Lipid II: a Central Component in Bacterial Cell Wall Synthesis and a Target for Antibiotics
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Synthesis and Biological Evaluation of Trisindolyl-Cycloalkanes and Bis- Indolyl Naphthalene Small Molecules As Potent Antibacterial and Antifungal Agents
Synthesis and Biological Evaluation of Trisindolyl-Cycloalkanes and Bis- Indolyl Naphthalene Small Molecules as Potent Antibacterial and Antifungal Agents Dissertation Zur Erlangung des akademischen Grades doctor rerum naturalium (Dr. rer. nat.) Vorgelegt der Naturwissenschaftlichen Fakultät I Institut für Pharmazie Fachbereich für Pharmazeutische Chemie der Martin-Luther-Universität Halle-Wittenberg von Kaveh Yasrebi Geboren am 09.14.1987 in Teheran/Iran (Islamische Republik) Gutachter: 1. Prof. Dr. Andreas Hilgeroth (Martin-Luther-Universität Halle-Wittenberg, Germany) 2. Prof. Dr. Sibel Süzen (Ankara Üniversitesi, Turkey) 3. Prof. Dr. Michael Lalk (Ernst-Moritz-Arndt-Universität Greifswald, Germany) Halle (Saale), den 21. Juli 2020 Selbstständigkeitserklärung Hiermit erkläre ich gemäß § 5 (2) b der Promotionsordnung der Naturwissenschaftlichen Fakultät I – Institut für Pharmazie der Martin-Luther-Universität Halle-Wittenberg, dass ich die vorliegende Arbeit selbstständig und ohne Benutzung anderer als der angegebenen Hilfsmittel und Quellen angefertigt habe. Alle Stellen, die wörtlich oder sinngemäß aus Veröffentlichungen entnommen sind, habe ich als solche kenntlich gemacht. Ich erkläre ferner, dass diese Arbeit in gleicher oder ähnlicher Form bisher keiner anderen Prüfbehörde zur Erlangung des Doktorgrades vorgelegt wurde. Halle (Saale), den 21. Juli 2020 Kaveh Yasrebi Acknowledgement This study was carried out from June 2015 to July 2017 in the Research Group of Drug Development and Analysis led by Prof. Dr. Andreas Hilgeroth at the Institute of Pharmacy, Martin-Luther-Universität Halle-Wittenberg. I would like to thank all the people for their participation who supported my work in this way and helped me obtain good results. First of all, I would like to express my gratitude to Prof. Dr. Andreas Hilgeroth for providing me with opportunity to carry out my Ph.D. -

A New Antibiotic Kills Pathogens Without Detectable Resistance
ARTICLE doi:10.1038/nature14098 A new antibiotic kills pathogens without detectable resistance Losee L. Ling1*, Tanja Schneider2,3*, Aaron J. Peoples1, Amy L. Spoering1, Ina Engels2,3, Brian P. Conlon4, Anna Mueller2,3, Till F. Scha¨berle3,5, Dallas E. Hughes1, Slava Epstein6, Michael Jones7, Linos Lazarides7, Victoria A. Steadman7, Douglas R. Cohen1, Cintia R. Felix1, K. Ashley Fetterman1, William P. Millett1, Anthony G. Nitti1, Ashley M. Zullo1, Chao Chen4 & Kim Lewis4 Antibiotic resistance is spreading faster than the introduction of new compounds into clinical practice, causing a public health crisis. Most antibiotics were produced by screening soil microorganisms, but this limited resource of cultivable bacteria was overmined by the 1960s. Synthetic approaches to produce antibiotics have been unable to replace this platform. Uncultured bacteria make up approximately 99% of all species in external environments, and are an untapped source of new antibiotics. We developed several methods to grow uncultured organisms by cultivation in situ or by using specific growth factors. Here we report a new antibiotic that we term teixobactin, discovered in a screen of uncultured bacteria. Teixobactin inhibits cell wall synthesis by binding to a highly conserved motif of lipid II (precursor of peptidoglycan) and lipid III (precursor of cell wall teichoic acid). We did not obtain any mutants of Staphylococcus aureus or Mycobacterium tuberculosis resistant to teixobactin. The properties of this compound suggest a path towards developing antibiotics that are likely to avoid development of resistance. Widespread introduction of antibiotics in the 1940s, beginning with factors through the chambers enables growth of uncultured bacteria in penicillin1,2 and streptomycin3, transformed medicine, providing effec- their natural environment. -

