Rapid Review: Post-Vaccination Transmission
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Vaccination Certificate Information Version: 21 July 2021
معلومات عن شهادة التطعيم، بالعربية 中文疫苗接种凭证信息 Informations sur le certificat de vaccination en FRANÇAIS Информация о сертификате вакцинации на РУССКОМ ЯЗЫКЕ Información sobre el Certificado de Vacunación en ESPAÑOL UN SYSTEM-WIDE COVID-19 VACCINATION PROGRAMME VACCINATION CERTIFICATE INFORMATION VERSION: 21 JULY 2021 ABOUT THE UN SYSTEM-WIDE COVID-19 VACCINATION PROGRAMME The UN is committed to ensuring the protection of its personnel. Tasked by the Secretary-General, the Department of Operational Support (DOS) is leading a coordinated, UN-system-wide effort to ensure the availability of vaccine to UN personnel, their dependents and implementing partners. The roll-out of the UN System-wide COVID-19 Vaccination Programme (the “Programme”) for UN personnel will provide a significant boost to the ability of personnel to stay and deliver on the Organization's mandates, to support beneficiaries in the communities they serve, and to contribute to our on-going work to recover better together from the pandemic. Information about the Programme is available at: https://www.un.org/en/coronavirus/vaccination ABOUT THE VACCINATION CERTIFICATE Upon administration of the requisite vaccine dosage, a certificate of vaccination is generated through the Programme’s registration platform (the “Platform”). The certificate generated by the Platform uses a standardized format, similar to the one used by the WHO in its “International Certificate of Vaccination or Prophylaxis” (Yellow) vaccination booklet. Note: This certificate may not dispense with travel restrictions put in place by the country(ies) of destination. As the nature of the pandemic continues to rapidly evolve, it is highly advisable to check the latest travel regulations. -

Global Dynamics of a Vaccination Model for Infectious Diseases with Asymptomatic Carriers
MATHEMATICAL BIOSCIENCES doi:10.3934/mbe.2016019 AND ENGINEERING Volume 13, Number 4, August 2016 pp. 813{840 GLOBAL DYNAMICS OF A VACCINATION MODEL FOR INFECTIOUS DISEASES WITH ASYMPTOMATIC CARRIERS Martin Luther Mann Manyombe1;2 and Joseph Mbang1;2 Department of Mathematics, Faculty of Science University of Yaounde 1, P.O. Box 812 Yaounde, Cameroon 1;2;3; Jean Lubuma and Berge Tsanou ∗ Department of Mathematics and Applied Mathematics University of Pretoria, Pretoria 0002, South Africa (Communicated by Abba Gumel) Abstract. In this paper, an epidemic model is investigated for infectious dis- eases that can be transmitted through both the infectious individuals and the asymptomatic carriers (i.e., infected individuals who are contagious but do not show any disease symptoms). We propose a dose-structured vaccination model with multiple transmission pathways. Based on the range of the explic- itly computed basic reproduction number, we prove the global stability of the disease-free when this threshold number is less or equal to the unity. Moreover, whenever it is greater than one, the existence of the unique endemic equilibrium is shown and its global stability is established for the case where the changes of displaying the disease symptoms are independent of the vulnerable classes. Further, the model is shown to exhibit a transcritical bifurcation with the unit basic reproduction number being the bifurcation parameter. The impacts of the asymptomatic carriers and the effectiveness of vaccination on the disease transmission are discussed through through the local and the global sensitivity analyses of the basic reproduction number. Finally, a case study of hepatitis B virus disease (HBV) is considered, with the numerical simulations presented to support the analytical results. -

