Structural Insight on the Recognition of Surface-Bound Opsonins by the Integrin I Domain of Complement Receptor 3
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Complement Recognition Pathways in Renal Transplantation
BRIEF REVIEW www.jasn.org Complement Recognition Pathways in Renal Transplantation Christopher L. Nauser, Conrad A. Farrar, and Steven H. Sacks Medical Research Council Centre for Transplantation, Division of Transplantation Immunology and Mucosal Biology, King’s College London, National Health Service Guy’s and St. Thomas’ Trust, London, United Kingdom ABSTRACT The complement system, consisting of soluble and cell membrane–bound compo- weigh its effect on organ injury and nents of the innate immune system, has defined roles in the pathophysiology of renal rejection with dampening the antimi- allograft rejection. Notably, the unavoidable ischemia-reperfusion injury inherent to crobial functions of the complement sys- transplantation is mediated through the terminal complement activation products tem, such as opsonisation, cell lysis, and C5a and C5b-9. Furthermore, biologically active fragments C3a and C5a, produced recruitment of neutrophils and other in- during complement activation, can modulate both antigen presentation and T cell flammatory cells.7 It is our belief that priming, ultimately leading to allograft rejection. Earlier work identified renal tubule therapeutic strategies can be designed cell synthesis of C3, rather than hepatic synthesis of C3, as the primary source of C3 to specificallytargetthekeyinitiators driving these effects. Recent efforts have focused on identifying the local triggers of of complement activation at the relevant complement activation. Collectin-11, a soluble C-type lectin expressed in renal tis- location, such as complement-binding sue, has been implicated as an important trigger of complement activation in renal anti-HLA antibodies in the vascular tissue. In particular, collectin-11 has been shown to engage L-fucose at sites of compartment or the initiator(s) of local ischemic stress, activating the lectin complement pathway and directing the innate complement activation in the extravas- immune response to the distressed renal tubule. -

Propranolol-Mediated Attenuation of MMP-9 Excretion in Infants with Hemangiomas
Supplementary Online Content Thaivalappil S, Bauman N, Saieg A, Movius E, Brown KJ, Preciado D. Propranolol-mediated attenuation of MMP-9 excretion in infants with hemangiomas. JAMA Otolaryngol Head Neck Surg. doi:10.1001/jamaoto.2013.4773 eTable. List of All of the Proteins Identified by Proteomics This supplementary material has been provided by the authors to give readers additional information about their work. © 2013 American Medical Association. All rights reserved. Downloaded From: https://jamanetwork.com/ on 10/01/2021 eTable. List of All of the Proteins Identified by Proteomics Protein Name Prop 12 mo/4 Pred 12 mo/4 Δ Prop to Pred mo mo Myeloperoxidase OS=Homo sapiens GN=MPO 26.00 143.00 ‐117.00 Lactotransferrin OS=Homo sapiens GN=LTF 114.00 205.50 ‐91.50 Matrix metalloproteinase‐9 OS=Homo sapiens GN=MMP9 5.00 36.00 ‐31.00 Neutrophil elastase OS=Homo sapiens GN=ELANE 24.00 48.00 ‐24.00 Bleomycin hydrolase OS=Homo sapiens GN=BLMH 3.00 25.00 ‐22.00 CAP7_HUMAN Azurocidin OS=Homo sapiens GN=AZU1 PE=1 SV=3 4.00 26.00 ‐22.00 S10A8_HUMAN Protein S100‐A8 OS=Homo sapiens GN=S100A8 PE=1 14.67 30.50 ‐15.83 SV=1 IL1F9_HUMAN Interleukin‐1 family member 9 OS=Homo sapiens 1.00 15.00 ‐14.00 GN=IL1F9 PE=1 SV=1 MUC5B_HUMAN Mucin‐5B OS=Homo sapiens GN=MUC5B PE=1 SV=3 2.00 14.00 ‐12.00 MUC4_HUMAN Mucin‐4 OS=Homo sapiens GN=MUC4 PE=1 SV=3 1.00 12.00 ‐11.00 HRG_HUMAN Histidine‐rich glycoprotein OS=Homo sapiens GN=HRG 1.00 12.00 ‐11.00 PE=1 SV=1 TKT_HUMAN Transketolase OS=Homo sapiens GN=TKT PE=1 SV=3 17.00 28.00 ‐11.00 CATG_HUMAN Cathepsin G OS=Homo -

