Monitoring of Common Sole Solea Solea (L) Captive Broodstock from Northern Adriatic Sea Over Consecutive Spawning Seasons
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Ashraf I. AHMED 1*, Mariam M. SHARAF 2, and Hebatullah A
ACTA ICHTHYOLOGICA ET PISCATORIA (2010) 40 (2): 161–166 DOI: 10.3750/AIP2010.40.2.09 REPRODUCTION OF THE EGYPTIAN SOLE, SOLEA AEGYPTIACA (ACTINOPTERYGII: PLEURONECTIFORMES: SOLEIDAE), FROM PORT SAID, EGYPT, MEDITERRANEAN SEA Ashraf I. AHMED 1* , Mariam M. SHARAF 2, and Hebatullah A. LABAN 2 1 Marine Science Department , 2 Zoology Department, Faculty of Science, Suez Canal University, Ismailia, Egypt Ahmed A.I., Sharaf M.M., Laban H.A. 2010. Reproduction of the Egyptian sole, Solea aegyptiaca (Actinopterygii: Pleuronectiformes: Soleidae), from Port Said, Egypt, Mediterranean Sea. Acta Ichthyol. Piscat. 40 (2): 161– 166. Background. Understanding of reproduction of the Egyptian sole, Solea aegyptiaca , has been considered a major step toward understanding its population dynamics, especially in the eastern Mediterranean Sea. In Egypt this species has highly commercial value and it has recently been successfully bred in aquaculture, but more data on the reproductive biology is still needed. So, the present work aimed to study the population structure and the reproductive dynamics of S. aegyptiaca over an annual reproductive cycle. Materials and Methods. A total of 594 specimens of S. aegyptiaca were obtained from October 2004 to September 2005 in a monthly basis from the commercial catch. Sex ratio, age, and length at first maturity, gona - do-somatic index, maturity stage, oocyte diameter, and fecundity were analysed in order to increase our knowl - edge on the reproductive dynamics of S. aegyptiaca . Results. The overall ratio between males and females was 1.0 : 1.15 and showed no significant deviation from the expected 1 : 1 ratio, males are dominant among small size, while females are dominant among larger size. -

Research Article Reproductive Biology of the Invasive Sharpbelly
Iran. J. Ichthyol. (March 2019), 6(1): 31-40 Received: August 17, 2018 © 2019 Iranian Society of Ichthyology Accepted: November 1, 2018 P-ISSN: 2383-1561; E-ISSN: 2383-0964 doi: 10.22034/iji.v6i1.285 http://www.ijichthyol.org Research Article Reproductive biology of the invasive sharpbelly, Hemiculter leucisculus (Basilewsky, 1855), from the southern Caspian Sea basin Hamed MOUSAVI-SABET*1,2, Adeleh HEIDARI1, Meysam SALEHI3 1Department of Fisheries, Faculty of Natural Resources, University of Guilan, Sowmeh Sara, Guilan, Iran. 2The Caspian Sea Basin Research Center, University of Guilan, Rasht, Iran. 3Abzi-Exir Aquaculture Co., Agriculture Section, Kowsar Economic Organization, Tehran, Iran. *Email: [email protected] Abstract: The sharpbelly, Hemiculter leucisculus, an invasive species, has expanded its range throughout much of Asia and into the Middle East. However, little is known of its reproductive information regarding spawning pattern and season that could possibly explain its success as an invasive species. This research is the first presentation of its reproductive characteristics, which