Novel Antimicrobial Agents Inhibiting Lipid II Incorporation Into Peptidoglycan Essay MBB
27 -7-2019 Novel antimicrobial agents inhibiting lipid II incorporation into peptidoglycan Essay MBB Mark Nijland S3265978 Supervisor: Prof. Dr. Dirk-Jan Scheffers Molecular Microbiology University of Groningen Content Abstract..............................................................................................................................................2 1.0 Peptidoglycan biosynthesis of bacteria ........................................................................................3 2.0 Novel antimicrobial agents ...........................................................................................................4 2.1 Teixobactin ...............................................................................................................................4 2.2 tridecaptin A1............................................................................................................................7 2.3 Malacidins ................................................................................................................................8 2.4 Humimycins ..............................................................................................................................9 2.5 LysM ........................................................................................................................................ 10 3.0 Concluding remarks .................................................................................................................... 11 4.0 references ................................................................................................................................. -
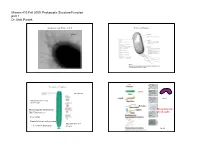
Parsek Micro410 Lecture
Microm 410 Fall 2009: Prokaryotic Structure/Function part 1 Dr. Matt Parsek Organization of the Prokaryotic Cell Prokaryotic Structures fimbriae Size Range of Prokaryotes bacillus See Table 4.1 (rigid) vibrio Nanobacteria 0.05‐0.2 µm (0.14‐0.2 µm) (flexible) Thiomargarita namibiensis Mycoplasma are 700‐750 µm (Fig. 4.2) pleomorphic green alga Nanochlorum eukaryotum Mycoplasma 0.1‐ ~1-2 µm in diameter 0.3 µm Fig. 4.1 Microm 410 Fall 2009: Prokaryotic Structure/Function part 1 Dr. Matt Parsek Staining cells for Microscopic observation Cell Arrangements Motility- ~80% of prokaryotes are motile streptococcus Staining properties: Gram Stain staphylococcus Fig. 2.3 Gram Stain (1884) (Bacteria) Gram-negative mixed culture Gram-positive Fig. 2.3 and 2.4 Microm 410 Fall 2009: Prokaryotic Structure/Function part 1 Dr. Matt Parsek Functions of the cytoplasmic membrane The phospholipid bi‐layer Fig. 4.9 Fig. 4.4 What is the structure of bacterial phospholipids? Other components of the cytoplasmic membrane Figs. 4.5‐4.6 Microm 410 Fall 2009: Prokaryotic Structure/Function part 1 Dr. Matt Parsek Archaeal membranes can be a lipid monolayer Archaeal phospholipids have an ether linkage Fig. 4.7 Fig. 4.8 Importance of Cell Wall Schematic diagram cell wall • Provides rigidity to cell allowing cell to withstand the large osmotic/ionic Fig. 4.16 changes a bacterium may experience in its environment, and turgor pressure of cytoplasm (conc. of solutes in cytoplasm). Cell lysis • May have a role in shape determination. • Provides a barrier against certain toxic chemical and biological agents. • Site of action of some of the most commonly used antibiotics used to treat bacterial infections (penicillin family). -
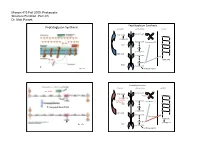
Parsek Lecture #3
Microm 410 Fall 2009: Prokaryotic Structure/Function: Part 2/3 Dr. Matt Parsek Peptidoglycan Synthesis Peptidoglycan Synthesis cytoplasm cell membrane cell wall Bactoprenol-P Pi UDP-NAM M G pentapeptide G M Bactoprenol Bactoprenol-P-P P M UMP G P NAM G M pentapeptide M G UDP-NAG Bactoprenol G P NAM‐NAG P NAM-NAG UMP pentapeptide Fig. 6.7a Interbridge peptide Peptidoglycan Synthesis Cross-linking of Peptidoglycan Strands cytoplasm cell membrane cell wall autolysins Bactoprenol-P Pi UDP-NAM Bacitracin M G pentapeptide G D-cycloserine Bactoprenol M (Oxamycin) Bactoprenol-P-P P M UMP G P Transpeptidase (FtsI) NAM G M pentapeptide M G UDP-NAG Bactoprenol G Vancomycin P NAM‐NAG P NAM-NAG pentapeptide UMP Fig. 6.7b pentapeptide Interbridge peptide Microm 410 Fall 2009: Prokaryotic Structure/Function: Part 2/3 Dr. Matt Parsek Cross-linking of Peptidoglycan Strands Antibiotic Resistance autolysins • Inactivate antibiotic β-lactamase (penicillinase) Clavulanic acid β-lactams Augmentin and Trimentin (combination of clavulanic acid and transpeptidase amoxicillin or ampicillin respectively) penicillins and cephalosporins lysozyme • Change chemistry of target site • Limit access of the antibiotic to target site Fig. 6.5 Cell Shape Determination • Modifications made to Peptidoglycan: ‐ lysozyme: Protoplasts/spheroplasts ‐ autolysins Bacillus subtilis ‐ endopeptidase Heliobacter pylori • Protein(s) may play a major role ‐ MreB protein Caulobacter crescentus ‐ MreB has homology to actin, a component of the cytoskeleton of eukaryotes. Shape determining protein‐ crescentin Fig. 6.4 Microm 410 Fall 2009: Prokaryotic Structure/Function: Part 2/3 Dr. Matt Parsek Cell Wall Gram-positive Bacteria intermediate filaments in the bacteria Caulobacter crescentus glycerol similar predicted structures of crescentin and intermediate filaments Fig. -