Commentary Disease Transmission Dynamics and the Evolution
Proc. Natl. Acad. Sci. USA Vol. 96, pp. 800–801, February 1999 Commentary Disease transmission dynamics and the evolution of antibiotic resistance in hospitals and communal settings Simon A. Levin*† and Viggo Andreasen‡ *Department of Ecology and Evolutionary Biology, Eno Hall, Princeton University, Princeton NJ 08544; and ‡Department of Mathematics, Roskilde University, DK-4000 Roskilde, Denmark Despite the tremendous benefits of antibiotics for dealing with sally, exercising little influence on their host under normal a wide range of pathogens, there is by now little doubt that conditions. Antibiotics are typically introduced to treat the their indiscriminate use has led to the emergence of novel true pathogens, and for them the problem of resistance resistant strains and a frightening new set of threats to public introduces questions concerning the length and intensity of health. Hospitals and other community settings provide an treatment, multidrug strategies, and patient compliance. (See, especially fertile ground for the spread of those types; in for example, refs. 2–4). Commensals provide a different sort particular, the recent emergence and proliferation of bacteria of problem, however. Under normal conditions, they live in resistant to both methicillin and vancomycin has engendered such places as the skin or upper respiratory tract, causing little serious concern, threatening the effectiveness of the last or no harm. On occasion, however, they become translocated available options for treatment of potentially fatal Staphylo- to sites that are normally sterile, such as the blood or lungs, coccus strains. where they may have serious harmful effects (6). Many viru- It seems clear that a considered and comprehensive strategy lence factors, for example in Staphylococcus aureus, are carried for antibiotic use is essential, permitting the intelligent de- on plasmids, and can be exchanged among different strains. -

Valacyclovir Reduced Genital Herpes Transmission in Couples Discordant for Herpes Simplex Virus Type 2 Infection Corey L, Wald A, Patel R, Et Al
146 THERAPEUTICS Evid Based Med: first published as 10.1136/ebm.9.5.146 on 30 September 2004. Downloaded from Valacyclovir reduced genital herpes transmission in couples discordant for herpes simplex virus type 2 infection Corey L, Wald A, Patel R, et al. Once-daily valacyclovir to reduce the risk of transmission of genital herpes. N Engl J Med 2004;350:11–20. Clinical impact ratings GP/FP/Primary care wwwwwwq Infectious diseases wwwwwqq ............................................................................................................................... In heterosexual couples who are serologically discordant for herpes simplex virus type 2 (HSV-2) infection, does once Q daily valacyclovir reduce the sexual transmission of genital herpes? METHODS MAIN RESULTS The table shows the results. Design: randomised placebo controlled trial. CONCLUSION In heterosexual couples discordant for herpes simplex virus type 2 infection, valacyclovir reduced transmission of infection. Allocation: concealed.* Commentary Blinding: blinded {participants, healthcare providers, data revious studies have shown that antiviral drugs can reduce the collectors, data analysts, and outcome assessors}À.* frequency of recurrence and subclinical shedding of viral particles.1 Follow up period: 8 months. P This important, large, well designed trial by Corey et al is the first to show an effect on the transmission of HSV-2 infection to an uninfected partner. In this study, the reduction in transmission was limited; 62 partners had Setting: 96 sites in the US, Canada, Europe, Latin America, and to take the drug daily for 8 months to prevent 1 infection, and 57 partners Australia. had to take the drug daily to prevent overall acquisition of HSV-2 infection. 2 independent predictors were found in addition to the drugs: Participants: 1498 couples >18 years who were women as the susceptible partners (hazard ratio [HR] 3.3) and duration immunocompetent, heterosexual, monogamous, in good health, of HSV-2 infection ,2 years (HR 2.9). -

Potential for Transmission of Antimicrobial Resistance In
Report of the Scientific Committee of the Food Safety Authority of Ireland 2015 Potential for Transmission of Antimicrobial Resistance in the Food Chain Report of the Scientific Committee of the Food Safety Authority of Ireland Potential for Transmission of Antimicrobial Resistance in the Food Chain Published by: Food Safety Authority of Ireland Abbey Court, Lower Abbey St Dublin 1 DO1 W2H4 Tel: +353 1 8171300 Fax: +353 1 8171301 [email protected] www.fsai.ie © FSAI 2015 Applications for reproduction should be made to the FSAI Information Unit ISBN 978-1-910348-01-7 Report of the Scientific Committee of the Food Safety Authority of Ireland Potential for Transmission of Antimicrobial Resistance in the Food Chain CONTENTS FOREWORD 4 ACKNOWLEDGEMENTS 5 ABBREVIATIONS 6 EXECUTIVE SUMMARY 7 CHAPTER 1. INTRODUCTION 10 1.1 The Scale of the Antimicrobial Resistance Problem ...................10 1.2 Human and Animals – The ‘One Health’ Concept.....................11 1.3 The Background to the Antimicrobial Resistance Problem . .11 1.4 Genetic Diversity, Genetic Determinants and Change in Bacteria . 14 1.5 Selection for Antimicrobial Resistance...............................15 1.6 Surveillance of Antimicrobial Resistance in Ireland and Europe . .15 1.7 Prudent Use of Antimicrobials – Definition . 15 1.8 Surveillance of Antimicrobial Use in Ireland and Europe ...............16 1.9 Antimicrobials and the Food Chain . .16 1.10 Antimicrobial-resistant Bacteria and the Food Chain ..................16 1.11 Summary . .17 1.12 Chapter Bibliography..............................................17 CHAPTER 2. TERMS OF REFERENCE 20 2.1 Chapter Bibliography..............................................20 1 of 58 Report of the Scientific Potential for Transmission of Committee of the Food Safety Authority of Ireland Antimicrobial Resistance in the Food Chain CHAPTER 3. -