was conducted based on 235 individuals collected monthly throughout a year from Sefid River, in the southern Caspian Sea basin. Age, sex ratio, fecundity, oocytes diameter and gonado-somatic index were calculated. Regression analyses were used to find relations among fecundity and fish size, gonad weight (Wg) and age. The mature males and females were longer than 93.0 and 99.7mm in total length, respectively (+1 in age). The average egg diameter ranged from 0.4mm (April) to 1.1mm (August). Spawning took place in August, when the water temperature was 23 to 26°C. -

Integrated Coastal Zone Management in the Northern Coast of Egypt
ADAPTATION TO CLIMATE CHANGE IN THE NILE DELTA THROUGH INTEGRATED COASTAL ZONE MANAGEMENT PROJECT INTEGRATED COASTAL ZONE MANAGEMENT IN THE NORTHERN COAST OF EGYPT – A SCOPING STUDY – ANNEX I: ANNEX I: HAZARD CHARACTERIZATION AND CLIMATE CHANGE DECEMBER 2016 ICZM IN THE NORTHERN COAST OF EGYPT – A SCOPING STUDY ANNEX I. HAZARD CHARACTERIZATION AND CLIMATE CHANGE INDEX ANNEX I: HAZARD CHARACTERIZATION AND CLIMATE CHANGE ........................................................ AI.1 1. INTRODUCTION ............................................................................................................................... AI.1 2. HISTORICAL CHARACTERIZATION OF CLIMATIC DRIVERS ............................................................... AI.1 2.1. Waves: GOW database (spatial resolution: 0.125°) ................................................................ AI.1 2.2. Storm surge: GOS database (spatial resolution: 0.064° x 0.114°) ........................................... AI.3 2.3. Sea surface temperature: GRHSST database (spatial resolution: 0.05°) ................................. AI.3 2.4. Air temperature: Seawind II database (spatial resolution: 0.125°)......................................... AI.4 2.5. Precipitation: Seawind II database (spatial resolution: 0.125°) .............................................. AI.4 3. NEAR‐ AND LONG‐TERM PROJECTIONS OF CLIMATIC DRIVERS ...................................................... AI.6 3.1. Waves: GOW projections (spatial resolution: 0.125°) ........................................................... -

Mucosal Health in Aquaculture Page Left Intentionally Blank Mucosal Health in Aquaculture
Mucosal Health in Aquaculture Page left intentionally blank Mucosal Health in Aquaculture Edited by Benjamin H. Beck Stuttgart National Aquaculture Research Center, Stuttgart, Arkansas, USA Eric Peatman School of Fisheries, Aquaculture, and Aquatic Sciences, Auburn University, Alabama, USA AMSTERDAM • BOSTON • HEIDELBERG • LONDON • NEW YORK OXFORD • PARIS • SAN DIEGO • SAN FRANCISCO • SINGAPORE SYDNEY • TOKYO Academic Press is an Imprint of Elsevier Academic Press is an imprint of Elsevier 125, London Wall, EC2Y 5AS, UK 525 B Street, Suite 1800, San Diego, CA 92101-4495, USA 225 Wyman Street, Waltham, MA 02451, USA The Boulevard, Langford Lane, Kidlington, Oxford OX5 1GB, UK Copyright © 2015 Elsevier Inc. All rights reserved. No part of this publication may be reproduced, stored in a retrieval system or transmitted