The Splicing Regulator PTBP1 Controls the Activity of the Transcription Factor
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Ptbp2 Represses Adult-Specific Splicing to Regulate the Generation of Neuronal Precursors in the Embryonic Brain
Downloaded from genesdev.cshlp.org on October 4, 2021 - Published by Cold Spring Harbor Laboratory Press Ptbp2 represses adult-specific splicing to regulate the generation of neuronal precursors in the embryonic brain Donny D. Licatalosi,1,4 Masato Yano,1,5 John J. Fak,1 Aldo Mele,1 Sarah E. Grabinski,2 Chaolin Zhang,1 and Robert B. Darnell1,3,6 1Laboratory of Molecular Neuro-Oncology, The Rockefeller University, New York, New York 10065, USA; 2Center for RNA Molecular Biology, Case Western Reserve University, Cleveland, Ohio 44106, USA; 3Howard Hughes Medical Institute, The Rockefeller University, New York, New York 10065, USA Two polypyrimidine tract RNA-binding proteins (PTBs), one near-ubiquitously expressed (Ptbp1) and another highly tissue-restricted (Ptbp2), regulate RNA in interrelated but incompletely understood ways. Ptbp1, a splicing regulator, is replaced in the brain and differentiated neuronal cell lines by Ptbp2. To define the roles of Ptbp2 in the nervous system, we generated two independent Ptbp2-null strains, unexpectedly revealing that Ptbp2 is expressed in neuronal progenitors and is essential for postnatal survival. A HITS-CLIP (high-throughput sequencing cross- linking immunoprecipitation)-generated map of reproducible Ptbp2–RNA interactions in the developing mouse neocortex, combined with results from splicing-sensitive microarrays, demonstrated that the major action of Ptbp2 is to inhibit adult-specific alternative exons by binding pyrimidine-rich sequences upstream of and/or within them. These regulated exons are present in mRNAs encoding proteins associated with control of cell fate, proliferation, and the actin cytoskeleton, suggesting a role for Ptbp2 in neurogenesis. Indeed, neuronal progenitors in the Ptbp2-null brain exhibited an aberrant polarity and were associated with regions of premature neurogenesis and reduced progenitor pools. -

Supplementary Methods
Supplementary methods Human lung tissues and tissue microarray (TMA) All human tissues were obtained from the Lung Cancer Specialized Program of Research Excellence (SPORE) Tissue Bank at the M.D. Anderson Cancer Center (Houston, TX). A collection of 26 lung adenocarcinomas and 24 non-tumoral paired tissues were snap-frozen and preserved in liquid nitrogen for total RNA extraction. For each tissue sample, the percentage of malignant tissue was calculated and the cellular composition of specimens was determined by histological examination (I.I.W.) following Hematoxylin-Eosin (H&E) staining. All malignant samples retained contained more than 50% tumor cells. Specimens resected from NSCLC stages I-IV patients who had no prior chemotherapy or radiotherapy were used for TMA analysis by immunohistochemistry. Patients who had smoked at least 100 cigarettes in their lifetime were defined as smokers. Samples were fixed in formalin, embedded in paraffin, stained with H&E, and reviewed by an experienced pathologist (I.I.W.). The 413 tissue specimens collected from 283 patients included 62 normal bronchial epithelia, 61 bronchial hyperplasias (Hyp), 15 squamous metaplasias (SqM), 9 squamous dysplasias (Dys), 26 carcinomas in situ (CIS), as well as 98 squamous cell carcinomas (SCC) and 141 adenocarcinomas. Normal bronchial epithelia, hyperplasia, squamous metaplasia, dysplasia, CIS, and SCC were considered to represent different steps in the development of SCCs. All tumors and lesions were classified according to the World Health Organization (WHO) 2004 criteria. The TMAs were prepared with a manual tissue arrayer (Advanced Tissue Arrayer ATA100, Chemicon International, Temecula, CA) using 1-mm-diameter cores in triplicate for tumors and 1.5 to 2-mm cores for normal epithelial and premalignant lesions. -

