Levels of Evidence
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
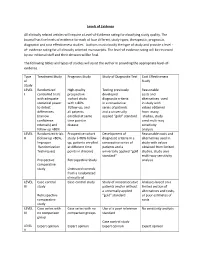
Levels of Evidence Table
Levels of Evidence All clinically related articles will require a Level-of-Evidence rating for classifying study quality. The Journal has five levels of evidence for each of four different study types; therapeutic, prognostic, diagnostic and cost effectiveness studies. Authors must classify the type of study and provide a level - of- evidence rating for all clinically oriented manuscripts. The level-of evidence rating will be reviewed by our editorial staff and their decision will be final. The following tables and types of studies will assist the author in providing the appropriate level-of- evidence. Type Treatment Study Prognosis Study Study of Diagnostic Test Cost Effectiveness of Study Study LEVEL Randomized High-quality Testing previously Reasonable I controlled trials prospective developed costs and with adequate cohort study diagnostic criteria alternatives used statistical power with > 80% in a consecutive in study with to detect follow-up, and series of patients values obtained differences all patients and a universally from many (narrow enrolled at same applied “gold” standard studies, study confidence time point in used multi-way intervals) and disease sensitivity follow up >80% analysis LEVEL Randomized trials Prospective cohort Development of Reasonable costs and II (follow up <80%, study (<80% follow- diagnostic criteria in a alternatives used in Improper up, patients enrolled consecutive series of study with values Randomization at different time patients and a obtained from limited Techniques) points in disease) universally -

TUTORIAL in BIOSTATISTICS: the Self-Controlled Case Series Method
STATISTICS IN MEDICINE Statist. Med. 2005; 0:1–31 Prepared using simauth.cls [Version: 2002/09/18 v1.11] TUTORIAL IN BIOSTATISTICS: The self-controlled case series method Heather J. Whitaker1, C. Paddy Farrington1, Bart Spiessens2 and Patrick Musonda1 1 Department of Statistics, The Open University, Milton Keynes, MK7 6AA, UK. 2 GlaxoSmithKline Biologicals, Rue de l’Institut 89, B-1330 Rixensart, Belgium. SUMMARY The self-controlled case series method was developed to investigate associations between acute outcomes and transient exposures, using only data on cases, that is, on individuals who have experienced the outcome of interest. Inference is within individuals, and hence fixed covariates effects are implicitly controlled for within a proportional incidence framework. We describe the origins, assumptions, limitations, and uses of the method. The rationale for the model and the derivation of the likelihood are explained in detail using a worked example on vaccine safety. Code for fitting the model in the statistical package STATA is described. Two further vaccine safety data sets are used to illustrate a range of modelling issues and extensions of the basic model. Some brief pointers on the design of case series studies are provided. The data sets, STATA code, and further implementation details in SAS, GENSTAT and GLIM are available from an associated website. key words: case series; conditional likelihood; control; epidemiology; modelling; proportional incidence Copyright c 2005 John Wiley & Sons, Ltd. 1. Introduction The self-controlled case series method, or case series method for short, provides an alternative to more established cohort or case-control methods for investigating the association between a time-varying exposure and an outcome event. -