WO 2015/028850 Al 5 March 2015 (05.03.2015) P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2015/028850 Al 5 March 2015 (05.03.2015) P O P C T (51) International Patent Classification: AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, C07D 519/00 (2006.01) A61P 39/00 (2006.01) BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, C07D 487/04 (2006.01) A61P 35/00 (2006.01) DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, A61K 31/5517 (2006.01) A61P 37/00 (2006.01) HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KN, KP, KR, A61K 47/48 (2006.01) KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, (21) International Application Number: OM, PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, PCT/IB2013/058229 SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, (22) International Filing Date: TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, 2 September 2013 (02.09.2013) ZW. (25) Filing Language: English (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (26) Publication Language: English GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, SZ, TZ, (71) Applicant: HANGZHOU DAC BIOTECH CO., LTD UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, RU, TJ, [US/CN]; Room B2001-B2019, Building 2, No 452 Sixth TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, DK, Street, Hangzhou Economy Development Area, Hangzhou EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, IT, LT, LU, LV, City, Zhejiang 310018 (CN). -

Escherichia Coli K–12 Derived from the Ecocyc Database Daniel S Weaver1*,Ingridmkeseler1,Amandamackie2, Ian T Paulsen2 and Peter D Karp1
Weaver et al. BMC Systems Biology 2014, 8:79 http://www.biomedcentral.com/1752-0509/8/79 RESEARCH ARTICLE OpenAccess A genome-scale metabolic flux model of Escherichia coli K–12 derived from the EcoCyc database Daniel S Weaver1*,IngridMKeseler1,AmandaMackie2, Ian T Paulsen2 and Peter D Karp1 Abstract Background: Constraint-based models of Escherichia coli metabolic flux have played a key role in computational studies of cellular metabolism at the genome scale. We sought to develop a next-generation constraint-based E. coli model that achieved improved phenotypic prediction accuracy while being frequently updated and easy to use. We also sought to compare model predictions with experimental data to highlight open questions in E. coli biology. Results: We present EcoCyc–18.0–GEM, a genome-scale model of the E. coli K–12 MG1655 metabolic network. The model is automatically generated from the current state of EcoCyc using the MetaFlux software, enabling the release of multiple model updates per year. EcoCyc–18.0–GEM encompasses 1445 genes, 2286 unique metabolic reactions, and 1453 unique metabolites. We demonstrate a three-part validation of the model that breaks new ground in breadth and accuracy: (i) Comparison of simulated growth in aerobic and anaerobic glucose culture with experimental results from chemostat culture and simulation results from the E. coli modeling literature. (ii) Essentiality prediction for the 1445 genes represented in the model, in which EcoCyc–18.0–GEM achieves an improved accuracy of 95.2% in predicting the growth phenotype of experimental gene knockouts. (iii) Nutrient utilization predictions under 431 different media conditions, for which the model achieves an overall accuracy of 80.7%. -