in any form or by any means electronic, mechanical, photocopying, recording or otherwise without the prior written permission of the publisher. Permissions may be sought directly from Elsevier’s Science & Technology Rights Department in Oxford, UK: phone (+44) (0) 1865 843830; fax (+44) (0) 1865 853333; email: [email protected]. Alternatively, visit the Science and Technology Books website at www.elsevierdirect.com/rights for further information. Notice No responsibility is assumed by the publisher for any injury and/or damage to persons or property as a matter of products liability, negligence or otherwise, or from any use or operation of any methods, products, instructions or ideas contained in the material herein. -

Field Identification Guide to the Living Marine Resources of the Eastern
Abdallah, M. 2002. Length-weight relationship of fishes caught by trawl off Alexandria, Egypt. Naga ICLARM Q. 25(1):19–20. Abdul Malak, D., Livingstone, S., Pollard, D., Polidoro, B., Cuttelod, A., Bariche, M., Bilecenoglu, M., Carpenter, K., Collette, B., Francour, P., Goren, M., Kara, M., Massutí, E., Papaconstantinou, C. & Tunesi L. 2011. Overview of the Conservation Status of the Marine Fishes of the Mediterranean Sea. Gland, Switzerland and Malaga, Spain: IUCN, vii + 61 pp. (also available at http://data.iucn.org/dbtw-wpd/edocs/RL-262-001.pdf). Abecasis, D., Bentes, L., Ribeiro, J., Machado, D., Oliveira, F., Veiga, P., Gonçalves, J.M.S & Erzini, K. 2008. First record of the Mediterranean parrotfish, Sparisoma cretense in Ria Formosa (south Portugal). Mar. Biodiv. Rec., 1: e27. DOI: 10.1017/5175526720600248x. Abella, A.J., Arneri, E., Belcari, P., Camilleri, M., Fiorentino, F., Jukic-Peladic, S., Kallianiotis, A., Lembo, G., Papacostantinou, C., Piccinetti, C., Relini, G. & Spedicato, M.T. 2002. Mediterranean stock assessment: current status, problems and perspective: Sub-Committee on Stock Assessment, Barcelona. 18 pp. Abellan, E. & Basurco, B. 1999. Finfish species diversification in the context of Mediterranean marine fish farming development. Marine finfish species diversification: current situation and prospects in Mediterranean aquaculture. CIHEAM/FAO, 9–27. CIHEAM/FAO, Zaragoza. ACCOBAMS, May 2009 www.accobams.org Agostini, V.N. & Bakun, A. 2002. “Ocean triads” in the Mediterranean Sea: physical mechanisms potentially structuring reproductive habitat suitability (with example application to European anchovy, Engraulis encrasicolus), Fish. Oceanogr., 3: 129–142. Akin, S., Buhan, E., Winemiller, K.O. & Yilmaz, H. 2005. Fish assemblage structure of Koycegiz Lagoon-Estuary, Turkey: spatial and temporal distribution patterns in relation to environmental variation. -

Are You Suprised ?