An Anticomplement Agent That Homes to the Damaged Brain and Promotes Recovery After Traumatic Brain Injury in Mice
An anticomplement agent that homes to the damaged brain and promotes recovery after traumatic brain injury in mice Marieta M. Rusevaa,1,2, Valeria Ramagliab,1, B. Paul Morgana, and Claire L. Harrisa,3 aInstitute of Infection and Immunity, School of Medicine, Cardiff University, Cardiff CF14 4XN, United Kingdom; and bDepartment of Genome Analysis, Academic Medical Center, Amsterdam 1105 AZ, The Netherlands Edited by Douglas T. Fearon, Cornell University, Cambridge, United Kingdom, and approved September 29, 2015 (received for review July 15, 2015) Activation of complement is a key determinant of neuropathology to rapidly and specifically inhibit MAC at sites of complement and disability after traumatic brain injury (TBI), and inhibition is activation, and test its therapeutic potential in experimental TBI. neuroprotective. However, systemic complement is essential to The construct, termed CD59-2a-CRIg, comprises CD59a linked fight infections, a critical complication of TBI. We describe a to CRIg via the murine IgG2a hinge. CD59a prevents assembly targeted complement inhibitor, comprising complement receptor of MAC in cell membranes (16), whereas CRIg binds C3b/iC3b of the Ig superfamily (CRIg) fused with complement regulator CD59a, deposited at sites of complement activation (17). The IgG2a designed to inhibit membrane attack complex (MAC) assembly at hinge promotes dimerization to increase ligand avidity. CD59- sites of C3b/iC3b deposition. CRIg and CD59a were linked via the 2a-CRIg protected in the TBI model, demonstrating that site- IgG2a hinge, yielding CD59-2a-CRIg dimer with increased iC3b/C3b targeted anti-MAC therapeutics may be effective in prevention binding avidity and MAC inhibitory activity. CD59-2a-CRIg inhibited of secondary neuropathology and improve neurologic recovery MAC formation and prevented complement-mediated lysis in vitro. -

Supplementary Table 1: Adhesion Genes Data Set
Supplementary Table 1: Adhesion genes data set PROBE Entrez Gene ID Celera Gene ID Gene_Symbol Gene_Name 160832 1 hCG201364.3 A1BG alpha-1-B glycoprotein 223658 1 hCG201364.3 A1BG alpha-1-B glycoprotein 212988 102 hCG40040.3 ADAM10 ADAM metallopeptidase domain 10 133411 4185 hCG28232.2 ADAM11 ADAM metallopeptidase domain 11 110695 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 195222 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 165344 8751 hCG20021.3 ADAM15 ADAM metallopeptidase domain 15 (metargidin) 189065 6868 null ADAM17 ADAM metallopeptidase domain 17 (tumor necrosis factor, alpha, converting enzyme) 108119 8728 hCG15398.4 ADAM19 ADAM metallopeptidase domain 19 (meltrin beta) 117763 8748 hCG20675.3 ADAM20 ADAM metallopeptidase domain 20 126448 8747 hCG1785634.2 ADAM21 ADAM metallopeptidase domain 21 208981 8747 hCG1785634.2|hCG2042897 ADAM21 ADAM metallopeptidase domain 21 180903 53616 hCG17212.4 ADAM22 ADAM metallopeptidase domain 22 177272 8745 hCG1811623.1 ADAM23 ADAM metallopeptidase domain 23 102384 10863 hCG1818505.1 ADAM28 ADAM metallopeptidase domain 28 119968 11086 hCG1786734.2 ADAM29 ADAM metallopeptidase domain 29 205542 11085 hCG1997196.1 ADAM30 ADAM metallopeptidase domain 30 148417 80332 hCG39255.4 ADAM33 ADAM metallopeptidase domain 33 140492 8756 hCG1789002.2 ADAM7 ADAM metallopeptidase domain 7 122603 101 hCG1816947.1 ADAM8 ADAM metallopeptidase domain 8 183965 8754 hCG1996391 ADAM9 ADAM metallopeptidase domain 9 (meltrin gamma) 129974 27299 hCG15447.3 ADAMDEC1 ADAM-like, -

APC Anti-Human Cd11b (ICRF44) Catalog Number: 20-0118
