Transcriptomic and Computational Analysis Identified LPA Metabolism
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Alzheimer's Disease Protective P522R Variant of PLCG2
bioRxiv preprint doi: https://doi.org/10.1101/2020.04.27.059600; this version posted April 28, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. The Alzheimer’s disease protective P522R variant of PLCG2, consistently enhances stimulus-dependent PLCγ2 activation, depleting substrate and altering cell function. Emily Maguire #1, Georgina E. Menzies#1, Thomas Phillips#1, Michael Sasner2, Harriet M. Williams2, Magdalena A. Czubala3, Neil Evans1, Emma L Cope4, Rebecca Sims5, Gareth R. Howell2, Emyr Lloyd-Evans4, Julie Williams†1,5, Nicholas D. Allen†4 and Philip R. Taylor†*1,3. 1 UK Dementia Research Institute at Cardiff, Hadyn Ellis Building, Maindy Road, Cardiff, CF24 4HQ, Wales, UK. 2 The Jackson Laboratory, Bar Harbor, Maine 04660, USA. 3 Systems Immunity University Research Institute, Tenovus Building, Heath Park, Cardiff CF 14 4XN, Wales, UK. 4 School of Biosciences, Cardiff University, Museum Avenue, Cardiff, CF10 3AX. 5 MRC Centre for Neuropsychiatric Genetics & Genomics, Hadyn Ellis Building, Maindy Road, Cardiff, CF24 4HQ, Wales, UK. #†These authors contributed equally *To whom correspondence should be addressed (lead contact): Prof Philip R. Taylor; Tel: +44(0)2920687328; Email: [email protected]. Abstract: Recent genome-wide association studies of Alzheimer’s disease (AD) have identified variants implicating immune pathways in disease development. A rare coding variant of PLCG2, which encodes PLCγ2, shows a significant protective effect for AD (rs72824905, P522R, P=5.38x10-10, Odds Ratio = 0.68). -

Lysophosphatidic Acid and Its Receptors: Pharmacology and Therapeutic Potential in Atherosclerosis and Vascular Disease
JPT-107404; No of Pages 13 Pharmacology & Therapeutics xxx (2019) xxx Contents lists available at ScienceDirect Pharmacology & Therapeutics journal homepage: www.elsevier.com/locate/pharmthera Lysophosphatidic acid and its receptors: pharmacology and therapeutic potential in atherosclerosis and vascular disease Ying Zhou a, Peter J. Little a,b, Hang T. Ta a,c, Suowen Xu d, Danielle Kamato a,b,⁎ a School of Pharmacy, University of Queensland, Pharmacy Australia Centre of Excellence, Woolloongabba, QLD 4102, Australia b Department of Pharmacy, Xinhua College of Sun Yat-sen University, Tianhe District, Guangzhou 510520, China c Australian Institute for Bioengineering and Nanotechnology, The University of Queensland, Brisbane, St Lucia, QLD 4072, Australia d Aab Cardiovascular Research Institute, Department of Medicine, University of Rochester School of Medicine and Dentistry, Rochester, NY 14642, USA article info abstract Available online xxxx Lysophosphatidic acid (LPA) is a collective name for a set of bioactive lipid species. Via six widely distributed G protein-coupled receptors (GPCRs), LPA elicits a plethora of biological responses, contributing to inflammation, Keywords: thrombosis and atherosclerosis. There have recently been considerable advances in GPCR signaling especially Lysophosphatidic acid recognition of the extended role for GPCR transactivation of tyrosine and serine/threonine kinase growth factor G-protein coupled receptors receptors. This review covers LPA signaling pathways in the light of new information. The use of transgenic and Atherosclerosis gene knockout animals, gene manipulated cells, pharmacological LPA receptor agonists and antagonists have Gproteins fi β-arrestins provided many insights into the biological signi cance of LPA and individual LPA receptors in the progression Transactivation of atherosclerosis and vascular diseases. -

