 T-box transcription factor TBX1 1.57 209.0 133.1 PAG1 Phosphoprotein associated with glycosphingolipid-enriched microdomains 1 2.19 290.9 133.0 NOL12 Nucleolar protein 12 1.61 203.7 126.5 ZNF606 Zinc finger protein 606 2.12 267.7 126.0 NFKBIE NF-kappa-B inhibitor epsilon 5.28 658.3 124.7 ZMYND10 Zinc finger MYND domain-containing protein 10 6.85 835.5 121.9 hCG_23177 - 14.94 1758.9 117.7 KIF3A Kinesin-like protein KIF3A 1.94 224.9 116.1 Q9C0K3_HUMAN Actin-related protein Arp11 3.38 368.0 108.9 NP_056263.1 DPCD protein 2.61 270.5 103.7 GBP1 Interferon-induced guanylate-binding protein 1 1.46 149.1 102.3 NP_660151.2 NAD(P) dependent steroid dehydrogenase-like 1.35 137.4 101.5 NP_689672.2 CDNA FLJ90761 fis, clone THYRO1000099 3.68 372.5 -
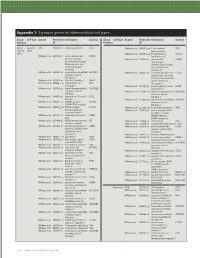
Technical Note, Appendix: an Analysis of Blood Processing Methods to Prepare Samples for Genechip® Expression Profiling (Pdf, 1
Appendix 1: Signature genes for different blood cell types. Blood Cell Type Source Probe Set Description Symbol Blood Cell Type Source Probe Set Description Symbol Fraction ID Fraction ID Mono- Lympho- GSK 203547_at CD4 antigen (p55) CD4 Whitney et al. 209813_x_at T cell receptor TRG nuclear cytes gamma locus cells Whitney et al. 209995_s_at T-cell leukemia/ TCL1A Whitney et al. 203104_at colony stimulating CSF1R lymphoma 1A factor 1 receptor, Whitney et al. 210164_at granzyme B GZMB formerly McDonough (granzyme 2, feline sarcoma viral cytotoxic T-lymphocyte- (v-fms) oncogene associated serine homolog esterase 1) Whitney et al. 203290_at major histocompatibility HLA-DQA1 Whitney et al. 210321_at similar to granzyme B CTLA1 complex, class II, (granzyme 2, cytotoxic DQ alpha 1 T-lymphocyte-associated Whitney et al. 203413_at NEL-like 2 (chicken) NELL2 serine esterase 1) Whitney et al. 203828_s_at natural killer cell NK4 (H. sapiens) transcript 4 Whitney et al. 212827_at immunoglobulin heavy IGHM Whitney et al. 203932_at major histocompatibility HLA-DMB constant mu complex, class II, Whitney et al. 212998_x_at major histocompatibility HLA-DQB1 DM beta complex, class II, Whitney et al. 204655_at chemokine (C-C motif) CCL5 DQ beta 1 ligand 5 Whitney et al. 212999_x_at major histocompatibility HLA-DQB Whitney et al. 204661_at CDW52 antigen CDW52 complex, class II, (CAMPATH-1 antigen) DQ beta 1 Whitney et al. 205049_s_at CD79A antigen CD79A Whitney et al. 213193_x_at T cell receptor beta locus TRB (immunoglobulin- Whitney et al. 213425_at Homo sapiens cDNA associated alpha) FLJ11441 fis, clone Whitney et al. 205291_at interleukin 2 receptor, IL2RB HEMBA1001323, beta mRNA sequence Whitney et al. -

Time Resolved Quantitative Phosphoproteomics Reveals Distinct Patterns of SHP2
bioRxiv preprint doi: https://doi.org/10.1101/598664; this version posted April 12, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Time resolved quantitative phosphoproteomics reveals distinct patterns of SHP2 dependence in EGFR signaling Vidyasiri Vemulapalli1,2, Lily Chylek3, Alison Erickson4, Jonathan LaRochelle1,2, Kartik Subramanian3, Morvarid Mohseni5, Matthew LaMarche5, Michael G. Acker5, Peter K. Sorger3, Steven P. Gygi4, and Stephen C. Blacklow1,2* 1Department of Cancer Biology, Dana-Farber Cancer Institute Boston, MA 02115, USA 2Department of Biological Chemistry & Molecular Pharmacology, Blavatnik Institute, Harvard Medical School, Boston, MA 02115, USA 3Laboratory of Systems Pharmacology, Harvard Medical School, Boston, MA 02115, USA 4Department of Cell Biology, Harvard Medical School, Boston, MA 02115, USA 5Novartis Institutes for Biomedical Research, Cambridge, MA, 02139, USA *To whom correspondence should be addressed: [email protected] bioRxiv preprint doi: https://doi.org/10.1101/598664; this version posted April 12, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Abstract SHP2 is a protein tyrosine phosphatase that normally potentiates intracellular signaling by growth factors, antigen receptors, and some cytokines; it is frequently mutated in childhood leukemias and other cancers. Here, we examine the role of SHP2 in the responses of breast cancer cells to EGF by monitoring phosphoproteome dynamics when SHP2 is allosterically inhibited by the small molecule SHP099. -

