Ptbp2 Represses Adult-Specific Splicing to Regulate the Generation of Neuronal Precursors in the Embryonic Brain
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Polypyrimidine Tract Binding Proteins Are Essential for B Cell Development
bioRxiv preprint doi: https://doi.org/10.1101/769141; this version posted September 14, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Polypyrimidine Tract Binding Proteins are essential for B cell development Authors: Elisa Monzón-Casanova1,2*, Louise S. Matheson1, Kristina Tabbada3, Kathi Zarnack4, Christopher W. J. Smith2 and Martin Turner1* Affiliations: 1Laboratory of Lymphocyte Signalling and Development, The Babraham Institute, Cambridge, UK; 2Department of Biochemistry, University of Cambridge, Cambridge, UK; 3Next Generation Sequencing Facility, The Babraham Institute, Cambridge, UK; 4Buchmann Institute for Molecular Life Sciences, Goethe University Frankfurt, Frankfurt am Main, Germany *Corresponding authors: Elisa Monzón-Casanova ([email protected]) and Martin Turner ([email protected]) 1 bioRxiv preprint doi: https://doi.org/10.1101/769141; this version posted September 14, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Abstract During B cell development, recombination of immunoglobulin loci is tightly coordinated with the cell cycle to avoid unwanted rearrangements of other genomic locations. Several factors have been identified that suppress proliferation in late-pre-B cells to allow light chain recombination. By comparison, our knowledge of factors limiting proliferation during heavy chain recombination at the pro-B cell stage is very limited. -
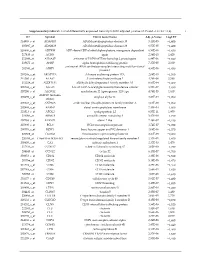
Supplementary Table S1. List of Differentially Expressed
Supplementary table S1. List of differentially expressed transcripts (FDR adjusted p‐value < 0.05 and −1.4 ≤ FC ≥1.4). 1 ID Symbol Entrez Gene Name Adj. p‐Value Log2 FC 214895_s_at ADAM10 ADAM metallopeptidase domain 10 3,11E‐05 −1,400 205997_at ADAM28 ADAM metallopeptidase domain 28 6,57E‐05 −1,400 220606_s_at ADPRM ADP‐ribose/CDP‐alcohol diphosphatase, manganese dependent 6,50E‐06 −1,430 217410_at AGRN agrin 2,34E‐10 1,420 212980_at AHSA2P activator of HSP90 ATPase homolog 2, pseudogene 6,44E‐06 −1,920 219672_at AHSP alpha hemoglobin stabilizing protein 7,27E‐05 2,330 aminoacyl tRNA synthetase complex interacting multifunctional 202541_at AIMP1 4,91E‐06 −1,830 protein 1 210269_s_at AKAP17A A‐kinase anchoring protein 17A 2,64E‐10 −1,560 211560_s_at ALAS2 5ʹ‐aminolevulinate synthase 2 4,28E‐06 3,560 212224_at ALDH1A1 aldehyde dehydrogenase 1 family member A1 8,93E‐04 −1,400 205583_s_at ALG13 ALG13 UDP‐N‐acetylglucosaminyltransferase subunit 9,50E‐07 −1,430 207206_s_at ALOX12 arachidonate 12‐lipoxygenase, 12S type 4,76E‐05 1,630 AMY1C (includes 208498_s_at amylase alpha 1C 3,83E‐05 −1,700 others) 201043_s_at ANP32A acidic nuclear phosphoprotein 32 family member A 5,61E‐09 −1,760 202888_s_at ANPEP alanyl aminopeptidase, membrane 7,40E‐04 −1,600 221013_s_at APOL2 apolipoprotein L2 6,57E‐11 1,600 219094_at ARMC8 armadillo repeat containing 8 3,47E‐08 −1,710 207798_s_at ATXN2L ataxin 2 like 2,16E‐07 −1,410 215990_s_at BCL6 BCL6 transcription repressor 1,74E‐07 −1,700 200776_s_at BZW1 basic leucine zipper and W2 domains 1 1,09E‐06 −1,570 222309_at -