Antibiotic Discovery
ANTIBIOTIC DISCOVERY RESISTANCE PROFILING OF MICROBIAL GENOMES TO REVEAL NOVEL ANTIBIOTIC NATURAL PRODUCTS By CHELSEA WALKER, H. BSc. A Thesis Submitted to the School of Graduate Studies in Partial Fulfilment of the Requirements for the Degree Master of Science McMaster University © Copyright by Chelsea Walker, May 2017 McMaster University MASTER OF SCIENCE (2017) Hamilton, Ontario (Biochemistry and Biomedical Sciences) TITLE: Resistance Profiling of Microbial Genomes to Reveal Novel Antibiotic Natural Products. AUTHOR: Chelsea Walker, H. BSc. (McMaster University) SUPERVISOR: Dr. Nathan A. Magarvey. COMMITTEE MEMBERS: Dr. Eric Brown and Dr. Michael G. Surette. NUMBER OF PAGES: xvii, 168 ii Lay Abstract It would be hard to imagine a world where we could no longer use the antibiotics we are routinely being prescribed for common bacterial infections. Currently, we are in an era where this thought could become a reality. Although we have been able to discover antibiotics in the past from soil dwelling microbes, this approach to discovery is being constantly challenged. At the same time, the bacteria are getting smarter in their ways to evade antibiotics, in the form of resistance, or self-protection mechanisms. As such is it essential to devise methods which can predict the potential for resistance to the antibiotics we use early in the discovery and isolation process. By using what we have learned in the past about how bacteria protect themselves for antibiotics, we can to stay one step ahead of them as we continue to search for new sources of antibiotics from bacteria. iii Abstract Microbial natural products have been an invaluable resource for providing clinically relevant therapeutics for almost a century, including most of the commonly used antibiotics that are still in medical use today. -

Synthesis and Structure−Activity Relationships of Teixobactin
Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Special Issue: Antimicrobial Therapeutics Reviews REVIEW Synthesis and structure−activity relationships of teixobactin John A. Karas,1 Fan Chen,1 Elena K. Schneider-Futschik,1,2 Zhisen Kang,1 Maytham Hussein,1 James Swarbrick,1 Daniel Hoyer,1,3,4 Andrew M. Giltrap,5 Richard J. Payne,5 Jian Li,6 and Tony Velkov1 1Department of Pharmacology & Therapeutics, School of Biomedical Sciences, Faculty of Medicine, Dentistry and Health Sciences, the University of Melbourne, Parkville, Victoria, Australia. 2Lung Health Research Centre, Department of Pharmacology & Therapeutics, the University of Melbourne, Parkville, Victoria, Australia. 3The Florey Institute of Neuroscience and Mental Health, the University of Melbourne, Parkville, Victoria, Australia. 4Department of Molecular Medicine, the Scripps Research Institute, La Jolla, California. 5School of Chemistry, the University of Sydney, Sydney, New South Wales, Australia. 6Monash Biomedicine Discovery Institute, Department of Microbiology, Monash University, Clayton, Victoria, Australia Address for correspondence: Tony Velkov and John A. Karas, Department of Pharmacology & Therapeutics, School of Biomedical Sciences, Faculty of Medicine, Dentistry and Health Sciences, the University of Melbourne, Parkville, VIC 3010, Australia. [email protected] OR [email protected] The discovery of antibiotics has led to the effective treatment of bacterial infections that were otherwise fatal and has had a transformative effect on modern medicine. Teixobactin is an unusual depsipeptide natural product that was recently discovered from a previously unculturable soil bacterium and found to possess potent antibacterial activity against several Gram positive pathogens, including methicillin-resistant Staphylococcus aureus and vancomycin- resistant Enterococci. -

