Oxidizing and Reducing Agents
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Process for Purification of Ethylene Compound Having Fluorine-Containing Organic Group
Office europeen des brevets (fi) Publication number : 0 506 374 A1 @ EUROPEAN PATENT APPLICATION @ Application number: 92302586.0 @ Int. CI.5: C07C 17/38, C07C 21/18 (g) Date of filing : 25.03.92 (30) Priority : 26.03.91 JP 86090/91 (72) Inventor : Kishita, Hirofumi 3-19-1, Isobe, Annaka-shi Gunma-ken (JP) (43) Date of publication of application : Inventor : Sato, Shinichi 30.09.92 Bulletin 92/40 3-5-5, Isobe, Annaka-shi Gunma-ken (JP) Inventor : Fujii, Hideki (84) Designated Contracting States : 3-12-37, Isobe, Annaka-shi DE FR GB Gunma-ken (JP) Inventor : Matsuda, Takashi 791 ~4 YdndSG Anri3k3~shi @ Applicant : SHIN-ETSU CHEMICAL CO., LTD. Gunma-ken (JP) 6-1, Ohtemachi 2-chome Chiyoda-ku Tokyo (JP) @) Representative : Votier, Sidney David et al CARPMAELS & RANSFORD 43, Bloomsbury Square London WC1A 2RA (GB) (54) Process for purification of ethylene compound having fluorine-containing organic group. (57) A process for purifying an ethylene compound having a fluorine-containing organic group (fluorine- containing ethylene compound) by mixing the fluorine-containing ethylene compound with an alkali metal or alkaline earth metal reducing agent, and subjecting the resulting mixture to irradiation with ultraviolet radiation, followed by washing with water. The purification process ensures effective removal of iodides which are sources of molecular iodine, from the fluorine-containing ethylene compound. < h- CO CO o 10 o Q_ LU Jouve, 18, rue Saint-Denis, 75001 PARIS EP 0 506 374 A1 BACKGROUND OF THE INVENTION 1. Field of the Invention 5 The present invention relates to a process for purifying an ethylene compound having a fluorine-containing organic group, and more particularly to a purification process by which iodine contained as impurity in an ethylene compound having a fluorine-containing organic group can be removed effectively. -
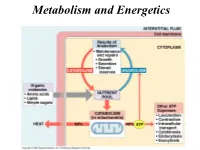
Metabolism and Energetics Oxidation of Carbon Atoms of Glucose Is the Major Source of Energy in Aerobic Metabolism
Metabolism and Energetics Oxidation of carbon atoms of glucose is the major source of energy in aerobic metabolism C6H1206 + 6O2 yields 6 CO2 + H20 + energy Energy released ΔG = - 686 kcal/mol Glucose oxidation requires over 25 discrete steps, with production of 36 ATP. Energy Transformations The mitochondrial synthesis of ATP is not stochiometric. Electron–motive force Proton-motive force Phosphoryl-transfer potential in the form of ATP. Substrate level phosphorylation The formation of ATP by substrate-level phosphorylation ADP ATP P CH O P CH2 O P 2 is used to represent HC OH HC OH a phosphate ester: phosphoglycerate CH OH OH CH2 O P kinase 2 P O bisphosphoglycerate 3-phosphoglycerate OH ADP ATP CH2 CH2 CH3 C O P C OH C O pyruvate kinase non-enzymic COOH COOH COOH phosphoenolpyruvate enolpyruvate pyruvate Why ATP? The reaction of ATP hydrolysis is very favorable ΔGo = - 30.5 kJ/mol = - 7.3 kCal/mol 1. Charge separation of closely packed phosphate groups provides electrostatic relief. Mg2+ 2. Inorganic Pi, the product of the reaction, is immediately resonance-stabilized (electron density spreads equally to all oxygens). 3. ADP immediately ionizes giving H+ into a low [H+] environment (pH~7). 4. Both Pi and ADP are more favorably solvated by water than one ATP molecule. 5. ATP is water soluble. The total body content of ATP and ADP is under 350 mmol – about 10 g, BUT … the amount of ATP synthesized and used each day is about 150 mol – about 110 kg. ATP Production - stage 1 - Glycolysis Glycolysis When rapid production of ATP is needed. -