TECHNICAL DATA SHEET APC Anti-Human CD11b (ICRF44) Catalog Number: 20-0118 PRODUCT INFORMATION Count Contents: APC Anti-Human CD11b (ICRF44) Isotype: Mouse IgG1, kappa 0 0 1 2 3 4 10 10 10 10 10 Human CD11b (ICRF44) APC Concentration: 5 uL (1 ug)/test Clone: ICRF44 Human peripheral blood monocytes were stained with 5 uL (1 ug) APC Anti-Human CD11b (20-0118) (solid line) or 1 ug APC Mouse Reactivity: Human IgG1 isotype control (dashed line). Formulation: 10 mM NaH2PO4, 150 mM NaCl, 0.09% NaN3, 0.1% gelatin, pH7.2 DESCRIPTION The ICRF44 antibody reacts with human CD11b, also known as integrin alpha M. This 165-170 kDa cell surface glycoprotein is part of a family of integrin receptors that mediate adhesion between cells (cell-cell) and components of the extracellular matrix, e.g. fibrinogen (cell-matrix). In addition, integrins are active signaling receptors which recruit leukocytes to inflammatory sites and promote cell activa- tion. Complete, functional integrin receptors consist of distinct combinations of integrin chains which are differentially expressed. Integrin alpha M (CD11b) assembles with Integrin beta-2 (CD18) into a receptor known as Macrophage Antigen-1 (Mac-1) or complement receptor type 3 (CR3). This receptor binds and induces intracellular signaling through ICAM-1, ICAM-2, ICAM-3 and ICAM-4 on endothelial cells and can also facilitate removal of iC3b bearing foreign cells.The ICRF44 antibody is widely used as a marker for CD11b expression on macrophages, granulocytes, and subsets of NK cells. It is reported to be cross-reactive with a number of non-human species including Baboon, Chimpanzee, Cynomolgus, Rhesus and Swine. -

CD Markers Are Routinely Used for the Immunophenotyping of Cells
ptglab.com 1 CD MARKER ANTIBODIES www.ptglab.com Introduction The cluster of differentiation (abbreviated as CD) is a protocol used for the identification and investigation of cell surface molecules. So-called CD markers are routinely used for the immunophenotyping of cells. Despite this use, they are not limited to roles in the immune system and perform a variety of roles in cell differentiation, adhesion, migration, blood clotting, gamete fertilization, amino acid transport and apoptosis, among many others. As such, Proteintech’s mini catalog featuring its antibodies targeting CD markers is applicable to a wide range of research disciplines. PRODUCT FOCUS PECAM1 Platelet endothelial cell adhesion of blood vessels – making up a large portion molecule-1 (PECAM1), also known as cluster of its intracellular junctions. PECAM-1 is also CD Number of differentiation 31 (CD31), is a member of present on the surface of hematopoietic the immunoglobulin gene superfamily of cell cells and immune cells including platelets, CD31 adhesion molecules. It is highly expressed monocytes, neutrophils, natural killer cells, on the surface of the endothelium – the thin megakaryocytes and some types of T-cell. Catalog Number layer of endothelial cells lining the interior 11256-1-AP Type Rabbit Polyclonal Applications ELISA, FC, IF, IHC, IP, WB 16 Publications Immunohistochemical of paraffin-embedded Figure 1: Immunofluorescence staining human hepatocirrhosis using PECAM1, CD31 of PECAM1 (11256-1-AP), Alexa 488 goat antibody (11265-1-AP) at a dilution of 1:50 anti-rabbit (green), and smooth muscle KD/KO Validated (40x objective). alpha-actin (red), courtesy of Nicola Smart. PECAM1: Customer Testimonial Nicola Smart, a cardiovascular researcher “As you can see [the immunostaining] is and a group leader at the University of extremely clean and specific [and] displays Oxford, has said of the PECAM1 antibody strong intercellular junction expression, (11265-1-AP) that it “worked beautifully as expected for a cell adhesion molecule.” on every occasion I’ve tried it.” Proteintech thanks Dr. -