The Splicing Regulator PTBP1 Controls the Activity of the Transcription Factor
1 The splicing regulator PTBP1 controls the activity of the 2 transcription factor Pbx1 during neuronal differentiation. 3 4 Anthony J Linares1, Chia-Ho Lin2, Andrey Damianov2, Katrina L Adams1,3,4, Bennett G 5 Novitch3,4, Douglas L Black2,4 6 1Molecular Biology Institute Graduate Program, University of California, Los Angeles; 2Department of 7 Microbiology, Immunology and Molecular Genetics, University of California, Los Angeles; 3Department of 8 Neurobiology, David Geffen School of Medicine at UCLA; 4Eli and Edythe Broad Center of Regenerative 9 Medicine and Stem Cell Research, David Geffen School of Medicine at UCLA, Los Angeles, United States. 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 1 27 ABSTRACT 28 29 The RNA-binding proteins PTBP1 and PTBP2 control programs of alternative splicing 30 during neuronal development. PTBP2 was found to maintain embryonic splicing patterns 31 of many synaptic and cytoskeletal proteins during differentiation of neuronal progenitor 32 cells (NPCs) into early neurons. However, the role of the earlier PTBP1 program in 33 embryonic stem cells (ESCs) and NPCs was not clear. We show that PTBP1 controls a 34 program of neuronal gene expression that includes the transcription factor Pbx1. We 35 identify exons specifically regulated by PTBP1 and not PTBP2 as mouse ESCs 36 differentiate into NPCs. We find that PTBP1 represses Pbx1 exon 7 and the expression 37 of the neuronal Pbx1a isoform in ESCs. Using CRISPR-Cas9 to delete regulatory 38 elements for exon 7, we induce Pbx1a expression in ESCs, finding that this activates 39 transcription of neuronal genes. -

A SARS-Cov-2 Protein Interaction Map Reveals Targets for Drug Repurposing
Article A SARS-CoV-2 protein interaction map reveals targets for drug repurposing https://doi.org/10.1038/s41586-020-2286-9 A list of authors and affiliations appears at the end of the paper Received: 23 March 2020 Accepted: 22 April 2020 A newly described coronavirus named severe acute respiratory syndrome Published online: 30 April 2020 coronavirus 2 (SARS-CoV-2), which is the causative agent of coronavirus disease 2019 (COVID-19), has infected over 2.3 million people, led to the death of more than Check for updates 160,000 individuals and caused worldwide social and economic disruption1,2. There are no antiviral drugs with proven clinical efcacy for the treatment of COVID-19, nor are there any vaccines that prevent infection with SARS-CoV-2, and eforts to develop drugs and vaccines are hampered by the limited knowledge of the molecular details of how SARS-CoV-2 infects cells. Here we cloned, tagged and expressed 26 of the 29 SARS-CoV-2 proteins in human cells and identifed the human proteins that physically associated with each of the SARS-CoV-2 proteins using afnity-purifcation mass spectrometry, identifying 332 high-confdence protein–protein interactions between SARS-CoV-2 and human proteins. Among these, we identify 66 druggable human proteins or host factors targeted by 69 compounds (of which, 29 drugs are approved by the US Food and Drug Administration, 12 are in clinical trials and 28 are preclinical compounds). We screened a subset of these in multiple viral assays and found two sets of pharmacological agents that displayed antiviral activity: inhibitors of mRNA translation and predicted regulators of the sigma-1 and sigma-2 receptors. -

Enriched Alternative Splicing in Islets of Diabetes-Susceptible Mice
International Journal of Molecular Sciences Article Enriched Alternative Splicing in Islets of Diabetes-Susceptible Mice Ilka Wilhelmi 1,2, Alexander Neumann 3 , Markus Jähnert 1,2 , Meriem Ouni 1,2,† and Annette Schürmann 1,2,4,5,*,† 1 Department of Experimental Diabetology, German Institute of Human Nutrition (DIfE), 14558 Potsdam, Germany; [email protected] (I.W.); [email protected] (M.J.); [email protected] (M.O.) 2 German Center for Diabetes Research (DZD), 85764 Munich, Germany 3 Omiqa Bioinformatics, 14195 Berlin, Germany; [email protected] 4 Institute of Nutritional Sciences, University of Potsdam, 14558 Nuthetal, Germany 5 Faculty of Health Sciences, Joint Faculty of the Brandenburg University of Technology Cottbus-Senftenberg, The Brandenburg Medical School Theodor Fontane and The University of Potsdam, 14469 Potsdam, Germany * Correspondence: [email protected] † These authors contributed equally to this work. Abstract: Dysfunctional islets of Langerhans are a hallmark of type 2 diabetes (T2D). We hypoth- esize that differences in islet gene expression alternative splicing which can contribute to altered protein function also participate in islet dysfunction. RNA sequencing (RNAseq) data from islets of obese diabetes-resistant and diabetes-susceptible mice were analyzed for alternative splicing and its putative genetic and epigenetic modulators. We focused on the expression levels of chromatin modifiers and SNPs in regulatory sequences. We identified alternative splicing events in islets of diabetes-susceptible mice amongst others in genes linked to insulin secretion, endocytosis or Citation: Wilhelmi, I.; Neumann, A.; ubiquitin-mediated proteolysis pathways. The expression pattern of 54 histones and chromatin Jähnert, M.; Ouni, M.; Schürmann, A. -