Elementary Iodine-Doped Activated Carbon As an Oxidizing Agent for the Treatment of Arsenic-Enriched Drinking Water
water Article Elementary Iodine-Doped Activated Carbon as an Oxidizing Agent for the Treatment of Arsenic-Enriched Drinking Water Fabio Spaziani 1,2,*, Yuli Natori 1, Yoshiaki Kinase 1, Tomohiko Kawakami 1 and Katsuyoshi Tatenuma 1 1 KAKEN Inc., Mito Institute 1044 Hori, Mito, Ibaraki 310-0903, Japan 2 ENEA Casaccia, Via Anguillarese 301, 00123 Roma, Italy * Correspondence: [email protected] Received: 18 July 2019; Accepted: 23 August 2019; Published: 27 August 2019 Abstract: An activated carbon impregnated with elementary iodine (I2), named IodAC, characterized by oxidation capability, was developed and applied to oxidize arsenite, As(III), to arsenate, As(V), in arsenic-rich waters. Batch and column experiments were conducted to test the oxidation ability of the material. Comparisons with the oxidizing agents usually used in arsenic treatment systems were also conducted. In addition, the material has been tested coupled with an iron-based arsenic sorbent, in order to verify its suitability for the dearsenication of drinking waters. IodAC exhibited a high and lasting oxidation potential, since the column tests executed on water spiked with 50 mg/L of arsenic (100% arsenite) showed that 1 cc of IodAC (30 wt% I2) can oxidize about 25 mg of As(III) (0.33 mmol) before showing a dwindling in the oxidation ability. Moreover, an improvement of the arsenic sorption capability of the tested sorbent was also proved. The results confirmed that IodAC is suitable for implementation in water dearsenication plants, in place of the commonly used oxidizing agents, such as sodium hypochlorite or potassium permanganate, and in association with arsenic sorbents. -

Reductions and Reducing Agents
REDUCTIONS AND REDUCING AGENTS 1 Reductions and Reducing Agents • Basic definition of reduction: Addition of hydrogen or removal of oxygen • Addition of electrons 9:45 AM 2 Reducible Functional Groups 9:45 AM 3 Categories of Common Reducing Agents 9:45 AM 4 Relative Reactivity of Nucleophiles at the Reducible Functional Groups In the absence of any secondary interactions, the carbonyl compounds exhibit the following order of reactivity at the carbonyl This order may however be reversed in the presence of unique secondary interactions inherent in the molecule; interactions that may 9:45 AM be activated by some property of the reacting partner 5 Common Reducing Agents (Borohydrides) Reduction of Amides to Amines 9:45 AM 6 Common Reducing Agents (Borohydrides) Reduction of Carboxylic Acids to Primary Alcohols O 3 R CO2H + BH3 R O B + 3 H 3 2 Acyloxyborane 9:45 AM 7 Common Reducing Agents (Sodium Borohydride) The reductions with NaBH4 are commonly carried out in EtOH (Serving as a protic solvent) Note that nucleophilic attack occurs from the least hindered face of the 8 carbonyl Common Reducing Agents (Lithium Borohydride) The reductions with LiBH4 are commonly carried out in THF or ether Note that nucleophilic attack occurs from the least hindered face of the 9:45 AM 9 carbonyl. Common Reducing Agents (Borohydrides) The Influence of Metal Cations on Reactivity As a result of the differences in reactivity between sodium borohydride and lithium borohydride, chemoselectivity of reduction can be achieved by a judicious choice of reducing agent. 9:45 AM 10 Common Reducing Agents (Sodium Cyanoborohydride) 9:45 AM 11 Common Reducing Agents (Reductive Amination with Sodium Cyanoborohydride) 9:45 AM 12 Lithium Aluminium Hydride Lithium aluminiumhydride reacts the same way as lithium borohydride. -