Proteomics Analysis of Cellular Proteins Co-Immunoprecipitated with Nucleoprotein of Influenza a Virus (H7N9)
Article Proteomics Analysis of Cellular Proteins Co-Immunoprecipitated with Nucleoprotein of Influenza A Virus (H7N9) Ningning Sun 1,:, Wanchun Sun 2,:, Shuiming Li 3, Jingbo Yang 1, Longfei Yang 1, Guihua Quan 1, Xiang Gao 1, Zijian Wang 1, Xin Cheng 1, Zehui Li 1, Qisheng Peng 2,* and Ning Liu 1,* Received: 26 August 2015 ; Accepted: 22 October 2015 ; Published: 30 October 2015 Academic Editor: David Sheehan 1 Central Laboratory, Jilin University Second Hospital, Changchun 130041, China; [email protected] (N.S.); [email protected] (J.Y.); [email protected] (L.Y.); [email protected] (G.Q.); [email protected] (X.G.); [email protected] (Z.W.); [email protected] (X.C.); [email protected] (Z.L.) 2 Key Laboratory of Zoonosis Research, Ministry of Education, Institute of Zoonosis, Jilin University, Changchun 130062, China; [email protected] 3 College of Life Sciences, Shenzhen University, Shenzhen 518057, China; [email protected] * Correspondence: [email protected] (Q.P.); [email protected] (N.L.); Tel./Fax: +86-431-8879-6510 (Q.P. & N.L.) : These authors contributed equally to this work. Abstract: Avian influenza A viruses are serious veterinary pathogens that normally circulate among avian populations, causing substantial economic impacts. Some strains of avian influenza A viruses, such as H5N1, H9N2, and recently reported H7N9, have been occasionally found to adapt to humans from other species. In order to replicate efficiently in the new host, influenza viruses have to interact with a variety of host factors. In the present study, H7N9 nucleoprotein was transfected into human HEK293T cells, followed by immunoprecipitated and analyzed by proteomics approaches. -

Polypyrimidine Tract Binding Proteins Are Essential for B Cell Development
bioRxiv preprint doi: https://doi.org/10.1101/769141; this version posted September 14, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Polypyrimidine Tract Binding Proteins are essential for B cell development Authors: Elisa Monzón-Casanova1,2*, Louise S. Matheson1, Kristina Tabbada3, Kathi Zarnack4, Christopher W. J. Smith2 and Martin Turner1* Affiliations: 1Laboratory of Lymphocyte Signalling and Development, The Babraham Institute, Cambridge, UK; 2Department of Biochemistry, University of Cambridge, Cambridge, UK; 3Next Generation Sequencing Facility, The Babraham Institute, Cambridge, UK; 4Buchmann Institute for Molecular Life Sciences, Goethe University Frankfurt, Frankfurt am Main, Germany *Corresponding authors: Elisa Monzón-Casanova ([email protected]) and Martin Turner ([email protected]) 1 bioRxiv preprint doi: https://doi.org/10.1101/769141; this version posted September 14, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Abstract During B cell development, recombination of immunoglobulin loci is tightly coordinated with the cell cycle to avoid unwanted rearrangements of other genomic locations. Several factors have been identified that suppress proliferation in late-pre-B cells to allow light chain recombination. By comparison, our knowledge of factors limiting proliferation during heavy chain recombination at the pro-B cell stage is very limited. -

Identification of Proteins Involved in the Maintenance of Genome Stability