The Autotaxin–Lysophosphatidic Acid Axis Modulates Histone Acetylation and Gene Expression During Oligodendrocyte Differentiation
The Journal of Neuroscience, August 12, 2015 • 35(32):11399–11414 • 11399 Cellular/Molecular The Autotaxin–Lysophosphatidic Acid Axis Modulates Histone Acetylation and Gene Expression during Oligodendrocyte Differentiation Natalie A. Wheeler,1 James A. Lister,2 and Babette Fuss1 Departments of 1Anatomy and Neurobiology and 2Human and Molecular Genetics, Virginia Commonwealth University School of Medicine, Richmond, Virginia 23298 During development, oligodendrocytes (OLGs), the myelinating cells of the CNS, undergo a stepwise progression during which OLG progenitors, specified from neural stem/progenitor cells, differentiate into fully mature myelinating OLGs. This progression along the OLG lineage is characterized by well synchronized changes in morphology and gene expression patterns. The latter have been found to be particularly critical during the early stages of the lineage, and they have been well described to be regulated by epigenetic mechanisms, especially by the activity of the histone deacetylases HDAC1 and HDAC2. The data presented here identify the extracellular factor autotaxin (ATX) as a novel upstream signal modulating HDAC1/2 activity and gene expression in cells of the OLG lineage. Using the zebrafish as an in vivo model system as well as rodent primary OLG cultures, this functional property of ATX was found to be mediated by its lysophospholipase D (lysoPLD) activity, which has been well characterized to generate the lipid signaling molecule lysophosphatidic acid (LPA). More specifically, the lysoPLD activity of ATX was found to modulate HDAC1/2 regulated gene expression during a time window coinciding with the transition from OLG progenitor to early differentiating OLG. In contrast, HDAC1/2 regulated gene expression during the transition from neural stem/progenitor to OLG progenitor appeared unaffected by ATX and its lysoPLD activity. -

Plasma Lipidome Is Dysregulated in Alzheimer's Disease and Is
Liu et al. Translational Psychiatry (2021) 11:344 https://doi.org/10.1038/s41398-021-01362-2 Translational Psychiatry ARTICLE Open Access Plasma lipidome is dysregulated in Alzheimer’s disease and is associated with disease risk genes Yue Liu1,2, Anbupalam Thalamuthu1, Karen A. Mather1,3, John Crawford1, Marina Ulanova1, Matthew Wai Kin Wong1, Russell Pickford4, Perminder S. Sachdev 1,5 and Nady Braidy 1,6 Abstract Lipidomics research could provide insights of pathobiological mechanisms in Alzheimer’s disease. This study explores a battery of plasma lipids that can differentiate Alzheimer’s disease (AD) patients from healthy controls and determines whether lipid profiles correlate with genetic risk for AD. AD plasma samples were collected from the Sydney Memory and Ageing Study (MAS) Sydney, Australia (aged range 75–97 years; 51.2% male). Untargeted lipidomics analysis was performed by liquid chromatography coupled–mass spectrometry (LC–MS/MS). We found that several lipid species from nine lipid classes, particularly sphingomyelins (SMs), cholesterol esters (ChEs), phosphatidylcholines (PCs), phosphatidylethanolamines (PIs), phosphatidylinositols (PIs), and triglycerides (TGs) are dysregulated in AD patients and may help discriminate them from healthy controls. However, when the lipid species were grouped together into lipid subgroups, only the DG group was significantly higher in AD. ChEs, SMs, and TGs resulted in good classification accuracy using the Glmnet algorithm (elastic net penalization for the generalized linear model [glm]) with more than 80% AUC. In general, group lipids and the lipid subclasses LPC and PE had less classification accuracy compared to the other subclasses. We also found significant increases in SMs, PIs, and the LPE/PE ratio in human U251 astroglioma cell lines exposed to pathophysiological concentrations of oligomeric Aβ42. -

