Vitro Generated to Ex Vivo CD8 T Cells Comparative and Functional
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Human and Mouse CD Marker Handbook Human and Mouse CD Marker Key Markers - Human Key Markers - Mouse
Welcome to More Choice CD Marker Handbook For more information, please visit: Human bdbiosciences.com/eu/go/humancdmarkers Mouse bdbiosciences.com/eu/go/mousecdmarkers Human and Mouse CD Marker Handbook Human and Mouse CD Marker Key Markers - Human Key Markers - Mouse CD3 CD3 CD (cluster of differentiation) molecules are cell surface markers T Cell CD4 CD4 useful for the identification and characterization of leukocytes. The CD CD8 CD8 nomenclature was developed and is maintained through the HLDA (Human Leukocyte Differentiation Antigens) workshop started in 1982. CD45R/B220 CD19 CD19 The goal is to provide standardization of monoclonal antibodies to B Cell CD20 CD22 (B cell activation marker) human antigens across laboratories. To characterize or “workshop” the antibodies, multiple laboratories carry out blind analyses of antibodies. These results independently validate antibody specificity. CD11c CD11c Dendritic Cell CD123 CD123 While the CD nomenclature has been developed for use with human antigens, it is applied to corresponding mouse antigens as well as antigens from other species. However, the mouse and other species NK Cell CD56 CD335 (NKp46) antibodies are not tested by HLDA. Human CD markers were reviewed by the HLDA. New CD markers Stem Cell/ CD34 CD34 were established at the HLDA9 meeting held in Barcelona in 2010. For Precursor hematopoetic stem cell only hematopoetic stem cell only additional information and CD markers please visit www.hcdm.org. Macrophage/ CD14 CD11b/ Mac-1 Monocyte CD33 Ly-71 (F4/80) CD66b Granulocyte CD66b Gr-1/Ly6G Ly6C CD41 CD41 CD61 (Integrin b3) CD61 Platelet CD9 CD62 CD62P (activated platelets) CD235a CD235a Erythrocyte Ter-119 CD146 MECA-32 CD106 CD146 Endothelial Cell CD31 CD62E (activated endothelial cells) Epithelial Cell CD236 CD326 (EPCAM1) For Research Use Only. -

Granzyme a Released Upon Stimulation of Cytotoxic T Lymphocytes Activates the Thrombin Receptor on Neuronal Cells and Astrocytes HANA S
Proc. Nail. Acad. Sci. USA Vol. 91, pp. 8112-8116, August 1994 Neurobiology Granzyme A released upon stimulation of cytotoxic T lymphocytes activates the thrombin receptor on neuronal cells and astrocytes HANA S. SUIDAN*, JACQUES BOUVIERt, ESTHER SCHAERERt, STUART R. STONES, DENIS MONARD*, AND JURG TSCHOPPt *Fnednch Miescher-Institut, P.O. Box 2543, CH-4002 Basel, Switzerland; tInstitute of Biochemistry, University of Lausanne, CH-1066 Epalinges, Switzerland; and tDepartment of Haematology, University of Cambridge, Medical Research Council Centre, Hills Road, Cambridge CB2 2QH, United Kingdom Communicated by Hans Neurath, May 4, 1994 ABSTRACT Granzymes are a family of serine proteases for example, myelin destruction is believed to be mediated by that are harbored in cytoplasmic granules of activated T these immune effector cells (19, 20). Experimental autoim- lymphocytes and are released upon target cell interaction. mune encephalomyelitis (EAE), a rodent multiple sclerosis- Immediate and complete neurite retraction was induced in a like disease, can be caused by T lymphocytes reactive against mouse neuronal cell line when total extracts ofgranule proteins myelin basic protein (MBP) (21). The encephalitogenic MBP- were added. This activity was isolated and identified as gran- specific