Identification of Proteins Involved in the Maintenance of Genome Stability by Edith Hang Yu Cheng A thesis submitted in conformity with the requirements for the degree of Doctor of Philosophy Department of Biochemistry University of Toronto ©Copyright by Edith Cheng2015 Identification of Proteins Involved in the Maintenance of Genome Stability Edith Cheng Doctor of Philosophy Department of Biochemistry University of Toronto 2015 Abstract Aberrant changes to the genome structure underlie numerous human diseases such as cancers. The functional characterization ofgenesand proteins that maintain chromosome stability will be important in understanding disease etiology and developing therapeutics. I took a multi-faceted approach to identify and characterize genes involved in the maintenance of genome stability. As biological pathways involved in genome maintenance are highly conserved in evolution, results from model organisms can greatly facilitate functional discovery in humans. In S. cerevisiae, I identified 47 essential gene depletions with elevated levels of spontaneous DNA damage foci and 92 depletions that caused elevated levels of chromosome rearrangements. Of these, a core subset of 15 DNA replication genes demonstrated both phenotypes when depleted. Analysis of rearrangement breakpoints revealed enrichment at yeast fragile sites, Ty retrotransposons, early origins of replication and replication termination sites. Together, thishighlighted the integral role of DNA replicationin genome maintenance. In light of my findings in S. cerevisiae, I identified a list of 153 human proteins that interact with the nascentDNA at replication forks, using a DNA pull down strategy (iPOND) in human cell lines. As a complementary approach for identifying human proteins involved in genome ii maintenance, I usedthe BioID techniqueto discernin vivo proteins proximal to the human BLM- TOP3A-RMI1-RMI2 genome stability complex, which has an emerging role in DNA replication progression. -
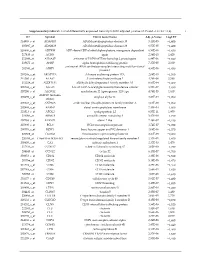
Supplementary Table S1. List of Differentially Expressed
Supplementary table S1. List of differentially expressed transcripts (FDR adjusted p‐value < 0.05 and −1.4 ≤ FC ≥1.4). 1 ID Symbol Entrez Gene Name Adj. p‐Value Log2 FC 214895_s_at ADAM10 ADAM metallopeptidase domain 10 3,11E‐05 −1,400 205997_at ADAM28 ADAM metallopeptidase domain 28 6,57E‐05 −1,400 220606_s_at ADPRM ADP‐ribose/CDP‐alcohol diphosphatase, manganese dependent 6,50E‐06 −1,430 217410_at AGRN agrin 2,34E‐10 1,420 212980_at AHSA2P activator of HSP90 ATPase homolog 2, pseudogene 6,44E‐06 −1,920 219672_at AHSP alpha hemoglobin stabilizing protein 7,27E‐05 2,330 aminoacyl tRNA synthetase complex interacting multifunctional 202541_at AIMP1 4,91E‐06 −1,830 protein 1 210269_s_at AKAP17A A‐kinase anchoring protein 17A 2,64E‐10 −1,560 211560_s_at ALAS2 5ʹ‐aminolevulinate synthase 2 4,28E‐06 3,560 212224_at ALDH1A1 aldehyde dehydrogenase 1 family member A1 8,93E‐04 −1,400 205583_s_at ALG13 ALG13 UDP‐N‐acetylglucosaminyltransferase subunit 9,50E‐07 −1,430 207206_s_at ALOX12 arachidonate 12‐lipoxygenase, 12S type 4,76E‐05 1,630 AMY1C (includes 208498_s_at amylase alpha 1C 3,83E‐05 −1,700 others) 201043_s_at ANP32A acidic nuclear phosphoprotein 32 family member A 5,61E‐09 −1,760 202888_s_at ANPEP alanyl aminopeptidase, membrane 7,40E‐04 −1,600 221013_s_at APOL2 apolipoprotein L2 6,57E‐11 1,600 219094_at ARMC8 armadillo repeat containing 8 3,47E‐08 −1,710 207798_s_at ATXN2L ataxin 2 like 2,16E‐07 −1,410 215990_s_at BCL6 BCL6 transcription repressor 1,74E‐07 −1,700 200776_s_at BZW1 basic leucine zipper and W2 domains 1 1,09E‐06 −1,570 222309_at -

The Splicing Factor PTBP1 Promotes Expression of Oncogenic Splice Variants And