CURRICULUM VITA Name: Prof. Dr. Sahar Fahmy Youssef Mehanna Birth date: 15.4.1964 - Port Said, Egypt Occupation: Professor of Fish Population Dynamics and fish stock assessment, National Institute of Oceanography and Fisheries NIOF, P. O. Box 182, Suez, Egypt. E-mail: [email protected] [email protected] Work phone: +2 - 062 - 3360015 Mobile: +201063770701 URL: http://livedna.org/20.257 Education 1997 Ph.D. (The study of biology and population dynamics of Lethrinus mahsena in the Gulf of Suez), Faculty of Science, Zoology Department, Zagazig University. 1993 M.Sc., (Rational exploitation of kuruma shrimp Penaeus japonicus Bate, 1888 in the Gulf of Suez), Faculty of Science, Zoology Department, Zagazig University. 1985 B.Sc., (Excellent) Zoology Department - Faculty of Science, Suez Canal University. Career history March 2020 – November 2020: Supervisor of the NIOF research station for aquaculture January 2018 – Present: Consultant for the National company for aquaculture and fisheries, Defense Ministry November 2017 – April 2018: Director of Red Sea Branch, NIOF March 2015 – October 2015: Director of Suez and Aqaba Gulfs Branch, NIOF December 2014 -March 2015: Head of Fisheries Division, NIOF March 2013- December 2013: Stock Assessment Program advisor in TCP/SNG/3402 Capacity building in fisheries stock assessment in GCC Project, FAO, GCC and Yemen, FAO. 1 August 2013-November 2020: Head of Fish Population Dynamics Lab, NIOF, Egypt. Sep 2011- Sep 2013: Stock assessment and Fisheries management expert in Marine sciences and Fisheries Center, Ministry of Agriculture and Fisheries Wealth, Sultanate of Oman. 2005 – 2011: Head of Fish Population Dynamics Lab, National Institute of Oceanography and Fisheries NIOF, Egypt. -
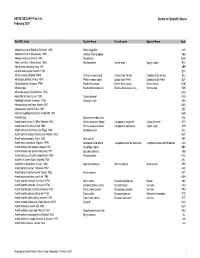
ASFIS ISSCAAP Fish List February 2007 Sorted on Scientific Name
ASFIS ISSCAAP Fish List Sorted on Scientific Name February 2007 Scientific name English Name French name Spanish Name Code Abalistes stellaris (Bloch & Schneider 1801) Starry triggerfish AJS Abbottina rivularis (Basilewsky 1855) Chinese false gudgeon ABB Ablabys binotatus (Peters 1855) Redskinfish ABW Ablennes hians (Valenciennes 1846) Flat needlefish Orphie plate Agujón sable BAF Aborichthys elongatus Hora 1921 ABE Abralia andamanika Goodrich 1898 BLK Abralia veranyi (Rüppell 1844) Verany's enope squid Encornet de Verany Enoploluria de Verany BLJ Abraliopsis pfefferi (Verany 1837) Pfeffer's enope squid Encornet de Pfeffer Enoploluria de Pfeffer BJF Abramis brama (Linnaeus 1758) Freshwater bream Brème d'eau douce Brema común FBM Abramis spp Freshwater breams nei Brèmes d'eau douce nca Bremas nep FBR Abramites eques (Steindachner 1878) ABQ Abudefduf luridus (Cuvier 1830) Canary damsel AUU Abudefduf saxatilis (Linnaeus 1758) Sergeant-major ABU Abyssobrotula galatheae Nielsen 1977 OAG Abyssocottus elochini Taliev 1955 AEZ Abythites lepidogenys (Smith & Radcliffe 1913) AHD Acanella spp Branched bamboo coral KQL Acanthacaris caeca (A. Milne Edwards 1881) Atlantic deep-sea lobster Langoustine arganelle Cigala de fondo NTK Acanthacaris tenuimana Bate 1888 Prickly deep-sea lobster Langoustine spinuleuse Cigala raspa NHI Acanthalburnus microlepis (De Filippi 1861) Blackbrow bleak AHL Acanthaphritis barbata (Okamura & Kishida 1963) NHT Acantharchus pomotis (Baird 1855) Mud sunfish AKP Acanthaxius caespitosa (Squires 1979) Deepwater mud lobster Langouste -

Wet) Housefly Maggots (Musca Domestica) Only Row Having Different Superscripts Are Differ Significantly (P≤0.05