The Complement System: a Powerful Modulator and Effector of Astrocyte Function in the Healthy and Diseased Central Nervous System
cells Review The Complement System: A Powerful Modulator and Effector of Astrocyte Function in the Healthy and Diseased Central Nervous System Marcela Pekna 1,3,4,* and Milos Pekny 2,3,4 1 Laboratory of Regenerative Neuroimmunology, Center for Brain Repair, Department of Clinical Neuroscience, Institute of Neuroscience and Physiology, Sahlgrenska Academy at the University of Gothenburg, 40530 Gothenburg, Sweden 2 Laboratory of Astrocyte Biology and CNS Regeneration, Center for Brain Repair, Department of Clinical Neuroscience, Institute of Neuroscience and Physiology, Sahlgrenska Academy at the University of Gothenburg, 40530 Gothenburg, Sweden; [email protected] 3 Florey Institute of Neuroscience and Mental Health, Parkville, Melbourne 3010, Australia 4 School of Medicine and Public Health, University of Newcastle, Newcastle 2308, Australia * Correspondence: [email protected]; Tel.: +46-31-786-3581 Abstract: The complement system, an effector arm of the innate immune system that plays a critical role in tissue inflammation, the elimination of pathogens and the clearance of dead cells and cell debris, has emerged as a regulator of many processes in the central nervous system, including neural cell genesis and migration, control of synapse number and function, and modulation of glial cell responses. Complement dysfunction has also been put forward as a major contributor to neurological disease. Astrocytes are neuroectoderm-derived glial cells that maintain water and ionic homeostasis, and control cerebral blood flow and multiple aspects of neuronal functioning. By virtue of their Citation: Pekna, M.; Pekny, M. The Complement System: A Powerful expression of soluble as well as membrane-bound complement proteins and receptors, astrocytes are Modulator and Effector of Astrocyte able to both send and receive complement-related signals. -
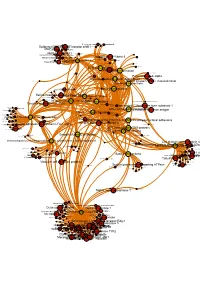
Brcaallwithlabelsintegrina1b1
Receptor-type tyrosine-proteinMuscle- skeletal receptor phosphatase tyrosine protein kinase S Dual specificity mitogen-activated protein kinase kinase 1 Plasminogen activator inhibitor-1 Epidermal growth factor receptor erbB1 Beta amyloid A4 protein MAPDual specificity Epidermalkinase mitogen-activated growth factorCasein proteinreceptor ERK2 kinase and ErbB2kinase; II (HER1 alphaMEK1/2 and (prime) HER2) Casein kinase II alpha Integrin alpha-IIb MAPCasein kinase kinase II beta ERK1 Dual specificity mitogen-activated protein kinase kinase 2 Lysyl oxidase Integrin alpha-IIb/beta-3 Furin Mitogen-activatedSerine/threonine-protein kinase RAF and Dual specificityEpidermal protein mitogen-activated growth proteinkinase; factor kinase kinase receptor 1 (Raf/MEK)ERK1/ERK2 Integrin alpha-V/beta-3 Integrin alpha-V/beta-3 and alpha-IIb/beta 3 Casein kinase II Signal transduction by L1 Integrin alpha-V/beta-6 Casein kinase II alpha/beta Vitronectin receptor alpha Bone morphogenetic protein 2 ECM proteoglycans Bone morphogenetic protein 4 VitronectinElastic fibre formation Peripheral plasma membrane protein CASK Neuropilin-1 Integrin alpha-V/beta-5 Protein kinaseProtein kinase C (PKC) alpha Integrin alpha-2/beta-3 Molecules associated with elastic fibres PKC alpha and beta-2 MER intracellular domain/EGFR extracellular domain chimera Fibronectin receptor alpha Protein kinase C- PKC; classical/novel Syndecan interactions Integrin alpha-2 von Willebrand factorIntegrin alpha-10 Laminin interactions Integrin alpha-11 Fibrinogen beta chain Serine/threonine-proteinVascular -

High Constitutive Cytokine Release by Primary Human Acute Myeloid Leukemia Cells Is Associated with a Specific Intercellular Communication Phenotype