T lymphocytes are in most cases CD4+ and major zyme A. This protease not only induced neurite retraction at histocompatibility complex class lI-restricted (17). The nanomolar concentrations but also reversed the stellation of pathogenicity in the central nervous system involves homing, astrocytes. Both effects were critically dependent on the ester- extravasation, and induction of tissue damage. olytic activity of granzyme A. As neurite retraction is known to Little is known about the molecular mechanism by which be induced by thrombin, possible cleavage and activation ofthe encephalitogenic T lymphocytes induce tissue destruction in thrombin receptor were investigated. -

Propranolol-Mediated Attenuation of MMP-9 Excretion in Infants with Hemangiomas
Supplementary Online Content Thaivalappil S, Bauman N, Saieg A, Movius E, Brown KJ, Preciado D. Propranolol-mediated attenuation of MMP-9 excretion in infants with hemangiomas. JAMA Otolaryngol Head Neck Surg. doi:10.1001/jamaoto.2013.4773 eTable. List of All of the Proteins Identified by Proteomics This supplementary material has been provided by the authors to give readers additional information about their work. © 2013 American Medical Association. All rights reserved. Downloaded From: https://jamanetwork.com/ on 10/01/2021 eTable. List of All of the Proteins Identified by Proteomics Protein Name Prop 12 mo/4 Pred 12 mo/4 Δ Prop to Pred mo mo Myeloperoxidase OS=Homo sapiens GN=MPO 26.00 143.00 ‐117.00 Lactotransferrin OS=Homo sapiens GN=LTF 114.00 205.50 ‐91.50 Matrix metalloproteinase‐9 OS=Homo sapiens GN=MMP9 5.00 36.00 ‐31.00 Neutrophil elastase OS=Homo sapiens GN=ELANE 24.00 48.00 ‐24.00 Bleomycin hydrolase OS=Homo sapiens GN=BLMH 3.00 25.00 ‐22.00 CAP7_HUMAN Azurocidin OS=Homo sapiens GN=AZU1 PE=1 SV=3 4.00 26.00 ‐22.00 S10A8_HUMAN Protein S100‐A8 OS=Homo sapiens GN=S100A8 PE=1 14.67 30.50 ‐15.83 SV=1 IL1F9_HUMAN Interleukin‐1 family member 9 OS=Homo sapiens 1.00 15.00 ‐14.00 GN=IL1F9 PE=1 SV=1 MUC5B_HUMAN Mucin‐5B OS=Homo sapiens GN=MUC5B PE=1 SV=3 2.00 14.00 ‐12.00 MUC4_HUMAN Mucin‐4 OS=Homo sapiens GN=MUC4 PE=1 SV=3 1.00 12.00 ‐11.00 HRG_HUMAN Histidine‐rich glycoprotein OS=Homo sapiens GN=HRG 1.00 12.00 ‐11.00 PE=1 SV=1 TKT_HUMAN Transketolase OS=Homo sapiens GN=TKT PE=1 SV=3 17.00 28.00 ‐11.00 CATG_HUMAN Cathepsin G OS=Homo -

Kinesin Family Member 18B Regulates the Proliferation and Invasion Of
Wu et al. Cell Death and Disease (2021) 12:302 https://doi.org/10.1038/s41419-021-03582-2 Cell Death & Disease ARTICLE Open Access Kinesin family member 18B regulates the proliferation and invasion of human prostate cancer cells Yu-Peng Wu 1,Zhi-BinKe 1, Wen-Cai Zheng 1, Ye-Hui Chen 1,Jun-MingZhu 1,FeiLin 1,Xiao-DongLi 1, Shao-Hao Chen 1,HaiCai 1, Qing-Shui Zheng 1, Yong Wei 1, Xue-Yi Xue 1 and Ning Xu 1 Abstract Expression of kinesin family member 18B (KIF18B), an ATPase with key roles in cell division, is deregulated in many cancers, but its involvement in prostate cancer (PCa) is unclear. Here, we investigated the expression and function of KIF18B in human PCa specimens and cell lines using bioinformatics analyses, immunohistochemical and immunofluorescence microscopy, and RT-qPCR and western blot analyses. KIF18B was overexpressed in PCa specimens compared with paracancerous tissues and was associated with poorer disease-free survival. In vitro, KIF18B knockdown in PCa cell lines promoted cell proliferation, migration, and invasion, and inhibited cell apoptosis, while KIF18B