Zoonotic Diseases Fact Sheet
ZOONOTIC DISEASES FACT SHEET s e ion ecie s n t n p is ms n e e s tio s g s m to a a o u t Rang s p t tme to e th n s n m c a s a ra y a re ho Di P Ge Ho T S Incub F T P Brucella (B. Infected animals Skin or mucous membrane High and protracted (extended) fever. 1-15 weeks Most commonly Antibiotic melitensis, B. (swine, cattle, goats, contact with infected Infection affects bone, heart, reported U.S. combination: abortus, B. suis, B. sheep, dogs) animals, their blood, tissue, gallbladder, kidney, spleen, and laboratory-associated streptomycina, Brucellosis* Bacteria canis ) and other body fluids causes highly disseminated lesions bacterial infection in tetracycline, and and abscess man sulfonamides Salmonella (S. Domestic (dogs, cats, Direct contact as well as Mild gastroenteritiis (diarrhea) to high 6 hours to 3 Fatality rate of 5-10% Antibiotic cholera-suis, S. monkeys, rodents, indirect consumption fever, severe headache, and spleen days combination: enteriditis, S. labor-atory rodents, (eggs, food vehicles using enlargement. May lead to focal chloramphenicol, typhymurium, S. rep-tiles [especially eggs, etc.). Human to infection in any organ or tissue of the neomycin, ampicillin Salmonellosis Bacteria typhi) turtles], chickens and human transmission also body) fish) and herd animals possible (cattle, chickens, pigs) All Shigella species Captive non-human Oral-fecal route Ranges from asymptomatic carrier to Varies by Highly infective. Low Intravenous fluids primates severe bacillary dysentery with high species. 16 number of organisms and electrolytes, fevers, weakness, severe abdominal hours to 7 capable of causing Antibiotics: ampicillin, cramps, prostration, edema of the days. -