Supplementary Information High Constitutive Cytokine Release by Primary Human Acute Myeloid Leukemia Cells Is Associated with a Specific Intercellular Communication Phenotype Håkon Reikvam 1,2,*, Elise Aasebø 1, Annette K. Brenner 2, Sushma Bartaula-Brevik 1, Ida Sofie Grønningsæter 2, Rakel Brendsdal Forthun 2, Randi Hovland 3,4 and Øystein Bruserud 1,2 1 Department of Clinical Science, University of Bergen, 5020, Bergen, Norway 2 Department of Medicine, Haukeland University Hospital, 5021, Bergen, Norway 3 Department of Medical Genetics, Haukeland University Hospital, 5021, Bergen, Norway 4 Institute of Biomedicine, University of Bergen, 5020, Bergen, Norway * Correspondence: [email protected]; Tel.: +55-97-50-00 J. Clin. Med. 2019, 8, x 2 of 36 Figure S1. Mutational studies in a cohort of 71 AML patients. The figure shows the number of patients with the various mutations (upper), the number of mutations in for each patient (middle) and the number of main classes with mutation(s) in each patient (lower). 2 J. Clin. Med. 2019, 8, x; doi: www.mdpi.com/journal/jcm J. Clin. Med. 2019, 8, x 3 of 36 Figure S2. The immunophenotype of primary human AML cells derived from 62 unselected patients. The expression of the eight differentiation markers CD13, CD14, CD15, CD33, CD34, CD45, CD117 and HLA-DR was investigated for 62 of the 71 patients included in our present study. We performed an unsupervised hierarchical cluster analysis and identified four patient main clusters/patient subsets. The mutational profile for each f the 62 patients is also given (middle), no individual mutation of main class of mutations showed any significant association with any of the for differentiation marker clusters (middle). -

Structural Insight on the Recognition of Surface-Bound Opsonins by the Integrin I Domain of Complement Receptor 3
Structural insight on the recognition of surface-bound opsonins by the integrin I domain of complement receptor 3 Goran Bajica, Laure Yatimea, Robert B. Simb, Thomas Vorup-Jensenc,1, and Gregers R. Andersena,1 Departments of aMolecular Biology and Genetics and cBiomedicine, Aarhus University, DK-8000 Aarhus, Denmark; and bDepartment of Pharmacology, University of Oxford, Oxford OX1 3QT, United Kingdom Edited by Douglas T. Fearon, University of Cambridge School of Clinical Medicine, Cambridge, United Kingdom, and approved August 28, 2013 (received for review June 13, 2013) Complement receptors (CRs), expressed notably on myeloid and degranulation, and changes in leukocyte cytokine production (2, lymphoid cells, play an essential function in the elimination of 5–7). CR3, and to a lesser degree CR4, are essential for the complement-opsonized pathogens and apoptotic/necrotic cells. In phagocytosis of complement-opsonized particles or complexes addition, these receptors are crucial for the cross-talk between the (6, 8, 9). Complement-opsonized immune complexes are cap- innate andadaptive branches ofthe immune system. CR3 (also known tured in the lymph nodes by CR3-positive subcapsular sinus as Mac-1, integrin α β , or CD11b/CD18) is expressed on all macro- macrophages (SSMs) and conveyed directly to naïve B cells or M 2 γ phages and recognizes iC3b on complement-opsonized objects, en- through follicular dendritic cells (10) using CR1, CR2, and Fc abling their phagocytosis. We demonstrate that the C3d moiety of receptors for antigen capture (11, 12). Hence, antigen-presenting iC3b harbors the binding site for the CR3 αI domain, and our structure cells such as SSMs may act as antigen storage and provide B of the C3d:αI domain complex rationalizes the CR3 selectivity for iC3b. -

Complement Receptor CD46 Co-Stimulates Optimal Human CD8 T