Journal of Zoological Research | Volume 02 | Issue 02 | April 2020 Journal of Zoological Research http://ojs.bilpublishing.com/index.php/jzr ARTICLE Effect of Feeding on Fresh (wet) Housefly Maggots (Musca domestica) with or without Artificial Diet on Water Quality and Growth Rates of African Catfish (Clarias gariepinus Burchell, 1822) Fry under Laboratory Conditions Hamed H.E. Saleh* Aquaculture Division, National Institute of Oceanography and Fisheries (NIOF), El-Fayoum, Egypt ARTICLE INFO ABSTRACT Article history No or little information on the use fresh (wet) housefly maggots Received: 24 June 2020 (Musca domestica) in African catfish Clarias( gariepinus) fry feeding. Therefore, this study was conducted to investigate the effect of feeding Accepted: 17 July 2020 on fresh (wet) housefly maggots with or without artificial diet on water Published Online: 30 July 2020 quality, growth performance, survival percentage and feed utilization of African catfish fry under laboratory conditions. Housefly maggots Keywords: produced from a mixture of poultry droppings and foods wastes, it African catfish was used to replace artificial feed at 0, 50 and 100% levels. Catfish were fed artificial diet alone (Feed 1), fresh (wet) housefly maggots Fresh (wet) housefly maggots alone (Feed 2), and 50% fresh housefly maggots with 50% artificial Artificial diet diet (Feed 3) were prepared and tested on triplicate groups of African Water quality catfish fry (initial weight of 0.25±0.02 g) for 60 days. Results showed that final weight (g/fish) was significantly (P≤0.05) higher in fish Growth performance fed on feed 3 (6.03±0.08), followed by fish fed feed 2 (4.62±0.27), Survival percentage and feed utilization followed by fish fed feed 1 (3.15±0.68). -

FAMILY Soleidae Bonaparte, 1833
FAMILY Soleidae Bonaparte, 1833 - true soles [=Soleini, Synapturiniae (Synapturinae), Brachirinae, Heteromycterina, Pardachirinae, Aseraggodinae, Aseraggodinae] Notes: Soleini Bonaparte 1833: Fasc. 4, puntata 22 [ref. 516] (subfamily) Solea Synapturniae [Synapturinae] Jordan & Starks, 1906:227 [ref. 2532] (subfamily) Synaptura [von Bonde 1922:21 [ref. 520] also used Synapturniae; stem corrected to Synaptur- by Jordan 1923a:170 [ref. 2421], confirmed by Chabanaud 1927:2 [ref. 782] and by Lindberg 1971:204 [ref. 27211]; senior objective synonym of Brachirinae Ogilby, 1916] Brachirinae Ogilby, 1916:136 [ref. 3297] (subfamily) Brachirus Swainson [junior objective synonym of Synapturinae Jordan & Starks, 1906, invalid, Article 61.3.2] Heteromycterina Chabanaud, 1930a:5, 20 [ref. 784] (section) Heteromycteris Pardachirinae Chabanaud, 1937:36 [ref. 793] (subfamily) Pardachirus Aseraggodinae Ochiai, 1959:154 [ref. 32996] (subfamily) Aseraggodus [unavailable publication] Aseraggodinae Ochiai, 1963:20 [ref. 7982] (subfamily) Aseraggodus GENUS Achiroides Bleeker, 1851 - true soles [=Achiroides Bleeker [P.], 1851:262, Eurypleura Kaup [J. J.], 1858:100] Notes: [ref. 325]. Masc. Plagusia melanorhynchus Bleeker, 1851. Type by monotypy. Apparently appeared first as Achiroïdes melanorhynchus Blkr. = Plagusia melanorhynchus Blkr." Species described earlier in same journal as