overexpression had the opposite effects. In a mouse xenograft model, KIF18B overexpression accelerated and promoted the growth of PCa tumors. Bioinformatics analysis of control and KIF18B-overexpressing PCa cells showed that genes involved in the PI3K–AKT–mTOR signaling pathway were significantly enriched among the differentially expressed genes. Consistent with this observation, we found that KIF18B overexpression activates the PI3K–AKT–mTOR signaling pathway in PCa cells both in vitro and in vivo. Collectively, our results suggest that KIF18B plays a crucial role – – 1234567890():,; 1234567890():,; 1234567890():,; 1234567890():,; in PCa via activation of the PI3K AKT mTOR signaling pathway, and raise the possibility that KIF18B could have utility as a novel biomarker for PCa. -

Defining the Characteristics of Serine Protease-Mediated Cell Death Cascades ⁎ A.R
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Biochimica et Biophysica Acta 1773 (2007) 1491–1499 www.elsevier.com/locate/bbamcr Minireview A more serine way to die: Defining the characteristics of serine protease-mediated cell death cascades ⁎ A.R. O’Connell, C. Stenson-Cox National Centre for Biomedical and Engineering Science, National University of Ireland, Galway, Ireland Received 23 February 2007; received in revised form 11 July 2007; accepted 1 August 2007 Available online 14 August 2007 Abstract The morphological features observed by Kerr, Wylie and Currie in 1972 define apoptosis, necrosis and autophagy. An appreciable number of alternative systems do not fall neatly under these categories, warranting a review of alternative proteolytic machinery and its contribution to cell death. This review aims to pinpoint key molecular features of serine protease-mediated pro-apoptotic signalling. The profile created will contribute to a standard set of biochemical criteria that can serve in differentiating within cell death subtypes. © 2007 Elsevier B.V. All rights reserved. Keywords: Apoptosis; Serine protease; Caspase; Mitochondria 1. Introduction ability (MOMP) triggering the release of apoptogenic factors. Although the underlying mechanism of MOMP induction has The knowledge that cell death is an essential event in the life not been fully ascertained it is known to be regulated by of multi-cellular organisms has been around for more than members of the Bcl-2 family (Fig. 11). Anti-apoptotic Bcl-2 and 150 years. In 1972 Kerr et al., coined the term ‘apoptosis’ to Bcl-xl proteins block MOMP, whilst pro-apoptotic BH-3 describe a distinct morphological pattern of physiologically domain only proteins; Bim and Bid promote MOMP through occurring cell death [1]. -

Anti-ITGAE (GW21349)
3050 Spruce Street, Saint Louis, MO 63103 USA Tel: (800) 521-8956 (314) 771-5765 Fax: (800) 325-5052 (314) 771-5757 email: [email protected] Product Information Anti-ITGAE antibody produced in chicken, affinity isolated antibody Catalog Number GW21349 Formerly listed as GenWay Catalog Number 15-288-21349, Integrin alpha-E Antibody. – Storage Temperature Store at 20 °C The product is a clear, colorless solution in phosphate buffered saline, pH 7.2, containing 0.02% sodium azide. Synonyms: Integrin, alpha E (antigen CD103, human mucosal lymphocyte antigen 1; alpha polypeptide), Mucosal Species Reactivity: Human lymphocyte 1 antigen; HML-1 antigen; Integrin alpha-IEL; Tested Applications: WB CD103 antigen Recommended Dilutions: Recommended starting dilution Product Description for Western blot analysis is 