Supplementary File
Supplementary file Table S1. Characteristics of studies included in the systematic review. Authors, reference Type of article Number of patients Type of stroke Valderrama [1] case report 1 AIS Oxley [2] case series 5 AIS Morassi [3] case series 6 AIS (n = 4), HS (n = 2) Tunc [4] case series 4 AIS Avula [5] case series 4 AIS (n = 3), TIA (n = 1) Oliver [6] case report 1 AIS Beyrouti [7] case series 6 AIS Al Saiegh [8] case series 2 AIS (n = 1), SAH (n = 1) Gunasekaran [9] case report 1 AIS Goldberg [10] case report 1 AIS Viguier [11] case report 1 AIS Escalard [12] case series/ case control 10 AIS Wang [13] case series 5 AIS Fara [14] case series 3 AIS Moshayedi [15] case report 1 AIS Deliwala [16] case report 1 AIS Sharafi-Razavi [17] case report 1 HS retrospective cohort study (case Jain [18] 35 AIS (n = 26) HS (n = 9) control) retrospective cohort study (case Yaghi [19] 32 AIS control) AIS (n = 35), TIA (n = 5), HS Benussi [20] retrospective cohort study 43 (n = 3) Rudilosso [21] case report 1 AIS Lodigiani [22] retrospective cohort study 9 AIS Malentacchi [23] case report 1 AIS J. Clin. Med. 2020, 9, x; doi: FOR PEER REVIEW www.mdpi.com/journal/jcm J. Clin. Med. 2020, 9, x FOR PEER REVIEW 2 of 8 retrospective cohort study/case Scullen [24] 2 HS + AIS (n = 1), AIS (n = 1) series retrospective cohort study/ case AIS (n = 17), ST (n = 2), HS Sweid [25] 22 series (n = 3) AIS – acute ischemic stroke; HS – haemorrhagic stroke; SAH – subarachnoid haemorrhage; ST – sinus thrombosis; TIA-transient ischemic attack. -

From Explanatory to Pragmatic Clinical Trials: a New Era for Effectiveness Research
From Explanatory to Pragmatic Clinical Trials: A New Era for Effectiveness Research Jerry Jarvik, M.D., M.P.H. Professor of Radiology, Neurological Surgery and Health Services Adjunct Professor Orthopedic Surgery & Sports Medicine and Pharmacy Director, Comparative Effectiveness, Cost and Outcomes Research Center (CECORC) UTSW April 2018 Acknowledgements •NIH: UH2 AT007766; UH3 AT007766 •NIH P30AR072572 •PCORI: CE-12-11-4469 Disclosures Physiosonix (ultrasound company): Founder/stockholder UpToDate: Section Editor Evidence-Based Neuroimaging Diagnosis and Treatment (Springer): Co-Editor The Big Picture Comparative Effectiveness Evidence Based Practice Health Policy Learning Healthcare System So we need to generate evidence Challenge #1: Clinical research is slow • Tradi>onal RCTs are slow and expensive— and rarely produce findings that are easily put into prac>ce. • In fact, it takes an average of 17 ye ars before research findings Howlead pragmaticto widespread clinical trials canchanges improve in care. practice & policy Challenge #1: Clinical research is slow “…rarely produce findings that are easily put into prac>ce.” Efficacy vs. Effectiveness Efficacy vs. Effectiveness • Efficacy: can it work under ideal conditions • Effectiveness: does it work under real-world conditions Challenge #2: Clinical research is not relevant to practice • Tradi>onal RCTs study efficacy “If we want of txs for carefully selected more evidence- popula>ons under ideal based pr actice, condi>ons. we need more • Difficult to translate to real practice-based evidence.” world. Green, LW. American Journal • When implemented into of Public Health, 2006. How pragmatic clinical trials everyday clinical prac>ce, oZen seecan a “ voltage improve drop practice”— drama>c & decreasepolicy from efficacy to effec>veness. -

Quasi-Experimental Studies in the Fields of Infection Control and Antibiotic Resistance, Ten Years Later: a Systematic Review
HHS Public Access Author manuscript Author ManuscriptAuthor Manuscript Author Infect Control Manuscript Author Hosp Epidemiol Manuscript Author . Author manuscript; available in PMC 2019 November 12. Published in final edited form as: Infect Control Hosp Epidemiol. 2018 February ; 39(2): 170–176. doi:10.1017/ice.2017.296. Quasi-experimental Studies in the Fields of Infection Control and Antibiotic Resistance, Ten Years Later: A Systematic Review Rotana Alsaggaf, MS, Lyndsay M. O’Hara, PhD, MPH, Kristen A. Stafford, PhD, MPH, Surbhi Leekha, MBBS, MPH, Anthony D. Harris, MD, MPH, CDC Prevention Epicenters Program Department of Epidemiology and Public Health, University of Maryland School of Medicine, Baltimore, Maryland. Abstract OBJECTIVE.—A systematic review of quasi-experimental studies in the field of infectious diseases was published in 2005. The aim of this study was to assess improvements in the design and reporting of quasi-experiments 10 years after the initial review. We also aimed to report the statistical methods used to analyze quasi-experimental data. DESIGN.—Systematic review of articles published from January 1, 2013, to December 31, 2014, in 4 major infectious disease journals. METHODS.—Quasi-experimental studies focused on infection control and antibiotic resistance were identified and classified based on 4 criteria: (1) type of quasi-experimental design used, (2) justification of the use of the design, (3) use of correct nomenclature to describe the design, and (4) statistical methods used. RESULTS.—Of 2,600 articles, 173 (7%) featured a quasi-experimental design, compared to 73 of 2,320 articles (3%) in the previous review (P<.01). Moreover, 21 articles (12%) utilized a study design with a control group; 6 (3.5%) justified the use of a quasi-experimental design; and 68 (39%) identified their design using the correct nomenclature. -