The Emerging Role of the RBM20 and PTBP1 Ribonucleoproteins in Heart Development and Cardiovascular Diseases
G C A T T A C G G C A T genes Review The Emerging Role of the RBM20 and PTBP1 Ribonucleoproteins in Heart Development and Cardiovascular Diseases Stefania Fochi, Pamela Lorenzi, Marilisa Galasso, Chiara Stefani , Elisabetta Trabetti, Donato Zipeto and Maria Grazia Romanelli * Department of Neurosciences, Biomedicine and Movement Sciences, Section of Biology and Genetics, University of Verona, 37134 Verona, Italy; [email protected] (S.F.); [email protected] (P.L.); [email protected] (M.G.); [email protected] (C.S.); [email protected] (E.T.); [email protected] (D.Z.) * Correspondence: [email protected]; Tel.: +39-045-802-7182 Received: 9 March 2020; Accepted: 6 April 2020; Published: 8 April 2020 Abstract: Alternative splicing is a regulatory mechanism essential for cell differentiation and tissue organization. More than 90% of human genes are regulated by alternative splicing events, which participate in cell fate determination. The general mechanisms of splicing events are well known, whereas only recently have deep-sequencing, high throughput analyses and animal models provided novel information on the network of functionally coordinated, tissue-specific, alternatively spliced exons. Heart development and cardiac tissue differentiation require thoroughly regulated splicing events. The ribonucleoprotein RBM20 is a key regulator of the alternative splicing events required for functional and structural heart properties, such as the expression of TTN isoforms. Recently, the polypyrimidine tract-binding protein PTBP1 has been demonstrated to participate with RBM20 in regulating splicing events. In this review, we summarize the updated knowledge relative to RBM20 and PTBP1 structure and molecular function; their role in alternative splicing mechanisms involved in the heart development and function; RBM20 mutations associated with idiopathic dilated cardiovascular disease (DCM); and the consequences of RBM20-altered expression or dysfunction. -

PTBP2 Exon 10 Inclusion Is Associated with the Progression of CML and It Is BCR-ABL1 Dependent T
International Journal of Biochemistry and Cell Biology 109 (2019) 69–75 Contents lists available at ScienceDirect International Journal of Biochemistry and Cell Biology journal homepage: www.elsevier.com/locate/biocel PTBP2 exon 10 inclusion is associated with the progression of CML and it is BCR-ABL1 dependent T S. Rajashree Nandagopalana, Subramaniam Agatheeswarana,1, Yellamandayya Vadlamudia, ⁎ Sutapa Biswasb, Ghanshyam Biswasb, Naresh Chandra Pattnayakc,2, Soumen Chakrabortya, a Institute of Life Sciences, Nalco Square, Bhubaneswar 751023, India b Sparsh Hospital and Critical Care, Bhubaneswar 751007, India c Lab Care and Diagnostics, Cuttack 753007, India ARTICLE INFO ABSTRACT Keywords: Altered or aberrant expression of several splicing factors leads to the progression of different cancers. Though Alternative splicing there are several ongoing studies underscoring the role of the splicing regulator polypyrimidine tract binding CML protein 2 (PTBP2) in neuronal cells, we unveil the role of PTBP2 in chronic myeloid leukemia (CML). Different RNA binding proteins RNA binding proteins (RBP's) earlier reported in chronic myeloid leukemia blast crisis (CML-BC) cases (n = 28) BCR-ABL1 from Radich Oncomine leukemia dataset, were compared. We observed increased expression of MSI2 followed PTBP2 by PTBP2 in BC cases and increased PTBP2 expression in relapsed cases (n = 10) from the same dataset com- Imatinib pared to other RBPs. We also observed increased PTBP2 exon 10 inclusion in KCL22, a granulocytic lineage CML cell line when compared to other CML cell lines of different lineages. As PTBP2 protein expression is associated with PTBP2 exon 10 inclusion, we observed in cell lines and in a set of progressed cases (n = 4) that increased BCR-ABL1 expression potentiates PTBP2 exon 10 inclusion and thus confers the existence of a functional protein. -