1:500, for tissue or cell staining Integrin alpha-E/beta-7 is a receptor for E-cadherin. It 1:200. mediates adhesion of intra-epithelial T-lymphocytes to epithelial cell monolayers. Note: Optimal concentrations and conditions for each application should be determined by the user. NCBI Accession number: NP_002199.2 Swiss Prot Accession number: P38570 Precautions and Disclaimer This product is for R&D use only, not for drug, household, or Gene Information: Human .. ITGAE (3682) other uses. Due to the sodium azide content a material Immunogen: Recombinant protein Integrin, alpha E (anti- safety data sheet (MSDS) for this product has been sent to gen CD103, human mucosal lymphocyte antigen 1; alpha the attention of the safety officer of your institution. Please polypeptide) consult the Material Safety Data Sheet for information regarding hazards and safe handling practices. Immunogen Sequence: GI # 6007851, sequence 773 - 817 Storage/Stability For continuous use, store at 2–8 °C for up to one week. -
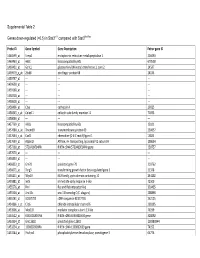
0.5) in Stat3∆/∆ Compared with Stat3flox/Flox
Supplemental Table 2 Genes down-regulated (<0.5) in Stat3∆/∆ compared with Stat3flox/flox Probe ID Gene Symbol Gene Description Entrez gene ID 1460599_at Ermp1 endoplasmic reticulum metallopeptidase 1 226090 1460463_at H60c histocompatibility 60c 670558 1460431_at Gcnt1 glucosaminyl (N-acetyl) transferase 1, core 2 14537 1459979_x_at Zfp68 zinc finger protein 68 24135 1459747_at --- --- --- 1459608_at --- --- --- 1459168_at --- --- --- 1458718_at --- --- --- 1458618_at --- --- --- 1458466_at Ctsa cathepsin A 19025 1458345_s_at Colec11 collectin sub-family member 11 71693 1458046_at --- --- --- 1457769_at H60a histocompatibility 60a 15101 1457680_a_at Tmem69 transmembrane protein 69 230657 1457644_s_at Cxcl1 chemokine (C-X-C motif) ligand 1 14825 1457639_at Atp6v1h ATPase, H+ transporting, lysosomal V1 subunit H 108664 1457260_at 5730409E04Rik RIKEN cDNA 5730409E04Rik gene 230757 1457070_at --- --- --- 1456893_at --- --- --- 1456823_at Gm70 predicted gene 70 210762 1456671_at Tbrg3 transforming growth factor beta regulated gene 3 21378 1456211_at Nlrp10 NLR family, pyrin domain containing 10 244202 1455881_at Ier5l immediate early response 5-like 72500 1455576_at Rinl Ras and Rab interactor-like 320435 1455304_at Unc13c unc-13 homolog C (C. elegans) 208898 1455241_at BC037703 cDNA sequence BC037703 242125 1454866_s_at Clic6 chloride intracellular channel 6 209195 1453906_at Med13l mediator complex subunit 13-like 76199 1453522_at 6530401N04Rik RIKEN cDNA 6530401N04 gene 328092 1453354_at Gm11602 predicted gene 11602 100380944 1453234_at -

Supplementary Table 1: Adhesion Genes Data Set
Supplementary Table 1: Adhesion genes data set PROBE Entrez Gene ID Celera Gene ID Gene_Symbol Gene_Name 160832 1 hCG201364.3 A1BG alpha-1-B glycoprotein 223658 1 hCG201364.3 A1BG alpha-1-B glycoprotein 212988 102 hCG40040.3 ADAM10 ADAM metallopeptidase domain 10 133411 4185 hCG28232.2 ADAM11 ADAM metallopeptidase domain 11 110695 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 195222 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 165344 8751 hCG20021.3 ADAM15 ADAM metallopeptidase domain 15 (metargidin) 189065 6868 null ADAM17 ADAM metallopeptidase domain 17 (tumor necrosis factor, alpha, converting enzyme) 108119 8728 hCG15398.4 ADAM19 ADAM metallopeptidase domain 19 (meltrin beta) 117763 8748 hCG20675.3 ADAM20 ADAM metallopeptidase domain 20 126448 8747 hCG1785634.2 