Antibody Response Cell Antigen Receptor Signaling And
Lysophosphatidic Acid Receptor 5 Inhibits B Cell Antigen Receptor Signaling and Antibody Response This information is current as Jiancheng Hu, Shannon K. Oda, Kristin Shotts, Erin E. of September 24, 2021. Donovan, Pamela Strauch, Lindsey M. Pujanauski, Francisco Victorino, Amin Al-Shami, Yuko Fujiwara, Gabor Tigyi, Tamas Oravecz, Roberta Pelanda and Raul M. Torres J Immunol 2014; 193:85-95; Prepublished online 2 June 2014; Downloaded from doi: 10.4049/jimmunol.1300429 http://www.jimmunol.org/content/193/1/85 Supplementary http://www.jimmunol.org/content/suppl/2014/05/31/jimmunol.130042 http://www.jimmunol.org/ Material 9.DCSupplemental References This article cites 63 articles, 17 of which you can access for free at: http://www.jimmunol.org/content/193/1/85.full#ref-list-1 Why The JI? Submit online. by guest on September 24, 2021 • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2014 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Lysophosphatidic Acid Receptor 5 Inhibits B Cell Antigen Receptor Signaling and Antibody Response Jiancheng Hu,*,1,2 Shannon K. -

Survival-Associated Metabolic Genes in Colon and Rectal Cancers
Survival-associated Metabolic Genes in Colon and Rectal Cancers Yanfen Cui ( [email protected] ) Tianjin Cancer Institute: Tianjin Tumor Hospital https://orcid.org/0000-0001-7760-7503 Baoai Han tianjin tumor hospital He Zhang tianjin tumor hospital Zhiyong Wang tianjin tumor hospital Hui Liu tianjin tumor hospital Fei Zhang tianjin tumor hospital Ruifang Niu tianjin tumor hospital Research Keywords: colon cancer, rectal cancer, prognosis, metabolism Posted Date: December 4th, 2020 DOI: https://doi.org/10.21203/rs.3.rs-117478/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/42 Abstract Background Uncontrolled proliferation is the most prominent biological feature of tumors. To rapidly proliferate and maximize the use of available nutrients, tumor cells regulate their metabolic behavior and the expression of metabolism-related genes (MRGs). In this study, we aimed to construct prognosis models for colon and rectal cancers, using MRGs to indicate the prognoses of patients. Methods We rst acquired the gene expression proles of colon and rectal cancers from the TCGA and GEO database, and utilized univariate Cox analysis, lasso regression, and multivariable cox analysis to identify MRGs for risk models. Then GSEA and KEGG functional enrichment analysis were utilized to identify the metabolism pathway of MRGs in the risk models and analyzed these genes comprehensively using GSCALite. Results Eight genes (CPT1C, PLCB2, PLA2G2D, GAMT, ENPP2, PIP4K2B, GPX3, and GSR) in the colon cancer risk model and six genes (TDO2, PKLR, GAMT, EARS2, ACO1, and WAS) in the rectal cancer risk model were identied successfully. Multivariate Cox analysis indicated that the models predicted overall survival accurately and independently for patients with colon or rectal cancer. -

Description Cy5 LG Cy3 MI Ratio(Cy3/Cy5) C9orf135