Vaccine Hesitancy
WHY CHILDREN WORKSHOP ON IMMUNIZATIONS ARE NOT VACCINATED? VACCINE HESITANCY José Esparza MD, PhD - Adjunct Professor, Institute of Human Virology, University of Maryland School of Medicine, Baltimore, MD, USA - Robert Koch Fellow, Robert Koch Institute, Berlin, Germany - Senior Advisor, Global Virus Network, Baltimore, MD, USA. Formerly: - Bill & Melinda Gates Foundation, Seattle, WA, USA - World Health Organization, Geneva, Switzerland The value of vaccination “The impact of vaccination on the health of the world’s people is hard to exaggerate. With the exception of safe water, no other modality has had such a major effect on mortality reduction and population growth” Stanley Plotkin (2013) VACCINES VAILABLE TO PROTECT AGAINST MORE DISEASES (US) BASIC VACCINES RECOMMENDED BY WHO For all: BCG, hepatitis B, polio, DTP, Hib, Pneumococcal (conjugated), rotavirus, measles, rubella, HPV. For certain regions: Japanese encephalitis, yellow fever, tick-borne encephalitis. For some high-risk populations: typhoid, cholera, meningococcal, hepatitis A, rabies. For certain immunization programs: mumps, influenza Vaccines save millions of lives annually, worldwide WHAT THE WORLD HAS ACHIEVED: 40 YEARS OF INCREASING REACH OF BASIC VACCINES “Bill Gates Chart” 17 M GAVI 5.6 M 4.2 M Today (ca 2015): <5% of children in GAVI countries fully immunised with the 11 WHO- recommended vaccines Seth Berkley (GAVI) The goal: 50% of children in GAVI countries fully immunised by 2020 Seth Berkley (GAVI) The current world immunization efforts are achieving: • Equity between high and low-income countries • Bringing the power of vaccines to even the world’s poorest countries • Reducing morbidity and mortality in developing countries • Eliminating and eradicating disease WHY CHILDREN ARE NOT VACCINATED? •Vaccines are not available •Deficient health care systems •Poverty •Vaccine hesitancy (reticencia a la vacunacion) VACCINE HESITANCE: WHO DEFINITION “Vaccine hesitancy refers to delay in acceptance or refusal of vaccines despite availability of vaccination services. -
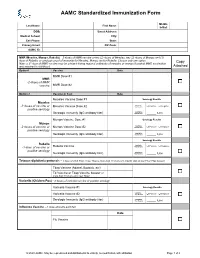
AAMC Standardized Immunization Form
AAMC Standardized Immunization Form Middle Last Name: First Name: Initial: DOB: Street Address: Medical School: City: Cell Phone: State: Primary Email: ZIP Code: AAMC ID: MMR (Measles, Mumps, Rubella) – 2 doses of MMR vaccine or two (2) doses of Measles, two (2) doses of Mumps and (1) dose of Rubella; or serologic proof of immunity for Measles, Mumps and/or Rubella. Choose only one option. Copy Note: a 3rd dose of MMR vaccine may be advised during regional outbreaks of measles or mumps if original MMR vaccination was received in childhood. Attached Option1 Vaccine Date MMR Dose #1 MMR -2 doses of MMR vaccine MMR Dose #2 Option 2 Vaccine or Test Date Measles Vaccine Dose #1 Serology Results Measles Qualitative -2 doses of vaccine or Measles Vaccine Dose #2 Titer Results: Positive Negative positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Mumps Vaccine Dose #1 Serology Results Mumps Qualitative -2 doses of vaccine or Mumps Vaccine Dose #2 Titer Results: Positive Negative positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Serology Results Rubella Qualitative Positive Negative -1 dose of vaccine or Rubella Vaccine Titer Results: positive serology Quantitative Serologic Immunity (IgG antibody titer) Titer Results: _____ IU/ml Tetanus-diphtheria-pertussis – 1 dose of adult Tdap; if last Tdap is more than 10 years old, provide date of last Td or Tdap booster Tdap Vaccine (Adacel, Boostrix, etc) Td Vaccine or Tdap Vaccine booster (if more than 10 years since last Tdap) Varicella (Chicken Pox) - 2 doses of varicella vaccine or positive serology Varicella Vaccine #1 Serology Results Qualitative Varicella Vaccine #2 Titer Results: Positive Negative Serologic Immunity (IgG antibody titer) Quantitative Titer Results: _____ IU/ml Influenza Vaccine --1 dose annually each fall Date Flu Vaccine © 2020 AAMC. -

Evolution of Disease Transmission During the COVID-19
www.nature.com/scientificreports OPEN Evolution of disease transmission during the COVID‑19 pandemic: patterns and determinants Jie Zhu & Blanca Gallego* Epidemic models are being used by governments to inform public health strategies to reduce the spread of SARS‑CoV‑2. They simulate potential scenarios by manipulating model parameters that control processes of disease transmission and recovery. However, the validity of these parameters is challenged by the uncertainty of the impact of public health interventions on disease transmission, and the forecasting accuracy of these models is rarely investigated during an outbreak. We ftted a stochastic transmission model on reported cases, recoveries and deaths associated with SARS‑CoV‑2 infection across 101 countries. The dynamics of disease transmission was represented in terms of the daily efective reproduction number ( Rt ). The relationship between public health interventions and Rt was explored, frstly using a hierarchical clustering algorithm on initial Rt patterns, and secondly computing the time‑lagged cross correlation among the daily number of policies implemented, Rt , and daily incidence counts in subsequent months. The impact of updating Rt every time a prediction is made on the forecasting accuracy of the model was investigated. We identifed 5 groups of countries with distinct transmission patterns during the frst 6 months of the pandemic. Early adoption of social distancing measures and a shorter gap between interventions were associated with a reduction on the duration of outbreaks. The lagged correlation analysis revealed that increased policy volume was associated with lower future Rt (75 days lag), while a lower Rt was associated with lower future policy volume (102 days lag). -