ARTICLE DOI: 10.1038/s41467-018-06706-z OPEN Complement receptor CD46 co-stimulates optimal human CD8+ T cell effector function via fatty acid metabolism Giuseppina Arbore1,2, Erin E. West3, Jubayer Rahman3, Gaelle Le Friec2, Nathalie Niyonzima3,4, Mehdi Pirooznia 3, Ilker Tunc3, Polychronis Pavlidis2, Nicholas Powell2, Yuesheng Li3, Poching Liu3, Aude Servais5, Lionel Couzi6, Veronique Fremeaux-Bacchi7, Leo Placais3, Alastair Ferraro8, Patrick R. Walsh9, David Kavanagh9, Behdad Afzali 3,10, Paul Lavender2, Helen J. Lachmann11 & Claudia Kemper2,3,12 1234567890():,; The induction of human CD4+ Th1 cells requires autocrine stimulation of the complement receptor CD46 in direct crosstalk with a CD4+ T cell-intrinsic NLRP3 inflammasome. However, it is unclear whether human cytotoxic CD8+ T cell (CTL) responses also rely on an intrinsic complement-inflammasome axis. Here we show, using CTLs from patients with CD46 deficiency or with constitutively-active NLRP3, that CD46 delivers co-stimulatory signals for optimal CTL activity by augmenting nutrient-influx and fatty acid synthesis. Sur- prisingly, although CTLs express NLRP3, a canonical NLRP3 inflammasome is not required for normal human CTL activity, as CTLs from patients with hyperactive NLRP3 activity function normally. These findings establish autocrine complement and CD46 activity as integral components of normal human CTL biology, and, since CD46 is only present in humans, emphasize the divergent roles of innate immune sensors between mice and men. 1 Division of Immunology, Transplantation and Infectious Diseases, San Raffaele Scientific Institute, Milano, Italy. 2 School of Immunology and Microbial Sciences, King’s College London, London, UK. 3 Laboratory of Molecular Immunology and the Immunology Center, National Heart, Lung, and Blood Institute (NHLBI), National Institutes of Health (NIH), Bethesda, MD, USA. -

Complement Receptor 1 Therapeutics for Prevention of Immune Hemolysis
Review: complement receptor 1 therapeutics for prevention of immune hemolysis K.YAZDANBAKHSH The complement system plays a crucial role in fighting infections biological activities, it has to be activated. Activation and is an important link between the innate and adaptive immune occurs in a sequence that involves proteolytic cleavage responses. However, inappropriate complement activation can cause tissue damage, and it underlies the pathology of many of the complement components, resulting in the diseases. In the transfusion medicine setting, complement release of active biological mediators and the assembly sensitization of RBCs can lead to both intravascular and of active enzyme molecules that result in cleavage of extravascular destruction. Moreover, complement deficiencies are 1 associated with autoimmune disorders, including autoimmune the next downstream complement component. hemolytic anemia (AIHA). Complement receptor 1 (CR1) is a large Depending on the nature of the activators, three single-pass glycoprotein that is expressed on a variety of cell types complement activation pathways have been described: in blood, including RBCs and immune cells. Among its multiple the antibody-dependent classical pathway and the functions is its ability to inhibit complement activation. Furthermore, gene knockout studies in mice implicate a role for antibody-independent alternative and lectin pathways CR1 (along with the alternatively spliced gene product CR2) in (Fig. 1).1 Common to all three pathways are two prevention of autoimmunity. This review discusses the possibility critical steps: the assembly of the C3 convertase that the CR1 protein may be manipulated to prevent and treat AIHA. In addition, it will be shown in an in vivo mouse model of enzymes and the activation of C5 convertases.