The Splicing Regulator PTBP2 Controls a Program of Embryonic Splicing Required for Neuronal Maturation
Faculty & Staff Scholarship 2014 The splicing regulator PTBP2 controls a program of embryonic splicing required for neuronal maturation Qin Li University of California, Los Angeles Sika Zhaeng University of California, Los Angeles Areum Han University of California, Los Angeles Chia-Ho Lin University of California, Los Angeles Peter Stoilov West Virginia University See next page for additional authors Follow this and additional works at: https://researchrepository.wvu.edu/faculty_publications Digital Commons Citation Li, Qin; Zhaeng, Sika; Han, Areum; Lin, Chia-Ho; Stoilov, Peter; Fu, Xiang-Dong; and Black, Douglas L., "The splicing regulator PTBP2 controls a program of embryonic splicing required for neuronal maturation" (2014). Faculty & Staff Scholarship. 2599. https://researchrepository.wvu.edu/faculty_publications/2599 This Article is brought to you for free and open access by The Research Repository @ WVU. It has been accepted for inclusion in Faculty & Staff Scholarship by an authorized administrator of The Research Repository @ WVU. For more information, please contact [email protected]. Authors Qin Li, Sika Zhaeng, Areum Han, Chia-Ho Lin, Peter Stoilov, Xiang-Dong Fu, and Douglas L. Black This article is available at The Research Repository @ WVU: https://researchrepository.wvu.edu/faculty_publications/ 2599 RESEARCH ARTICLE elife.elifesciences.org The splicing regulator PTBP2 controls a program of embryonic splicing required for neuronal maturation Qin Li1, Sika Zheng1, Areum Han2, Chia-Ho Lin1, Peter Stoilov3, Xiang-Dong -

PTBP2-Dependent Alternative Splicing Regulates Protein Transport and Mitochondria
bioRxiv preprint doi: https://doi.org/10.1101/497461; this version posted December 15, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. PTBP2-dependent alternative splicing regulates protein transport and mitochondria morphology in post-meiotic germ cells. Molly M. Hannigan1, Hisashi Fujioka2, Adina Brett-Morris3, Jason A. Mears3,4, and Donny D. Licatalosi1,* 1Center for RNA Science and Therapeutics, 2EM Core Facility, 3Department of Pharmacology and Center for Mitochondrial Diseases, 4Cleveland Center for Membrane and Structural Biology, Case Western Reserve University, School of Medicine, Cleveland, Ohio, 44106, USA *Correspondence Donny D. Licatalosi Center for RNA Science and Therapeutics Case Western Reserve University Cleveland, OH, 44106, USA Tel: (216) 368-4757 Fax: (216) 368-2010 Email: [email protected] 1 bioRxiv preprint doi: https://doi.org/10.1101/497461; this version posted December 15, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. ABSTRACT The RNA binding protein PTBP2 (polypyrimidine tract binding protein 2) is a key regulator of tissue-specific alternative RNA splicing. In the testis, PTBP2 is expressed in meiotic and post-meiotic germ cells (spermatocytes and spermatids, respectively). In these cells, PTBP2 is required for proper alternative mRNA splicing for over 200 genes, disproportionately affecting genes encoding proteins involved in protein trafficking via transport vesicles. -