Supplementary Table S2. DNA microarray dataset of top 30 differentially expressed genes and housekeeping genes Up-regulated in SI cancer cells Symbol Description Cy5_LG Cy3_MI Ratio(Cy3/Cy5) C9orf135 Uncharacterized protein C9orf135 1.37 307.8 224.3 KIAA1245 Notch homolog 2 N-terminal like protein 1.82 406.8 223.6 APITD1 Centromere protein S (CENP-S) 2.56 560.3 218.7 PPIL6 Peptidyl-prolyl cis-trans isomerase-like 6 1.85 343.0 185.7 MYCBP2 Probable E3 ubiquitin-protein ligase MYCBP2 2.01 332.1 165.6 ANGPTL4 Angiopoietin-related protein 4 precursor 10.26 1500.1 146.3 C10orf79 Novel protein (Fragment) 2.86 415.5 145.5 NP_653323.1 KPL2 protein isoform 2 1.82 257.5 141.6 ZNF345 Zinc finger protein 345 1.58 215.7 136.8 SIX1 Homeobox protein SIX1 1.59 216.1 136.0 KLHL7 Kelch-like protein 7 1.91 254.4 133.3 TBX1 T-box transcription factor TBX1 1.57 209.0 133.1 PAG1 Phosphoprotein associated with glycosphingolipid-enriched microdomains 1 2.19 290.9 133.0 NOL12 Nucleolar protein 12 1.61 203.7 126.5 ZNF606 Zinc finger protein 606 2.12 267.7 126.0 NFKBIE NF-kappa-B inhibitor epsilon 5.28 658.3 124.7 ZMYND10 Zinc finger MYND domain-containing protein 10 6.85 835.5 121.9 hCG_23177 - 14.94 1758.9 117.7 KIF3A Kinesin-like protein KIF3A 1.94 224.9 116.1 Q9C0K3_HUMAN Actin-related protein Arp11 3.38 368.0 108.9 NP_056263.1 DPCD protein 2.61 270.5 103.7 GBP1 Interferon-induced guanylate-binding protein 1 1.46 149.1 102.3 NP_660151.2 NAD(P) dependent steroid dehydrogenase-like 1.35 137.4 101.5 NP_689672.2 CDNA FLJ90761 fis, clone THYRO1000099 3.68 372.5 -
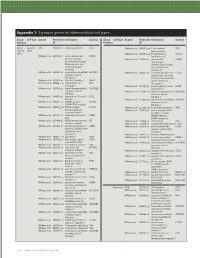
Technical Note, Appendix: an Analysis of Blood Processing Methods to Prepare Samples for Genechip® Expression Profiling (Pdf, 1
Appendix 1: Signature genes for different blood cell types. Blood Cell Type Source Probe Set Description Symbol Blood Cell Type Source Probe Set Description Symbol Fraction ID Fraction ID Mono- Lympho- GSK 203547_at CD4 antigen (p55) CD4 Whitney et al. 209813_x_at T cell receptor TRG nuclear cytes gamma locus cells Whitney et al. 209995_s_at T-cell leukemia/ TCL1A Whitney et al. 203104_at colony stimulating CSF1R lymphoma 1A factor 1 receptor, Whitney et al. 210164_at granzyme B GZMB formerly McDonough (granzyme 2, feline sarcoma viral cytotoxic T-lymphocyte- (v-fms) oncogene associated serine homolog esterase 1) Whitney et al. 203290_at major histocompatibility HLA-DQA1 Whitney et al. 210321_at similar to granzyme B CTLA1 complex, class II, (granzyme 2, cytotoxic DQ alpha 1 T-lymphocyte-associated Whitney et al. 203413_at NEL-like 2 (chicken) NELL2 serine esterase 1) Whitney et al. 203828_s_at natural killer cell NK4 (H. sapiens) transcript 4 Whitney et al. 212827_at immunoglobulin heavy IGHM Whitney et al. 203932_at major histocompatibility HLA-DMB constant mu complex, class II, Whitney et al. 212998_x_at major histocompatibility HLA-DQB1 DM beta complex, class II, Whitney et al. 204655_at chemokine (C-C motif) CCL5 DQ beta 1 ligand 5 Whitney et al. 212999_x_at major histocompatibility HLA-DQB Whitney et al. 204661_at CDW52 antigen CDW52 complex, class II, (CAMPATH-1 antigen) DQ beta 1 Whitney et al. 205049_s_at CD79A antigen CD79A Whitney et al. 213193_x_at T cell receptor beta locus TRB (immunoglobulin- Whitney et al. 213425_at Homo sapiens cDNA associated alpha) FLJ11441 fis, clone Whitney et al. 205291_at interleukin 2 receptor, IL2RB HEMBA1001323, beta mRNA sequence Whitney et al. -

Phosphodiesterase (PDE)