Comparative Study of Oxidants in Removal of Chemical Oxygen Demand from the Wastewater (IJIRST/ Volume 4 / Issue 1/ 011)
IJIRST –International Journal for Innovative Research in Science & Technology| Volume 4 | Issue 1 | June 2017 ISSN (online): 2349-6010 Comparative Study of Oxidants in Removal of Chemical Oxygen Demand from the Wastewater Prof Dr K N Sheth Kavita V Italia Director PG Student Geetanjali Institute of Technical Studies, Udaipur Department of Environmental Engineering Institute of Science & Technology for Advanced Studies & Research, Vallabh Vidyanagar Abstract Chemical Oxygen Demand (COD) test involves chemical oxidation using chromic acid as a strong oxidizer. COD of a wastewater sample is the amount of oxidant- potassium dichromate that reacts with the sample when it is heated for 2 hours under controlled environmental conditions and result is expressed as mg of oxidant consumed per liter of a given sample of wastewater. COD can be helpful in determining the quantity required for dilution needed for conducting Biochemical Oxygen Demand (BOD) five day test. In the present investigation attempts have been made to use other oxidants like hydrogen peroxide H2O2, sodium hypo-chlorite, calcium hypo-chlorite and potassium permanganate for measurement of COD by standard method and compare the results obtained using potassium dichromate as strong oxidant. Experiments were carried out for each oxidizing agent for COD removal at temperatures like 1000C, 750C and 500C. The duration of exposure time in each experimental set up was taken as temperature wise, for 1000C, it was 1-5 minutes , for 750C it was 20-40 minutes and for 500C it was 45-75 minutes. It was found that temperature plays very important role in in deciding the exposure time to be allocated for reduction of COD. -

Chapter 20 Electrochemistry
Chapter 20 Electrochemistry Learning goals and key skills: Identify oxidation, reduction, oxidizing agent, and reducing agent in a chemical equation Complete and balance redox equations using the method of half-reactions. Sketch a voltaic cell and identify its cathode, anode, and the directions in which electrons and ions move. o Calculate standard emfs (cell potentials), E cell, from standard reduction potentials. Use reduction potentials to predict whether a redox reaction is spontaneous. o o Relate E cell to DG and equilibrium constants. Calculate emf under nonstandard conditions. Identify the components of common batteries. Describe the construction of a lithium-ion battery and explain how it works. Describe the construction of a fuel cell and explain how it generates electrical energy. Explain how corrosion occurs and how it is prevented by cathodic protection. Describe the reactions in electrolytic cells. Relate the amounts of products and reactants in redox reactions to electrical charge. Electrochemistry Electrochemistry is the study of the relationships between electricity and chemical reactions. • It includes the study of both spontaneous and nonspontaneous processes. 1 Redox reactions: assigning oxidation numbers Oxidation numbers help keep track of what species loses electrons and what species gains them. • An element is oxidized when the oxidation number increases • An element is reduced when the oxidation number decreases • an oxidizing agent causes another element to be oxidized • a reducing agent causes another element to be reduced. Assigning oxidation numbers (sect. 4.4) 1. Elemental form, each atom has ox. # = 0. Zn O2 O3 I2 S8 P4 2. Simple ions, = charge on ion. -1 for Cl-, +2 for Mg2+ 3. -

Ch. 21.1 Redox Reactions and Electrochemical Cells
Pre-Health Post-Baccalaureate Program Study Guide and Practice Problems Course: CHM2046 Textbook Chapter: 21.1 (Silberberg 6e) Topics Covered: Redox Reactions and Electrochemical Cells Created by Isaac Loy 1. Review Understanding this chapter’s material will depend on an in- depth understanding of redox reactions, which were first covered last semester in ch. 4. We will review redox reactions in this study guide, but it would be wise to review ch. 4 if you are having difficulty with this material. Redox reactions will also be incredibly important moving forward into organic chemistry and biochemistry. 2. Oxidation-Reduction Reactions The mnemonic that you will come back to time and time again for this topic is: “LEO the lion says GER” Where “LEO” stands for Loss of Electrons = Oxidation And “GER” stands for Gain of Electrons = Reduction The oxidizing agent (the substance that is being reduced) pulls electrons from the substance that is being oxidized. The reducing agent (the substance that is being oxidized) gives electrons to the substance that is being reduced. Oxidation and reduction are simultaneous processes. In order for a redox reaction to take place, one substance must be oxidized and the other must be reduced. When working with oxidation numbers to solve problems, the substance being oxidized (LEO -> loss of electrons) becomes more positive. Likewise, the substance being reduced (GER -> gaining electrons) becomes more negative. 3. Using Half-Reactions to Solve Redox Problems Follow the steps below to create half-reactions. It is crucial that you follow the steps in order! A. Split up the overall reaction into two half-reactions, where the species of one reaction is being oxidized, and the species of the other reaction is being reduced. -