Coordinated Regulation of Neuronal Mrna Steady-State Levels Through Developmentally Controlled Intron Retention
Downloaded from genesdev.cshlp.org on September 24, 2021 - Published by Cold Spring Harbor Laboratory Press Coordinated regulation of neuronal mRNA steady-state levels through developmentally controlled intron retention Karen Yap,1,4 Zhao Qin Lim,1,4 Piyush Khandelia,1,4 Brad Friedman,2,3,5 and Eugene V. Makeyev1,6 1School of Biological Sciences, Nanyang Technological University, Singapore, 637551; 2Department of Molecular and Cell Biology, Harvard University, Cambridge, Massachusetts 02138, USA; 3The Koch Institute for Integrative Cancer Research, Massachusetts Institute of Technology, Cambridge, Massachusetts 02139, USA Differentiated cells acquire unique structural and functional traits through coordinated expression of lineage- specific genes. An extensive battery of genes encoding components of the synaptic transmission machinery and specialized cytoskeletal proteins is activated during neurogenesis, but the underlying regulation is not well understood. Here we show that genes encoding critical presynaptic proteins are transcribed at a detectable level in both neurons and nonneuronal cells. However, in nonneuronal cells, the splicing of 39-terminal introns within these genes is repressed by the polypyrimidine tract-binding protein (Ptbp1). This inhibits the export of incompletely spliced mRNAs to the cytoplasm and triggers their nuclear degradation. Clearance of these intron- containing transcripts occurs independently of the nonsense-mediated decay (NMD) pathway but requires components of the nuclear RNA surveillance machinery, including the nuclear pore-associated protein Tpr and the exosome complex. When Ptbp1 expression decreases during neuronal differentiation, the regulated introns are spliced out, thus allowing the accumulation of translation-competent mRNAs in the cytoplasm. We propose that this mechanism counters ectopic and precocious expression of functionally linked neuron-specific genes and ensures their coherent activation in the appropriate developmental context. -
Supplemental Material
SUPPLEMENTAL MATERIAL Online OnlY Supplemental material miRNA expression profiling of cerebrospinal fluid in patients with cerebral aneurysmal subarachnoid hemorrhage S. S. Stylli et al. http://thejns.org/doi/abs/10.3171/2016.1.JNS151454 DisClaimer The Journal of Neurosurgery acknowledges that the following section is published verbatim as submitted by the authors and did not go through either the Journal’s peer-review or editing process. ©AANS, 2016 J Neurosurg Supplementary Table 1 nSolver Differential Expression Analysis Comparison Groups Comparison 1 No SAH SAH / No vasospasm (combined) CSF0012, CSF0018, CSF0030, CSF0034 CSF0020, CSF0023, CSF0024, CSF0025, CSF0038, CSF0039, CSF0040, CSF0041, CSF0042 Comparison 2 No SAH SAH / No vasospasm (Sample Day 1) CSF0012, CSF0018, CSF0030, CSF0034 CSF0020, CSF0023, CSF0024, CSF0027, CSF0041, CSF0038 Comparison 3 No SAH SAH / Vasospasm (combined) CSF0012, CSF0018, CSF0030, CSF0034 CSF0 022, CSF0027, CSF0032, CSF0033, CSF0036, CSF0044, CSF0045, CSF0048, CSF0052, CSF0053 Comparison 4 No SAH SAH / Vasospasm (Sample Day 1) CSF0012, CSF0018, CSF0030, CSF0034 CSF0027, CSF0032, CSF0036, CSF0044, CSF0052 Comparison 5 No SAH SAH / Vasospasm (post Sample Day 1) CSF0012, CSF0018, CSF0030, CSF0034 CSF0022, CSF0033, CSF0045, CSF0048, CSF0053 Comparison 6 SAH / No vasospasm (combined) SAH / Vasospasm (combined) CSF0020, CSF0023, CSF0024, CSF0025, CSF0022, CSF0027, CSF0032, CSF0033, CSF0036, CSF0038, CSF0039, CSF0040, CSF0041, CSF0044, CSF0045, CSF0048, CSF0052, CSF0053 CSF0042 Comparison 7 SAH / No vasospasm -

1 Title: Networks of Splice Factor Regulation by Unproductive
bioRxiv preprint doi: https://doi.org/10.1101/2020.05.20.107375. this version posted May 22, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. It is made available under a CC-BY 4.0 International license. Title: Networks of Splice Factor Regulation by Unproductive Splicing Coupled With Nonsense Mediated mRNA Decay Anna Desai1#, Zhiqiang Hu2#, Courtney E. French3, James P. B. Lloyd2,4, and Steven E. Brenner1,2,3,4* 1 Comparative Biochemistry Graduate Program, University of California, Berkeley, California 94720, USA 2 Department of Plant and Microbial Biology, University of California, Berkeley, California 94720, USA 3 Department of Molecular & Cell Biology, University of California, Berkeley, California 94720, USA 4 Center for RNA Systems Biology, University of California, Berkeley, California 94720, USA # Co-first authors * Corresponding author E-mail: [email protected] 1 bioRxiv preprint doi: https://doi.org/10.1101/2020.05.20.107375. this version posted May 22, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. It is made available under a CC-BY 4.0 International license. Abstract Background: Nonsense mediated mRNA decay (NMD) is an RNA surveillance pathway that degrades aberrant transcripts harboring premature termination codons. This pathway, in conjunction with alternative splicing, regulates gene expression post-transcriptionally. Nearly all serine and arginine-rich (SR) proteins and many heterogeneous nuclear ribonucleoproteins (hnRNPs) produce isoforms that can be degraded by the NMD pathway. Many splicing factors have been reported to be regulated via alternative splicing coupled to NMD.