Phosphodiesterase (PDE) Phosphodiesterase (PDE) is any enzyme that breaks a phosphodiester bond. Usually, people speaking of phosphodiesterase are referring to cyclic nucleotide phosphodiesterases, which have great clinical significance and are described below. However, there are many other families of phosphodiesterases, including phospholipases C and D, autotaxin, sphingomyelin phosphodiesterase, DNases, RNases, and restriction endonucleases, as well as numerous less-well-characterized small-molecule phosphodiesterases. The cyclic nucleotide phosphodiesterases comprise a group of enzymes that degrade the phosphodiester bond in the second messenger molecules cAMP and cGMP. They regulate the localization, duration, and amplitude of cyclic nucleotide signaling within subcellular domains. PDEs are therefore important regulators ofsignal transduction mediated by these second messenger molecules. www.MedChemExpress.com 1 Phosphodiesterase (PDE) Inhibitors, Activators & Modulators (+)-Medioresinol Di-O-β-D-glucopyranoside (R)-(-)-Rolipram Cat. No.: HY-N8209 ((R)-Rolipram; (-)-Rolipram) Cat. No.: HY-16900A (+)-Medioresinol Di-O-β-D-glucopyranoside is a (R)-(-)-Rolipram is the R-enantiomer of Rolipram. lignan glucoside with strong inhibitory activity Rolipram is a selective inhibitor of of 3', 5'-cyclic monophosphate (cyclic AMP) phosphodiesterases PDE4 with IC50 of 3 nM, 130 nM phosphodiesterase. and 240 nM for PDE4A, PDE4B, and PDE4D, respectively. Purity: >98% Purity: 99.91% Clinical Data: No Development Reported Clinical Data: No Development Reported Size: 1 mg, 5 mg Size: 10 mM × 1 mL, 10 mg, 50 mg (R)-DNMDP (S)-(+)-Rolipram Cat. No.: HY-122751 ((+)-Rolipram; (S)-Rolipram) Cat. No.: HY-B0392 (R)-DNMDP is a potent and selective cancer cell (S)-(+)-Rolipram ((+)-Rolipram) is a cyclic cytotoxic agent. (R)-DNMDP, the R-form of DNMDP, AMP(cAMP)-specific phosphodiesterase (PDE) binds PDE3A directly. -

Macrophages/Microglia in the Glioblastoma Tumor Microenvironment
International Journal of Molecular Sciences Review Macrophages/Microglia in the Glioblastoma Tumor Microenvironment Jun Ma, Clark C. Chen * and Ming Li * Department of Neurosurgery, University of Minnesota, Minneapolis, MN 55455, USA; [email protected] * Correspondence: [email protected] (C.C.C.); [email protected] (M.L.) Abstract: The complex interaction between glioblastoma and its microenvironment has been rec- ognized for decades. Among various immune profiles, the major population is tumor-associated macrophage, with microglia as its localized homolog. The present definition of such myeloid cells is based on a series of cell markers. These good sentinel cells experience significant changes, facilitat- ing glioblastoma development and protecting it from therapeutic treatments. Huge, complicated mechanisms are involved during the overall processes. A lot of effort has been dedicated to crack the mysterious codes in macrophage/microglia recruiting, activating, reprogramming, and functioning. We have made our path. With more and more key factors identified, a lot of new therapeutic methods could be explored to break the ominous loop, to enhance tumor sensitivity to treatments, and to improve the prognosis of glioblastoma patients. However, it might be a synergistic system rather than a series of clear, stepwise events. There are still significant challenges before the light of truth can shine onto the field. Here, we summarize recent advances in this field, reviewing the path we have been on and where we are now. Keywords: glioblastoma; tumor microenvironment; glioblastoma-associated macrophages/microglia; macrophage; microglia Citation: Ma, J.; Chen, C.C.; Li, M. Macrophages/Microglia in the Glioblastoma Tumor Int. J. Mol. Sci. Microenvironment. 1. Glioblastoma Associated Macrophages/Microglia 2021, 22, 5775. -

Low GSTM3 Expression Is Associated with Poor Disease‐Free Survival In