Case Series: COVID-19 Infection Causing New-Onset Diabetes Mellitus?
Endocrinology & Metabolism International Journal Case Series Open Access Case series: COVID-19 infection causing new-onset diabetes mellitus? Abstract Volume 9 Issue 1 - 2021 Objectives: This paper seeks to explore the hypothesis of the potential diabetogenic effect 1 2 of SARS-COV-2 (Severe Acute respiratory syndrome coronavirus). Rujuta Katkar, Narasa Raju Madam 1Consultant Endocrinologist at Yuma Regional Medical Center, Case series presentation: We present a case series of observation among 8 patients of age USA 2 group ranging from 34 to 74 years with a BMI range of 26.61 to 53.21 Kilogram/square Hospitalist at Yuma Regional Medical Center, USA meters that developed new-onset diabetes after COVID-19 infection. Correspondence: Rujuta Katkar, MD, Consultant Severe Acute Respiratory Syndrome Coronavirus (SARS-COV-2), commonly known as Endocrinologist at Yuma Regional Medical Center, 2851 S Coronavirus or COVID-19(Coronavirus infectious disease), gains entry into the cells by Avenue B, Bldg.20, Yuma, AZ-85364, USA, Tel 2034359976, binding to the Angiotensin-converting enzyme-2(ACE-2) receptors located in essential Email metabolic tissues including the pancreas, adipose tissue, small intestine, and kidneys. Received: March 25, 2021 | Published: April 02, 2021 The evidence reviewed from the scientific literature describes how ACE 2 receptors play a role in the pathogenesis of diabetes and the plausible interaction of SARS-COV-2 with ACE 2 receptors in metabolic organs and tissues. Conclusion: The 8 patients without a past medical history of diabetes admitted with COVID-19 infection developed new-onset diabetes mellitus due to plausible interaction of SARS-COV-2 with ACE 2 receptors. -

Using Randomized Evaluations to Improve the Efficiency of US Healthcare Delivery
Using Randomized Evaluations to Improve the Efficiency of US Healthcare Delivery Amy Finkelstein Sarah Taubman MIT, J-PAL North America, and NBER MIT, J-PAL North America February 2015 Abstract: Randomized evaluations of interventions that may improve the efficiency of US healthcare delivery are unfortunately rare. Across top journals in medicine, health services research, and economics, less than 20 percent of studies of interventions in US healthcare delivery are randomized. By contrast, about 80 percent of studies of medical interventions are randomized. The tide may be turning toward making randomized evaluations closer to the norm in healthcare delivery, as the relevant actors have an increasing need for rigorous evidence of what interventions work and why. At the same time, the increasing availability of administrative data that enables high-quality, low-cost RCTs and of new sources of funding that support RCTs in healthcare delivery make them easier to conduct. We suggest a number of design choices that can enhance the feasibility and impact of RCTs on US healthcare delivery including: greater reliance on existing administrative data; measuring a wide range of outcomes on healthcare costs, health, and broader economic measures; and designing experiments in a way that sheds light on underlying mechanisms. Finally, we discuss some of the more common concerns raised about the feasibility and desirability of healthcare RCTs and when and how these can be overcome. _____________________________ We are grateful to Innessa Colaiacovo, Lizi Chen, and Belinda Tang for outstanding research assistance, to Katherine Baicker, Mary Ann Bates, Kelly Bidwell, Joe Doyle, Mireille Jacobson, Larry Katz, Adam Sacarny, Jesse Shapiro, and Annetta Zhou for helpful comments, and to the Laura and John Arnold Foundation for financial support. -