P. melanorhynchus (also spelled melanorhijnchus). Diagnosis provided in Bleeker 1851:404 [ref. 6831] in same journal with second species leucorhynchos added. •Valid as Achiroides Bleeker, 1851 -- (Kottelat 1989:20 [ref. 13605], Roberts 1989:183 [ref. 6439], Munroe 2001:3880 [ref. 26314], Kottelat 2013:463 [ref. 32989]). Current status: Valid as Achiroides Bleeker, 1851. Soleidae. (Eurypleura) [ref. 2578]. Fem. Plagusia melanorhynchus Bleeker, 1851. Type by being a replacement name. Unneeded substitute for Achiroides Bleeker, 1851. •Objective synonym of Achiroides Bleeker, 1851 -- (Kottelat 2013:463 [ref. -

Socio-Economic Analysis of Egyptian Fisheries: Options for Improvement
FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS SOCIOSOCIOSOCIO---ECONOMIC ANALYSISANALYSISANALYSIS OF EEEGYPTIAN FISHERIES: OPTIONOPTIONOPTIONSSS FOR IMPROVEMENT GGCP/INT/041/ECCP/INT/041/EC ––– GRE ––– ITAITAITA Athens (Greece),),), AprilAprilApril 2012012014201444 i The conclusions and recommendations given in this and in other documents in the Scientific and Institutional Cooperation to Support Responsible Fisheries in the Eastern Mediterranean series are those considered appropriate at the time of preparation. They may be modified in the light of further knowledge gained in subsequent stages of the Project. The designations employed and the presentation of material in this publication do not imply the expression of any opinion on the part of FAO or donors concerning the legal status of any country, territory, city or area, or concerning the determination of its frontiers or boundaries. ii Preface The Project “Scientific and Institutional Cooperation to Support Responsible Fisheries in the Eastern Mediterranean - EastMed is executed by the Food and Agriculture Organization of the United Nations (FAO) and funded by Greece, Italy and EC. The Eastern Mediterranean countries have for long lacked a cooperation framework as created for other areas of the Mediterranean, namely the FAO sub-regional projects AdriaMed, MedSudMed, CopeMed II and ArtFiMed. This made it more difficult for some countries in the region to participate fully in international and regional initiatives for cooperation on fishery research and management. Following the very encouraging experience of technical and institutional assistance provided to countries by the other FAO sub-regional Projects, EastMed was born to support the development of regional cooperation and the further development of multidisciplinary expertise necessary to formulate appropriate management measures under the FAO Code of Conduct for Responsible Fisheries and the principles of the Ecosystem Approach to Fisheries (EAF) to ensure rational, responsible and participative fisheries management. -

Download (4MB)