E. Coli Pbp1b, Moenomycin-Based
Investigating the Ligand Interactions Between E. coli PBP1b, Moenomycin-based Compounds, and Beta-Lactam Compounds Peter Alexander MSc by Research 2017 i CERTIFICATE OF ORIGINALITY This is to certify that I am responsible for the work submitted in this thesis, that the original work is my own, except as specified in the acknowledgements and in references, and that neither the thesis nor the original work contained therein has been previously submitted to any institution for a degree. Signature: Name: Date: CERTIFICATE OF COMPLIANCE This is to certify that this project has been carried out in accordance with University principles regarding ethics and health and safety. Forms are available to view on request. Signature: Name: Date: ii Abstract Antimicrobial resistance is a growing problem in this era. Resistance to the majority of clinical antibiotics including those of a ‘last line of defence’ nature has been seen in a number of laboratory and clinical settings. One method aiming at reducing this problem is altering existing antimicrobial compounds, in order to improve pharmacological effects (avoiding resistance mechanisms, improved spectrum of use). Analysis of the interactions between the antimicrobial compounds and their targets can determine whether modifications to current antimicrobials (such as moenomycin A, a glycosyltransferase inhibitor) have altered the mode of action. ecoPBP1B is a bifunctional glycosyltransferase that could be used as a model for beta lactams and moenomycins, aiding in the design and development of novel antimicrobials based on these families. Moenomycin A has not seen high clinical usage due to poor pharmacokinetics and bioavailability. This project aimed to show whether ecoPBP1b can be used as a model for novel antimicrobials, such as seeing whether Moenomycin A analogues (with cell penetrating peptides to facilitate entry into the bacterial cell) still retain their ability to bind to glycosyltransferases. -

Lipid II As a Target for Antibiotics
Nature Reviews Drug Discovery | AOP, published online 10 March 2006; doi:10.1038/nrd2004 REVIEWS Lipid II as a target for antibiotics Eefjan Breukink and Ben de Kruijff Abstract | Lipid II is a membrane-anchored cell-wall precursor that is essential for bacterial cell-wall biosynthesis. The effectiveness of targeting Lipid II as an antibacterial strategy is highlighted by the fact that it is the target for at least four different classes of antibiotic, including the clinically important glycopeptide antibiotic vancomycin. However, the growing problem of bacterial resistance to many current drugs, including vancomycin, has led to increasing interest in the therapeutic potential of other classes of compound that target Lipid II. Here, we review progress in understanding of the antibacterial activities of these compounds, which include lantibiotics, mannopeptimycins and ramoplanin, and consider factors that will be important in exploiting their potential as new treatments for bacterial infections. Since the discovery of penicillin more than 75 years ago, for novel antibacterial drugs, and here we review their antibiotics have had an immense impact on the treatment mode of action and their antibacterial activities, and use of infections caused by bacteria. However, the widespread, this as a basis to discuss their potential as novel drugs for and sometimes inappropriate, use of antibiotics has gen- combating antibiotic-resistant bacteria. erated a strong evolutionary pressure for the emergence of bacteria that either have an inherent resistance to a The role of Lipid II in cell-wall synthesis particular antibiotic or have the capacity to acquire such The cell wall (FIG. 1) of all bacteria comprises a polymer of resistance. -

Alternative Functions of ABC Transporters in Bacillus Subtilis and Listeria Monocytogenes
microorganisms Review Not Just Transporters: Alternative Functions of ABC Transporters in Bacillus subtilis and Listeria monocytogenes Jeanine Rismondo * and Lisa Maria Schulz Department of General Microbiology, GZMB, Georg-August-University Göttingen, Grisebachstr. 8, D-37077 Göttingen, Germany; [email protected] * Correspondence: [email protected]; Tel.: +49-551-39-33796 Abstract: ATP-binding cassette (ABC) transporters are usually involved in the translocation of their cognate substrates, which is driven by ATP hydrolysis. Typically, these transporters are required for the import or export of a wide range of substrates such as sugars, ions and complex organic molecules. ABC exporters can also be involved in the export of toxic compounds such as antibiotics. However, recent studies revealed alternative detoxification mechanisms of ABC transporters. For instance, the ABC transporter BceAB of Bacillus subtilis seems to confer resistance to bacitracin via target protection. In addition, several transporters with functions other than substrate export or import have been identified in the past. Here, we provide an overview of recent findings on ABC transporters of the Gram-positive organisms B. subtilis and Listeria monocytogenes with transport or regulatory functions affecting antibiotic resistance, cell wall biosynthesis, cell division and sporulation. Keywords: ABC transporter; antibiotic resistance; Gram-positive bacteria; cell wall 1. Introduction ATP-binding cassette (ABC) transporters can be found in all kingdoms of life and Citation: Rismondo, J.; Schulz, L.M. can be subdivided in three main groups: eukaryotic transporters, bacterial importers and Not Just Transporters: Alternative bacterial exporters. In these transporters, the translocation of cognate substrates is driven Functions of ABC Transporters in by ATP hydrolysis [1].