Title: a TRANSMISSION-VIRULENCE EVOLUTIONARY TRADE-OFF
1 Title: A TRANSMISSION-VIRULENCE EVOLUTIONARY TRADE-OFF EXPLAINS 2 ATTENUATION OF HIV-1 IN UGANDA 3 Short title: EVOLUTION OF VIRULENCE IN HIV 4 François Blanquart1, Mary Kate Grabowski2, Joshua Herbeck3, Fred Nalugoda4, David 5 Serwadda4,5, Michael A. Eller6, 7, Merlin L. Robb6,7, Ronald Gray2,4, Godfrey Kigozi4, Oliver 6 Laeyendecker8, Katrina A. Lythgoe1,9, Gertrude Nakigozi4, Thomas C. Quinn8, Steven J. 7 Reynolds8, Maria J. Wawer2, Christophe Fraser1 8 1. MRC Centre for Outbreak Analysis and Modelling, Department of Infectious Disease 9 Epidemiology, School of Public Health, Imperial College London, United Kingdom 10 2. Department of Epidemiology, Bloomberg School of Public Health, Johns Hopkins University, 11 Baltimore, MD, USA 12 3. International Clinical Research Center, Department of Global Health, University of 13 Washington, Seattle, WA, USA 14 4. Rakai Health Sciences Program, Entebbe, Uganda 15 5. School of Public Health, Makerere University, Kampala, Uganda 16 6. U.S. Military HIV Research Program, Walter Reed Army Institute of Research, Silver Spring, 17 MD, USA 18 7. Henry M. Jackson Foundation for the Advancement of Military Medicine, Bethesda, MD, USA 19 8. Laboratory of Immunoregulation, Division of Intramural Research, National Institute of 20 Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD, USA 21 9. Department of Zoology, University of Oxford, United Kingdom 22 23 Abstract 24 Evolutionary theory hypothesizes that intermediate virulence maximizes pathogen fitness as 25 a result of a trade-off between virulence and transmission, but empirical evidence remains scarce. 26 We bridge this gap using data from a large and long-standing HIV-1 prospective cohort, in 27 Uganda. -

Catalogue of Clinical Trials and Cohort Studies to Identify Biological
Catalogue of clinical trials and cohort studies to identify biological specimens of relevance to the development of assays for acute and early HIV infection: Final Report Catalogue of clinical trials and cohort studies to identify biological specimens of relevance to the development of assays for recent HIV infection Final Report February 2010 This final report was prepared by Dr Joanne Micallef and Professor John Kaldor, The University of New South Wales, under subcontract with Family Health International, funded by the Bill and Melinda Gates Foundation under the grant titled “Development of Assays for Acute HIV Infection and Estimation and HIV Incidence in Population”. 1 Catalogue of clinical trials and cohort studies to identify biological specimens of relevance to the development of assays for acute and early HIV infection: Final Report Table of Contents Table of Contents .................................................................................................................................................................... 2 List of acronyms ...................................................................................................................................................................... 4 1 Introduction ..................................................................................................................................................................... 6 2 Methods ............................................................................................................................................................................ -