Author Manuscript Published OnlineFirst on July 16, 2018; DOI: 10.1158/1078-0432.CCR-17-3850 Author manuscripts have been peer reviewed and accepted for publication but have not yet been edited. The splicing factor PTBP1 promotes expression of oncogenic splice variants and predicts poor prognosis in patients with non-muscle invasive bladder cancer Pamela Bielli1,2, Valentina Panzeri2,3,4, Rossano Lattanzio5,6, Simona Mutascio1,2, Marco Pieraccioli1,2, Elisabetta Volpe2, Vincenzo Pagliarulo7, Mauro Piantelli5, Antonella Giannantoni8; Savino M. Di Stasi9,10 and Claudio Sette2,4,10 1Department of Biomedicine and Prevention, University of Rome Tor Vergata, Rome, Italy; 2Fondazione Santa Lucia IRCCS, Rome, Italy; 3Department of science medical/chirurgic and translational medicine, University of Rome “Sapienza”, Rome, Italy; 4Institute of Human Anatomy and Cell Biology, Università Cattolica del Sacro Cuore, Rome, Italy; 5Department of Medical, Oral & Biotechnological Sciences, G. d'Annunzio University, Chieti, Italy; 6 Center of Excellence on Aging and Translational Medicine (CeSi-Met), G. d'Annunzio University, Chieti, Italy; 7Department of Emergency and Organ Transplantation, University Aldo Moro , Bari, Italy; 8Department of Surgical and Biomedical Sciences, University of Perugia, Perugia, Italy; 9Department of Experimental Medicine and Surgery, University of Rome Tor Vergata, Rome, Italy. 10These authors equally contributed to the work. Running title: PTBP1 is overexpressed in bladder cancer Keywords: PTBP1, alternative splicing, bladder cancer, risk factor, prognostic marker Conflict of interest statement: The Authors have no conflicts of interest to disclose. Financial Support: The research was supported by the Associazione Italiana Ricerca sul Cancro (AIRC; IG18790) and by Italian Ministry of Health “Ricerca Finalizzata 2011” (GR- 2011-02348423). -

Significance of Polypyrimidine Tract–Binding Protein 1 Expression In
Published OnlineFirst April 22, 2015; DOI: 10.1158/1535-7163.MCT-14-0142 Cancer Biology and Signal Transduction Molecular Cancer Therapeutics Significance of Polypyrimidine Tract–Binding Protein 1 Expression in Colorectal Cancer Hidekazu Takahashi1, Junichi Nishimura1, Yoshinori Kagawa1,2, Yoshihiro Kano3, Yusuke Takahashi1,4, Xin Wu1, Masayuki Hiraki1, Atsushi Hamabe1, Masamitsu Konno3, Naotsugu Haraguchi1, Ichiro Takemasa1, Tsunekazu Mizushima1, Masaru Ishii2, Koshi Mimori4, Hideshi Ishii3, Yuichiro Doki1, Masaki Mori1, and Hirofumi Yamamoto1 Abstract Polypyrimidine tract–binding protein (PTBP1) is an RNA- observed that PTBP1 affects cell invasion, which was partially binding protein with various molecular functions related to correlated to CD44 splicing, and this correlation was also RNA metabolism and a major repressive regulator of alternative confirmed in clinical samples. PTBP1 expression also affected splicing, causing exon skipping in numerous alternatively anchorage-independent growth in colorectal cancer cell lines. spliced pre-mRNAs. Here, we have investigated the role of PTBP1 expression also affected cell proliferation. Using time- PTBP1 in colorectal cancer. PTBP1 expression levels were sig- lapse imaging analysis, PTBP1 was implicated in prolonged nificantly overexpressed in cancerous tissues compared with G2–M phase in HCT116 cells. As for the mechanism of pro- corresponding normal mucosal tissues. We also observed that longed G2–M phase in HCT116 siPTBP1 cells, Western blotting PTBP1 expression levels, c-MYC expression levels, and PKM2: revealed that PTBP1 expression level was correlated to PKM1 ratio were positively correlated in colorectal cancer CDK11p58 expression level, which was reported to play an specimens. Moreover, PTBP1 expression levels were positively important role on progression to complete mitosis. These correlated to poor prognosis and lymph node metastasis. -