Gen Chem II Jasperse Ch. 19 Electrochemistry 1
Gen Chem II Jasperse Ch. 19 Electrochemistry 1 Chapter 19 Electrochemistry Math Summary Relating Standard Cell Potential to Standard Half Cell Potentials Eºcell=Eºoxidation + Eºreduction (standard conditions assume 1.0 M concentrations) Relating Half Cell Potentials when Written in Opposite Directions Eºox = -Eºred for half reactions written in opposite directions Relating Standard Cell Potentials to ∆G ∆Gº = -nFE˚cell (to give answer in kJ, use F = 96.485) F = 96,500 C/mol n=number of electrons transferred Relating Actual Cell Potential to Standard Cell Potential when Concentrations aren't 1.0-M Ecell = Eºcell -[0.0592/n] log Q (Q = ratio of actual concentrations) Relating Standard Cell Potential to Equilibrium Constant log K = nEº/0.0592 Relating Actual Cell Potential to Actual Concentrations in Concentration Cells Ecell = -[0.0592/n] log Q for concentration cells, where anode and cathode differ only in concentration, but otherwise have same ions Relating # of Moles of Electrons Transferred as a Function of Time and Current in Electrolysis 1 mol e- = 96,500 C moles of electrons = [current (A)•time (sec)]/96,500 for electrolysis, moles, current, and time are related. rearranged: time (sec)=(moles of electrons)(96500)/current (in A) Note: 3600 sec/hour so time (hours)=(moles of electrons)(26.8)/current (in A) Electrochemistry-Related Units C = Coulomb = quantity of electrical charge = 6.24 • 1018 electrons • 1 mole of electrons = 96,500 C A = amp = rate of charge flow per time = C/sec V = volt = electrical power/force/strength = J/C 96,500C 96.5 kJ F = Faraday = = mole e− mole e− •V € € Gen Chem II Jasperse Ch. -

Chemistry – Midterm Part 3 Material Unit Review Packet 2014 ALLEN
Chemistry – Midterm Part 3 Material Unit Review packet 2014 ALLEN 1. Label the following as chemical or physical change: Grass growing_____physical______ Dissolving sugar in water____ physical___ Evaporating water__physical____ Na in water appears to dissolve and forms NaOH__chemical__ 2. An element is found which is a good conductor of electricity, is ductile, brittle and does not react with acid. Is this new element a metal, nonmetal or metalloid? metalloid 3. The current periodic table is arranged according to increasing____atomic number__. 4. Elements in the same vertical column are in the same group and have similar properties due to similar electron configurations. 5. An element is discovered which bonds to oxygen in a 1:1 ratio. Where would it be placed on the periodic table? ________group 2________ 6. Estimate the boiling point of Kr if the boiling point of argon is -186°C and xenon -112°C. (-186+-112)/2 = -149°C 7. Give the characteristics of each group and label the periodic table with their correct positions: Alkali metals alkaline earth metals noble gases Periods Halogens transitional metals lanthanide and actinide metals Groups or families Group 1 = alkali metals (+1), highly reactive Group 2 = alkaline metals (+2) , highly reactive but less reactive than group 1 D block = transition metals (varied charges), strong color (pigments) Group 7 = halogens, highly reactive Group 8 = noble gases, unreactive Top row of inner transition metals = lanthanide series Bottom row of “ “ “ = actinide series Periods = horizontal Groups/families = vertical 8.) Balance Equations: _2_H2 + __O2 2__H2O 9.) Define Law of Conservation of Matter Matter can be neither created or destroyed during a chemical reaction. -