Chapter 2 Disease and Disease Transmission
DISEASE AND DISEASE TRANSMISSION Chapter 2 Disease and disease transmission An enormous variety of organisms exist, including some which can survive and even develop in the body of people or animals. If the organism can cause infection, it is an infectious agent. In this manual infectious agents which cause infection and illness are called pathogens. Diseases caused by pathogens, or the toxins they produce, are communicable or infectious diseases (45). In this manual these will be called disease and infection. This chapter presents the transmission cycle of disease with its different elements, and categorises the different infections related to WES. 2.1 Introduction to the transmission cycle of disease To be able to persist or live on, pathogens must be able to leave an infected host, survive transmission in the environment, enter a susceptible person or animal, and develop and/or multiply in the newly infected host. The transmission of pathogens from current to future host follows a repeating cycle. This cycle can be simple, with a direct transmission from current to future host, or complex, where transmission occurs through (multiple) intermediate hosts or vectors. This cycle is called the transmission cycle of disease, or transmission cycle. The transmission cycle has different elements: The pathogen: the organism causing the infection The host: the infected person or animal ‘carrying’ the pathogen The exit: the method the pathogen uses to leave the body of the host Transmission: how the pathogen is transferred from host to susceptible person or animal, which can include developmental stages in the environment, in intermediate hosts, or in vectors 7 CONTROLLING AND PREVENTING DISEASE The environment: the environment in which transmission of the pathogen takes place. -

Statements Contained in This Release As the Result of New Information Or Future Events Or Developments
Pfizer and BioNTech Provide Update on Booster Program in Light of the Delta-Variant NEW YORK and MAINZ, GERMANY, July 8, 2021 — As part of Pfizer’s and BioNTech’s continued efforts to stay ahead of the virus causing COVID-19 and circulating mutations, the companies are providing an update on their comprehensive booster strategy. Pfizer and BioNTech have seen encouraging data in the ongoing booster trial of a third dose of the current BNT162b2 vaccine. Initial data from the study demonstrate that a booster dose given 6 months after the second dose has a consistent tolerability profile while eliciting high neutralization titers against the wild type and the Beta variant, which are 5 to 10 times higher than after two primary doses. The companies expect to publish more definitive data soon as well as in a peer-reviewed journal and plan to submit the data to the FDA, EMA and other regulatory authorities in the coming weeks. In addition, data from a recent Nature paper demonstrate that immune sera obtained shortly after dose 2 of the primary two dose series of BNT162b2 have strong neutralization titers against the Delta variant (B.1.617.2 lineage) in laboratory tests. The companies anticipate that a third dose will boost those antibody titers even higher, similar to how the third dose performs for the Beta variant (B.1.351). Pfizer and BioNTech are conducting preclinical and clinical tests to confirm this hypothesis. While Pfizer and BioNTech believe a third dose of BNT162b2 has the potential to preserve the highest levels of protective efficacy against all currently known variants including Delta, the companies are remaining vigilant and are developing an updated version of the Pfizer-BioNTech COVID-19 vaccine that targets the full spike protein of the Delta variant. -

COVID-19 Vaccines Frequently Asked Questions
Page 1 of 12 COVID-19 Vaccines 2020a Frequently Asked Questions Michigan.gov/Coronavirus The information in this document will change frequently as we learn more about COVID-19 vaccines. There is a lot we are learning as the pandemic and COVID-19 vaccines evolve. The approach in Michigan will adapt as we learn more. September 29, 2021. Quick Links What’s new | Why COVID-19 vaccination is important | Booster and additional doses | What to expect when you get vaccinated | Safety of the vaccine | Vaccine distribution/prioritization | Additional vaccine information | Protecting your privacy | Where can I get more information? What’s new − Pfizer booster doses recommended for some people to boost waning immunity six months after completing the Pfizer vaccine. Why COVID-19 vaccination is important − If you are fully vaccinated, you don’t have to quarantine after being exposed to COVID-19, as long as you don’t have symptoms. This means missing less work, school, sports and other activities. − COVID-19 vaccination is the safest way to build protection. COVID-19 is still a threat, especially to people who are unvaccinated. Some people who get COVID-19 can become severely ill, which could result in hospitalization, and some people have ongoing health problems several weeks or even longer after getting infected. Even people who did not have symptoms when they were infected can have these ongoing health problems. − After you are fully vaccinated for COVID-19, you can resume many activities that you did before the pandemic. CDC recommends that fully vaccinated people wear a mask in public indoor settings if they are in an area of substantial or high transmission.