A SARS-Cov-2 Protein Interaction Map Reveals Targets for Drug Repurposing
Article A SARS-CoV-2 protein interaction map reveals targets for drug repurposing https://doi.org/10.1038/s41586-020-2286-9 A list of authors and affiliations appears at the end of the paper Received: 23 March 2020 Accepted: 22 April 2020 A newly described coronavirus named severe acute respiratory syndrome Published online: 30 April 2020 coronavirus 2 (SARS-CoV-2), which is the causative agent of coronavirus disease 2019 (COVID-19), has infected over 2.3 million people, led to the death of more than Check for updates 160,000 individuals and caused worldwide social and economic disruption1,2. There are no antiviral drugs with proven clinical efcacy for the treatment of COVID-19, nor are there any vaccines that prevent infection with SARS-CoV-2, and eforts to develop drugs and vaccines are hampered by the limited knowledge of the molecular details of how SARS-CoV-2 infects cells. Here we cloned, tagged and expressed 26 of the 29 SARS-CoV-2 proteins in human cells and identifed the human proteins that physically associated with each of the SARS-CoV-2 proteins using afnity-purifcation mass spectrometry, identifying 332 high-confdence protein–protein interactions between SARS-CoV-2 and human proteins. Among these, we identify 66 druggable human proteins or host factors targeted by 69 compounds (of which, 29 drugs are approved by the US Food and Drug Administration, 12 are in clinical trials and 28 are preclinical compounds). We screened a subset of these in multiple viral assays and found two sets of pharmacological agents that displayed antiviral activity: inhibitors of mRNA translation and predicted regulators of the sigma-1 and sigma-2 receptors. -

Enriched Alternative Splicing in Islets of Diabetes-Susceptible Mice
International Journal of Molecular Sciences Article Enriched Alternative Splicing in Islets of Diabetes-Susceptible Mice Ilka Wilhelmi 1,2, Alexander Neumann 3 , Markus Jähnert 1,2 , Meriem Ouni 1,2,† and Annette Schürmann 1,2,4,5,*,† 1 Department of Experimental Diabetology, German Institute of Human Nutrition (DIfE), 14558 Potsdam, Germany; [email protected] (I.W.); [email protected] (M.J.); [email protected] (M.O.) 2 German Center for Diabetes Research (DZD), 85764 Munich, Germany 3 Omiqa Bioinformatics, 14195 Berlin, Germany; [email protected] 4 Institute of Nutritional Sciences, University of Potsdam, 14558 Nuthetal, Germany 5 Faculty of Health Sciences, Joint Faculty of the Brandenburg University of Technology Cottbus-Senftenberg, The Brandenburg Medical School Theodor Fontane and The University of Potsdam, 14469 Potsdam, Germany * Correspondence: [email protected] † These authors contributed equally to this work. Abstract: Dysfunctional islets of Langerhans are a hallmark of type 2 diabetes (T2D). We hypoth- esize that differences in islet gene expression alternative splicing which can contribute to altered protein function also participate in islet dysfunction. RNA sequencing (RNAseq) data from islets of obese diabetes-resistant and diabetes-susceptible mice were analyzed for alternative splicing and its putative genetic and epigenetic modulators. We focused on the expression levels of chromatin modifiers and SNPs in regulatory sequences. We identified alternative splicing events in islets of diabetes-susceptible mice amongst others in genes linked to insulin secretion, endocytosis or Citation: Wilhelmi, I.; Neumann, A.; ubiquitin-mediated proteolysis pathways. The expression pattern of 54 histones and chromatin Jähnert, M.; Ouni, M.; Schürmann, A. -