Observational Clinical Research
E REVIEW ARTICLE Clinical Research Methodology 2: Observational Clinical Research Daniel I. Sessler, MD, and Peter B. Imrey, PhD * † Case-control and cohort studies are invaluable research tools and provide the strongest fea- sible research designs for addressing some questions. Case-control studies usually involve retrospective data collection. Cohort studies can involve retrospective, ambidirectional, or prospective data collection. Observational studies are subject to errors attributable to selec- tion bias, confounding, measurement bias, and reverse causation—in addition to errors of chance. Confounding can be statistically controlled to the extent that potential factors are known and accurately measured, but, in practice, bias and unknown confounders usually remain additional potential sources of error, often of unknown magnitude and clinical impact. Causality—the most clinically useful relation between exposure and outcome—can rarely be defnitively determined from observational studies because intentional, controlled manipu- lations of exposures are not involved. In this article, we review several types of observa- tional clinical research: case series, comparative case-control and cohort studies, and hybrid designs in which case-control analyses are performed on selected members of cohorts. We also discuss the analytic issues that arise when groups to be compared in an observational study, such as patients receiving different therapies, are not comparable in other respects. (Anesth Analg 2015;121:1043–51) bservational clinical studies are attractive because Group, and the American Society of Anesthesiologists they are relatively inexpensive and, perhaps more Anesthesia Quality Institute. importantly, can be performed quickly if the required Recent retrospective perioperative studies include data O 1,2 data are already available. -

Interpreting Indirect Treatment Comparisons and Network Meta
VALUE IN HEALTH 14 (2011) 417–428 available at www.sciencedirect.com journal homepage: www.elsevier.com/locate/jval SCIENTIFIC REPORT Interpreting Indirect Treatment Comparisons and Network Meta-Analysis for Health-Care Decision Making: Report of the ISPOR Task Force on Indirect Treatment Comparisons Good Research Practices: Part 1 Jeroen P. Jansen, PhD1,*, Rachael Fleurence, PhD2, Beth Devine, PharmD, MBA, PhD3, Robbin Itzler, PhD4, Annabel Barrett, BSc5, Neil Hawkins, PhD6, Karen Lee, MA7, Cornelis Boersma, PhD, MSc8, Lieven Annemans, PhD9, Joseph C. Cappelleri, PhD, MPH10 1Mapi Values, Boston, MA, USA; 2Oxford Outcomes, Bethesda, MD, USA; 3Pharmaceutical Outcomes Research and Policy Program, School of Pharmacy, School of Medicine, University of Washington, Seattle, WA, USA; 4Merck Research Laboratories, North Wales, PA, USA; 5Eli Lilly and Company Ltd., Windlesham, Surrey, UK; 6Oxford Outcomes Ltd., Oxford, UK; 7Canadian Agency for Drugs and Technologies in Health (CADTH), Ottawa, ON, Canada; 8University of Groningen / HECTA, Groningen, The Netherlands; 9University of Ghent, Ghent, Belgium; 10Pfizer Inc., New London, CT, USA ABSTRACT Evidence-based health-care decision making requires comparisons of all of randomized, controlled trials allow multiple treatment comparisons of relevant competing interventions. In the absence of randomized, con- competing interventions. Next, an introduction to the synthesis of the avail- trolled trials involving a direct comparison of all treatments of interest, able evidence with a focus on terminology, assumptions, validity, and statis- indirect treatment comparisons and network meta-analysis provide use- tical methods is provided, followed by advice on critically reviewing and in- ful evidence for judiciously selecting the best choice(s) of treatment. terpreting an indirect treatment comparison or network meta-analysis to Mixed treatment comparisons, a special case of network meta-analysis, inform decision making. -

Patient Reported Outcomes (PROS) in Performance Measurement
Patient-Reported Outcomes Table of Contents Introduction ............................................................................................................................................ 2 Defining Patient-Reported Outcomes ............................................................................................ 2 How Are PROs Used? ............................................................................................................................ 3 Measuring research study endpoints ........................................................................................................ 3 Monitoring adverse events in clinical research ..................................................................................... 3 Monitoring symptoms, patient satisfaction, and health care performance ................................ 3 Example from the NIH Collaboratory ................................................................................................................... 4 Measuring PROs: Instruments, Item Banks, and Devices ....................................................... 5 PRO Instruments ............................................................................................................................................... 5 Item Banks........................................................................................................................................................... 8 Devices .................................................................................................................................................................