Harnessing the Immune System to Overcome Cytokine Storm And
Khadke et al. Virol J (2020) 17:154 https://doi.org/10.1186/s12985-020-01415-w REVIEW Open Access Harnessing the immune system to overcome cytokine storm and reduce viral load in COVID-19: a review of the phases of illness and therapeutic agents Sumanth Khadke1, Nayla Ahmed2, Nausheen Ahmed3, Ryan Ratts2,4, Shine Raju5, Molly Gallogly6, Marcos de Lima6 and Muhammad Rizwan Sohail7* Abstract Background: Coronavirus disease 2019 (COVID-19) is caused by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2, previously named 2019-nCov), a novel coronavirus that emerged in China in December 2019 and was declared a global pandemic by World Health Organization by March 11th, 2020. Severe manifestations of COVID-19 are caused by a combination of direct tissue injury by viral replication and associated cytokine storm resulting in progressive organ damage. Discussion: We reviewed published literature between January 1st, 2000 and June 30th, 2020, excluding articles focusing on pediatric or obstetric population, with a focus on virus-host interactions and immunological mechanisms responsible for virus associated cytokine release syndrome (CRS). COVID-19 illness encompasses three main phases. In phase 1, SARS-CoV-2 binds with angiotensin converting enzyme (ACE)2 receptor on alveolar macrophages and epithelial cells, triggering toll like receptor (TLR) mediated nuclear factor kappa-light-chain-enhancer of activated B cells (NF-ƙB) signaling. It efectively blunts an early (IFN) response allowing unchecked viral replication. Phase 2 is characterized by hypoxia and innate immunity mediated pneumocyte damage as well as capillary leak. Some patients further progress to phase 3 characterized by cytokine storm with worsening respiratory symptoms, persistent fever, and hemodynamic instability. -

Immune Dysfunction and Neuroinflammation in Autism
Review Acta Neurobiol Exp 2016, 76: 257–268 Immune dysfunction and neuroinflammation in autism spectrum disorder Geir Bjørklund1*, Khaled Saad2, Salvatore Chirumbolo3, Janet K. Kern4, David A. Geier4, Mark R. Geier4, and Mauricio A. Urbina5 1 Council for Nutritional and Environmental Medicine, Mo i Rana, Norway, 2 Department of Pediatrics, Faculty of Medicine, Assiut University, Assiut, Egypt, 3 Department of Neurological and Movement Science, University of Verona, Verona, Italy, 4 Institute of Chronic Illnesses, Inc., Silver Spring, MD, USA, 5 Departamento de Zoología, Facultad de Ciencias Naturales y Oceanográficas, Universidad de Concepción, Concepción, Chile, * Email: [email protected] Autism spectrum disorder (ASD) is a complex heterogeneous neurodevelopmental disorder with a complex pathogenesis. Many studies over the last four decades have recognized altered immune responses among individuals diagnosed with ASD. The purpose of this critical and comprehensive review is to examine the hypothesis that immune dysfunction is frequently present in those with ASD. It was found that often individuals diagnosed with ASD have alterations in immune cells such as T cells, B cells, monocytes, natural killer cells, and dendritic cells. Also, many individuals diagnosed with ASD have alterations in immunoglobulins and increased autoantibodies. Finally, a significant portion of individuals diagnosed with ASD have elevated peripheral cytokines and chemokines and associated neuroinflammation. In conclusion, immune dysregulation and inflammation are important components in the diagnosis and treatment of ASD. Key words: autism, cytokines, innate immunity, neuroinflammation INTRODUCTION a fundamental role in ASD development, despite some concern about whether it causes ASD onset or regulates Autism spectrum disorder (ASD) is considered ASD pathogenesis and symptomatology. -

Mutational Landscape and Clinical Outcome of Patients with De Novo Acute Myeloid Leukemia and Rearrangements Involving 11Q23/KMT2A
Mutational landscape and clinical outcome of patients with de novo acute myeloid leukemia and rearrangements involving 11q23/KMT2A Marius Billa,1,2, Krzysztof Mrózeka,1,2, Jessica Kohlschmidta,b, Ann-Kathrin Eisfelda,c, Christopher J. Walkera, Deedra Nicoleta,b, Dimitrios Papaioannoua, James S. Blachlya,c, Shelley Orwicka,c, Andrew J. Carrolld, Jonathan E. Kolitze, Bayard L. Powellf, Richard M. Stoneg, Albert de la Chapelleh,i,2, John C. Byrda,c, and Clara D. Bloomfielda,c aThe Ohio State University Comprehensive Cancer Center, Columbus, OH 43210; bAlliance for Clinical Trials in Oncology Statistics and Data Center, The Ohio State University Comprehensive Cancer Center, Columbus, OH 43210; cDivision of Hematology, Department of Internal Medicine, The Ohio State University Comprehensive Cancer Center, Columbus, OH 43210; dDepartment of Genetics, University of Alabama at Birmingham, Birmingham, AL 35294; eNorthwell Health Cancer Institute, Zucker School of Medicine at Hofstra/Northwell, Lake Success, NY 11042; fDepartment of Internal Medicine, Section on Hematology & Oncology, Wake Forest Baptist Comprehensive Cancer Center, Winston-Salem, NC 27157; gDepartment of Medical Oncology, Dana-Farber/Partners Cancer Care, Boston, MA 02215; hHuman Cancer Genetics Program, Comprehensive Cancer Center, The Ohio State University, Columbus, OH 43210; and iDepartment of Cancer Biology and Genetics, Comprehensive Cancer Center, The Ohio State University, Columbus, OH 43210 Contributed by Albert de la Chapelle, August 28, 2020 (sent for review July 17, 2020; reviewed by Anne Hagemeijer and Stefan Klaus Bohlander) Balanced rearrangements involving the KMT2A gene, located at patterns that include high expression of HOXA genes and thereby 11q23, are among the most frequent chromosome aberrations in contribute to leukemogenesis (14–16).