Disruption of Nucleocytoplasmic Trafficking As a Cellular Senescence
www.nature.com/emm ARTICLE OPEN Disruption of nucleocytoplasmic trafficking as a cellular senescence driver Ji-Hwan Park1,14, Sung Jin Ryu2,13,14, Byung Ju Kim3,13,14, Hyun-Ji Cho3, Chi Hyun Park4, Hyo Jei Claudia Choi2, Eun-Jin Jang3, Eun Jae Yang5, Jeong-A Hwang5, Seung-Hwa Woo5, Jun Hyung Lee5, Ji Hwan Park5, Kyung-Mi Choi6, Young-Yon Kwon6, 6 7 3 3 8 9 10 5 ✉ Cheol-Koo Lee , Joon✉ Tae Park , Sung✉ Chun Cho , Yun-Il Lee , Sung✉ Bae Lee , Jeong A. Han , Kyung A. Cho , Min-Sik Kim , Daehee Hwang11 , Young-Sam Lee3,5 and Sang Chul Park3,12 © The Author(s) 2021 Senescent cells exhibit a reduced response to intrinsic and extrinsic stimuli. This diminished reaction may be explained by the disrupted transmission of nuclear signals. However, this hypothesis requires more evidence before it can be accepted as a mechanism of cellular senescence. A proteomic analysis of the cytoplasmic and nuclear fractions obtained from young and senescent cells revealed disruption of nucleocytoplasmic trafficking (NCT) as an essential feature of replicative senescence (RS) at the global level. Blocking NCT either chemically or genetically induced the acquisition of an RS-like senescence phenotype, named nuclear barrier-induced senescence (NBIS). A transcriptome analysis revealed that, among various types of cellular senescence, NBIS exhibited a gene expression pattern most similar to that of RS. Core proteomic and transcriptomic patterns common to both RS and NBIS included upregulation of the endocytosis-lysosome network and downregulation of NCT in senescent cells, patterns also observed in an aging yeast model. -

Roles of PTBP1 in Alternative Splicing, Glycolysis, and Oncogensis*
122 Zhu et al. / J Zhejiang Univ-Sci B (Biomed & Biotechnol) 2020 21(2):122-136 Journal of Zhejiang University-SCIENCE B (Biomedicine & Biotechnology) ISSN 1673-1581 (Print); ISSN 1862-1783 (Online) www.jzus.zju.edu.cn; www.springerlink.com E-mail: [email protected] Review: Roles of PTBP1 in alternative splicing, glycolysis, and oncogensis* Wei ZHU§, Bo-lun ZHOU§, Li-juan RONG, Li YE, Hong-juan XU, Yao ZHOU, Xue-jun YAN, Wei-dong LIU, Bin ZHU, Lei WANG, Xing-jun JIANG, Cai-ping REN†‡ NHC Key Laboratory of Carcinogenesis (Central South University) and the Key Laboratory of Carcinogenesis and Cancer Invasion of the Chinese Ministry of Education, Cancer Research Institute, Department of Neurosurgery, Xiangya Hospital, Central South University, Changsha 410008, China †E-mail: [email protected] Received July 16, 2019; Revision accepted Oct. 21, 2019; Crosschecked Dec. 17, 2019; Published online Feb. 5, 2020 Abstract: Polypyrimidine tract-binding protein 1 (PTBP1) plays an essential role in splicing and is expressed in almost all cell types in humans, unlike the other proteins of the PTBP family. PTBP1 mediates several cellular processes in certain types of cells, including the growth and differentiation of neuronal cells and activation of immune cells. Its function is regulated by various molecules, including microRNAs (miRNAs), long non-coding RNAs (lncRNAs), and RNA-binding proteins. PTBP1 plays roles in various diseases, particularly in some cancers, including colorectal cancer, renal cell cancer, breast cancer, and glioma. In cancers, it acts mainly as a regulator of glycolysis, apoptosis, prolif- eration, tumorigenesis, invasion, and migration. The role of PTBP1 in cancer has become a popular research topic in recent years, and this research has contributed greatly to the formulation of a useful therapeutic strategy for cancer.