Status of the tiger tooth croaker, Otolithes ruber (Schneider, 1801) and jinga shrimp, Metapenaeus affinis(H. Milne Edwards, 1837) stocks in Khouzestan coastal waters Item Type monograph Authors Eskandari, Gholamreza; Hashemi, S.A.; Taghavi motlagh, A.; Dehghan madiseh, S.; Mayahi, Y.; Albuobaid, S. Publisher Iranian Fisheries Science Research Institute Download date 07/10/2021 17:26:24 Link to Item http://hdl.handle.net/1834/39797 وزارت ﺟﻬﺎد ﻛﺸﺎورزي ﺳﺎزﻣﺎن ﺗﺤﻘﻴﻘﺎت ، آﻣﻮزش و ﺗﺮوﻳﺞﻛ ﺸﺎورزي ﻣﺆﺳﺴﻪ ﺗﺤﻘﻴﻘﺎت ﻋﻠﻮم ﺷﻴﻼﺗ ﻲ ﻛﺸﻮر – ﭘﮋوﻫﺸﻜﺪه آﺑﺰي ﭘﺮوري ﺟﻨﻮب ﻛﺸﻮر ﻋﻨﻮان : ﺑﺮرﺳﻲ وﺿﻌﻴﺖ ذﺧﺎﻳﺮ ﻣﺎﻫﻲ ﺷﻮرﻳﺪه و ﻣﻴﮕﻮي ﺳﻔﻴﺪ در ﺳﻮاﺣﻞ ﺧﻮزﺳﺘﺎن ﻣﺠﺮي : ﻏﻼﻣﺮﺿﺎ اﺳﻜﻨﺪري ﺷﻤﺎره ﺛﺒﺖ 42040 وزارت ﺟﻬﺎد ﻛﺸﺎورزي ﺳﺎزﻣﺎن ﺗﺤﻘﻴﻘﺎت، آﻣﻮزش و ﺗﺮوﻳﭻ ﻛﺸﺎورزي ﻣﺆﺳﺴﻪ ﺗﺤﻘﻴﻘﺎت ﻋﻠﻮم ﺷﻴﻼﺗﻲ ﻛﺸﻮر - ﭘﮋوﻫﺸﻜﺪه آﺑﺰي ﭘﺮوري ﺟﻨﻮب ﻛﺸﻮر ﻋﻨﻮان ﭘﺮوژه ﻃﺮح : ﺑﺮرﺳﻲ وﺿﻌﻴﺖ ذﺧﺎﻳﺮ ﻣﺎﻫﻲ ﺷﻮرﻳﺪه و ﻣﻴﮕﻮي ﺳﻔﻴﺪ در ﺳﻮاﺣﻞ ﺧﻮزﺳﺘﺎن ﺷﻤﺎره ﻣﺼﻮب : -89045 12- 74-4 74-4 ﻧﺎم و ﻧﺎم ﺧﺎﻧﻮادﮔﻲ ﻧﮕﺎرﻧﺪه / ﻧﮕﺎرﻧﺪﮔﺎن : ﻏﻼﻣﺮﺿﺎ اﺳﻜﻨﺪري ﻧﺎم و ﻧﺎم ﺧﺎﻧﻮادﮔﻲ ﻣﺠﺮي ﻣﺴﺌﻮل ( اﺧﺘﺼﺎص ﺑﻪ ﭘﺮوژه ﻫﺎ و ﻃﺮﺣﻬﺎي ﻣﻠﻲ و ﻣﺸﺘﺮك دارد ) : - ﻧﺎم و ﻧﺎم ﺧﺎﻧﻮادﮔﻲ ﻣﺠﺮي / ﻣﺠﺮﻳﺎن : ﻏﻼﻣﺮﺿﺎ اﺳﻜﻨﺪري ﻧﺎم و ﻧﺎم ﺧﺎﻧﻮادﮔﻲ ﻫﻤﻜﺎران : ﺳﻴﺪ اﺣﻤﺪ رﺿﺎ ﻫﺎﺷﻤﻲ، ﺳﻴﺪ اﻣﻴﻦ اﷲ ﺗﻘﻮي، ﺳﻴﻤﻴﻦ دﻫﻘﺎن ﻣﺪﻳﺴﻪ، ﻳﻮﺳﻒ ﻣﻴﺎﺣﻲ و ﺻﺎدق آﻟﺒﻮﻋﺒﻴﺪ ﻧﺎم و ﻧﺎم ﺧﺎﻧﻮادﮔﻲ ﻣﺸﺎوران : - ﻧﺎم و ﻧﺎم ﺧﺎﻧﻮادﮔﻲ ﻧﺎﻇﺮ : ﻓﺮﻫﺎد ﻛﻴﻤﺮام ﻣﺤﻞ اﺟﺮا : اﺳﺘﺎن ﺧﻮزﺳﺘﺎن ﺗﺎرﻳﺦ ﺷﺮوع : /1/4 89 89 ﻣﺪت اﺟﺮا : 1 ﺳﺎل و 9 ﻣﺎه ﻧﺎﺷﺮ : ﻣﻮﺳﺴﻪ ﺗﺤﻘﻴﻘﺎت ﻋﻠﻮم ﺷﻴﻼﺗﻲ ﻛﺸﻮر ﺗﺎرﻳﺦ اﻧﺘﺸﺎر : ﺳﺎل1392 ﺣﻖ ﭼﺎپ ﺑﺮاي ﻣﺆﻟﻒ ﻣﺤﻔﻮظ اﺳﺖ . ﻧﻘﻞ ﻣﻄﺎﻟﺐ ، ﺗﺼﺎوﻳﺮ ، ﺟﺪاول ، ﻣﻨﺤﻨﻲ ﻫﺎ و ﻧﻤﻮدارﻫﺎ ﺑﺎ ذﻛﺮ ﻣﺄﺧﺬ ﺑﻼﻣﺎﻧﻊ اﺳﺖ . « ﺳﻮاﺑﻖ ﻃﺮح ﻳﺎ ﭘﺮوژه و ﻣﺠﺮي» ﭘﺮوژه : ﺑﺮرﺳﻲ وﺿﻌﻴﺖ ذﺧﺎﻳﺮ ﻣﺎﻫﻲ ﺷﻮرﻳﺪه و ﻣﻴﮕﻮي ﺳﻔﻴﺪ در ﺳﻮاﺣﻞ ﺧﻮزﺳﺘﺎن ﻛﺪ ﻣﺼﻮب : -89045 12- 74-4 ﺷﻤﺎره ﺛﺒﺖ ( ﻓﺮوﺳﺖ ) : 42040 ﺗﺎرﻳﺦ : /27/9 91 91 ﺑﺎ ﻣﺴﺌﻮﻟﻴﺖ اﺟﺮاﻳﻲ ﺟﻨﺎب آﻗﺎي ﻏﻼﻣﺮﺿﺎ اﺳﻜﻨﺪري داراي ﻣﺪرك ﺗﺤﺼﻴﻠﻲ دﻛﺘﺮي در رﺷﺘﻪ ﺷﻴﻼت ﻣﻲ ﺑﺎﺷﺪ . -

The Utilization of Soybean Meal in Formulated Diet for Marble Goby, Oxyeleotris Marmoratus
Journal of Agricultural Science; Vol. 5, No. 11; 2013 ISSN 1916-9752 E-ISSN 1916-9760 Published by Canadian Center of Science and Education The Utilization of Soybean Meal in Formulated Diet for Marble Goby, Oxyeleotris marmoratus Annita Seok Kian Yong1, Shing Yau Ooi1 & Rossita Shapawi1 1 Borneo Marine Research Institute, Universiti Malaysia Sabah, Malaysia Correspondence: Annita Seok Kian Yong, Universiti Malaysia Sabah, Jalan UMS 88400, Kota Kinabalu, Sabah, Malaysia. Tel: 60-8832-0000 ext. 2589. E-mail: [email protected] Received: August 13, 2013 Accepted: September 22, 2013 Online Published: October 15, 2013 doi:10.5539/jas.v5n11p139 URL: http://dx.doi.org/10.5539/jas.v5n11p139 Abstract Marble goby, Oxyeleotris marmoratus is a carnivorous fish that highly demanded in Asia region and has great potential in aquaculture industry. A feeding trial was conducted to investigate the possibility of replacing fishmeal with soybean meal in the diet for marble goby juvenile. Fish (initial body weight and length 0.28±0.01 g and 2.60±0.04 cm respectively) were fed with four isonitrogenous and isolipidic diets, which contained 0%, 10% and 20% of defatted soybean meal (SB0, SB10 and SB20, respectively) and 20% of soybean meal supplemented with 2000 FTU/kg phytase (SB20+P). The fish were randomly distributed into 12L aquariums and hand-fed till apparent satiation twice daily. After 40-day of feeding trial, highest growth was observed in fish fed SB0, without significant difference with fish fed SB10; both groups were significant better than juveniles fed SB20 and SB20+P. Treatment SB20+P obtained slightly higher growth than those fed SB20.