Yang et al. Diagnostic Pathology (2021) 16:10 https://doi.org/10.1186/s13000-021-01069-4 RESEARCH Open Access Low GSTM3 expression is associated with poor disease‐free survival in resected esophageal squamous cell carcinoma Fu Yang1,2,3,4, Jing Wen2,3,4, Kongjia Luo2,3,4 and Jianhua Fu2,3,4* Abstract Background: Glutathione S-transferase mu 3 (GSTM3) plays a crucial role in tumor progression in various cancers. However, the relationship between GSTM3 expression and the clinical prognosis of esophageal squamous cell carcinoma (ESCC) has not been studied to date. We aimed to characterize the role of GSTM3 in predicting postoperative prognosis of ESCC patients. Methods: In the retrospective study, GSTM3 mRNA levels in 184 ESCC tissues and matched 43 adjacent nontumorous tissues were measured by quantitative real-time PCR. GSTM3 protein levels in 247 ESCC tissues were measured by immunohistochemistry. Results: Downregulation of GSTM3 occurred in 62.8 % of primary ESCC tissues compared with their nontumor counterparts. Patients with low GSTM3 expression tended to exhibit an increased rate of poor differentiation in both the mRNA cohort (p = 0.024) and protein cohort (p = 0.004). In the mRNA cohort, low GSTM3 expression was associated with unfavorable 3-year disease-free survival (DFS) (39.2 % vs. 57.4 %) and 5-year DFS (26.8 % vs. 45.1 %) (p = 0.023). The result was confirmed in the protein cohort. Patients with low GSTM3 expression had unfavorable 3-year disease-free survival (DFS) (18.7 % vs. 33.5 %) and 5-year DFS (5.3 % vs. 30.5 %) (p = 0.006). -

Phosphoproteomics Identify Arachidonic-Acid-Regulated Signal Transduction Pathways Modulating Macrophage Functions with Implications for Ovarian Cancer
Phosphoproteomics identify arachidonic-acid-regulated signal transduction pathways modulating macrophage functions with implications for ovarian cancer Raimund Dietze1¶, Mohamad K. Hammoud1¶, María Gómez-Serrano1, Annika Unger1, Tim Bieringer1§, Florian Finkernagel1, Anna M. Sokol2,3, Andrea Nist4, Thorsten Stiewe4, Silke Reinartz1, Viviane Ponath5, Christian Preußer5, Elke Pogge von Strandmann5, Sabine Müller- Brüsselbach1, Johannes Graumann2,3 and Rolf Müller1* 1Tranlational OncologY Group, Center for Tumor BiologY and Immunology, Philipps University, Marburg, GermanY 2Biomolecular Mass Spectrometry, Max-Planck-Institute for Heart and Lung Research, Bad Nauheim, GermanY 3The German Centre for Cardiovascular Research (DZHK), Partner Site Rhine-Main, Max Planck Institute for Heart and Lung Research, Bad Nauheim, GermanY 4Genomics Core Facility, Philipps UniversitY, Marburg, GermanY 5Institute for Tumor Immunology, Philipps University, Marburg, GermanY §Present address: Hochschule Landshut, 84036 Landshut, GermanY ¶Equal contribution *Corresponding author: Rolf Müller, Center for Tumor Biology and Immunology (ZTI), Philipps University, Hans-Meerwein-Strasse 3, 35043 Marburg, GermanY. Email: [email protected]. Phone: +49 6421 2866236. Running title: Signaling pathways of arachidonic acid in macrophages 1 Abstract Arachidonic acid (AA) is a polYunsaturated fatty acid present at high concentrations in the ovarian cancer (OC) microenvironment and associated with a poor clinical outcome. In the present studY, we have unraveled a potential link between AA and macrophage functions. Methods: AA-triggered signal transduction was studied in primary monocyte-derived macrophages (MDMs) by phosphoproteomics, transcriptional profiling, measurement of intracellular Ca2+ accumulation and reactive oxygen species production in conjunction with bioinformatic analyses. Functional effects were investigated by actin filament staining, quantification of macropinocytosis and analysis of extracellular vesicle release.