Evolution of the First Metabolic Cycles
Proc. Natl. Acad. Sci. USA Vol. 87, pp. 200-204, January 1990 Evolution Evolution of the first metabolic cycles (chemoautotrophy/reductive citric acid cycle/origin of life/pyrite) GUNTER WACHTERSHAUSER 8000 Munich 2, Tal 29, Federal Republic of Germany Communicated by Karl Popper, October 12, 1989 (received for review February 28, 1989) ABSTRACT There are two alternatives concerning the genobacter thermophilus (13), and Desulfobacter hydro- origin of life: the origin may be heterotrophic or autotrophic. genophilus (14) and also in the sulfur-associated archaebac- The central problem within the theory of an autotrophic origin teria Thermoproteus neutrophilus (15) and, partly demon- is the first process of carbon fixation. I here propose the strated, in Sulfolobus brierleyi (16). As suggested by Kandler hypothesis that this process is an autocatalytic cycle that can be and Stetter (16) and previously by Hartmann (17), it may be retrodictively constructed from the extant reductive citric acid considered to be of great antiquity and the evolutionary cycle by replacing thioesters by thioacids and by assuming that precursor ofthe oxidative Krebs cycle. It is here conjectured the required reducing power is obtained from the oxidative to be the extant candidate for the reconstruction of the formation of pyrite (FeS2). This archaic cycle is strictly archaic autocatalytic cycle of carbon fixation. chemoautotrophic: photoautotrophy is not required. The cycle The presently accepted form of the extant reductive citric is catalytic for pyrite formation and autocatalytic for its own acid cycle is shown in Fig. 1 in a somewhat unusual repre- multiplication. It is a consequence of this hypothesis that the sentation, twisted in an 8. -

20 More About Oxidation–Reduction Reactions
More About 20 Oxidation–Reduction Reactions OOC n important group of organic reactions consists of those that O A involve the transfer of electrons C from one molecule to another. Organic chemists H OH use these reactions—called oxidation–reduction reactions or redox reactions—to synthesize a large O variety of compounds. Redox reactions are also important C in biological systems because many of these reactions produce HH energy. You have seen a number of oxidation and reduction reactions in other chapters, but discussing them as a group will give you the opportunity to CH3OH compare them. In an oxidation–reduction reaction, one compound loses electrons and one com- pound gains electrons. The compound that loses electrons is oxidized, and the one that gains electrons is reduced. One way to remember the difference between oxidation and reduction is with the phrase “LEO the lion says GER”: Loss of Electrons is Oxi- dation; Gain of Electrons is Reduction. The following is an example of an oxidation–reduction reaction involving inorganic reagents: Cu+ + Fe3+ ¡ Cu2+ + Fe2+ In this reaction,Cu+ loses an electron, so Cu+ is oxidized. Fe3+ gains an electron, so Fe3+ is reduced. The reaction demonstrates two important points about oxidation– reduction reactions. First, oxidation is always coupled with reduction. In other words, a compound cannot gain electrons (be reduced) unless another compound in the reaction simultaneously loses electrons (is oxidized). Second, the compound that is oxidized (Cu+) is called the reducing agent because it loses the electrons that are used to reduce the other compound (Fe3+). Similarly, the compound that is reduced (Fe3+) is called the oxidizing agent because it gains the electrons given up by the other compound (Cu+) when it is oxidized. -

Hydrogen Peroxide Cas #7722-84-1
HYDROGEN PEROXIDE CAS #7722-84-1 Division of Toxicology ToxFAQsTM April 2002 This fact sheet answers the most frequently asked health questions (FAQs) about hydrogen peroxicde. For more information, call the ATSDR Information Center at 1-888-422-8737. This fact sheet is one in a series of summaries about hazardous substances and their health effects. It is important you understand this information because this substance may harm you. The effects of exposure to any hazardous substance depend on the dose, the duration, how you are exposed, personal traits and habits, and whether other chemicals are present. HIGHLIGHTS: Hydrogen peroxide is a manufactured chemical, although small amounts of hydrogen peroxide gas may occur naturally in the air. Low exposure may occur from use at home; higher exposures may occur from industrial use. Exposure to hydrogen peroxide can cause irritation of the eyes, throat, respiratory airway, and skin. Drinking concentrated liquid can cause mild to severe gastrointestinal effects. This substance has been found in at least 18 of the 1,585 National Priorities List sites identified by the Environmental Protection Agency (EPA). What is hydrogen peroxide? ‘ If released to soil, hydrogen peroxide will be broken down Hydrogen peroxide is a colorless liquid at room temperature by reacting with other compounds. with a bitter taste. Small amounts of gaseous hydrogen peroxide occur naturally in the air. Hydrogen peroxide is ‘ Hydrogen peroxide does not accumulate in the food chain. unstable, decomposing readily to oxygen and water with release of heat. Although nonflammable, it is a powerful How might I be exposed to hydrogen peroxide? oxidizing agent that can cause spontaneous combustion when it comes in contact with organic material.