ADAM21 ADAM metallopeptidase domain 21 208981 8747 hCG1785634.2|hCG2042897 ADAM21 ADAM metallopeptidase domain 21 180903 53616 hCG17212.4 ADAM22 ADAM metallopeptidase domain 22 177272 8745 hCG1811623.1 ADAM23 ADAM metallopeptidase domain 23 102384 10863 hCG1818505.1 ADAM28 ADAM metallopeptidase domain 28 119968 11086 hCG1786734.2 ADAM29 ADAM metallopeptidase domain 29 205542 11085 hCG1997196.1 ADAM30 ADAM metallopeptidase domain 30 148417 80332 hCG39255.4 ADAM33 ADAM metallopeptidase domain 33 140492 8756 hCG1789002.2 ADAM7 ADAM metallopeptidase domain 7 122603 101 hCG1816947.1 ADAM8 ADAM metallopeptidase domain 8 183965 8754 hCG1996391 ADAM9 ADAM metallopeptidase domain 9 (meltrin gamma) 129974 27299 hCG15447.3 ADAMDEC1 ADAM-like, -

The Role of Fc Gamma Receptors in the Activity of Therapeutic Monoclonal Antibodies
UNIVERSITY OF SOUTHAMPTON FACULTY OF MEDICINE Cancer Sciences Unit Volume 1 of 1 The Role of Fc Gamma Receptors in the Activity of Therapeutic Monoclonal Antibodies by Robert James Oldham Thesis for the degree of Doctor of Philosophy September 2016 UNIVERSITY OF SOUTHAMPTON ABSTRACT FACULTY OF MEDICINE Biomedicine Thesis for the degree of Doctor of Philosophy THE ROLE OF FC GAMMA RECEPTORS IN THE ACTIVITY OF THERAPEUTIC MONOCLONAL ANTIBODIES Robert James Oldham Fc gamma receptors (FcγRs) are the major family of receptors responsible for interacting with immunoglobulin G (IgG). They are known to be required for the anti-tumour activity of direct targeting mAbs through expression on NK cells and macrophages. Furthermore, recent work has suggested that cross-linking via FcγRs is required for the activity of agonistic, immune modulatory mAb. This thesis sought to investigate the requirement for these receptors for different aspects of mAb activity; from T cell activation to tumour depletion, using a combination of in vitro and in vivo systems. A panel of CHO-K1 cells were generated and transfected to express the polymorphic variants of human FcγRs. These were characterised for their ability to bind IgG before being used as feeder cells in T cell proliferation assays. The assays found that cross-linking of the anti-CD28 mAb, TGN1412 by FcγRIIb (CD32b) or FcγRIIa (CD32a) but not FcγRIIIa (CD16a) transfected cells induced T cell proliferation. Furthermore, this was accompanied by the release of pro-inflammatory cytokines including TNF-α, IFN-γ and IL-2. With the importance of cross-linking via CD32b demonstrated, experiments probed the mechanism of expression using Ramos and Raji cells. -

APC Anti-Human Cd11b (ICRF44) Catalog Number: 20-0118
TECHNICAL DATA SHEET APC Anti-Human CD11b (ICRF44) Catalog Number: 20-0118 PRODUCT INFORMATION Count Contents: APC Anti-Human CD11b (ICRF44) Isotype: Mouse IgG1, kappa 0 0 1 2 3 4 10 10 10 10 10 Human CD11b (ICRF44) APC Concentration: 5 uL (1 ug)/test Clone: ICRF44 Human peripheral blood monocytes were stained with 5 uL (1 ug) APC Anti-Human CD11b (20-0118) (solid line) or 1 ug APC Mouse Reactivity: Human IgG1 isotype control (dashed line). Formulation: 10 mM NaH2PO4, 150 mM NaCl, 0.09% NaN3, 0.1% gelatin, pH7.2 DESCRIPTION The ICRF44 antibody reacts with human CD11b, also known as integrin alpha M. This 165-170 kDa cell surface glycoprotein is part of a family of integrin receptors that mediate adhesion between cells (cell-cell) and components of the extracellular matrix, e.g. fibrinogen (cell-matrix). In addition, integrins are active signaling receptors which recruit leukocytes to inflammatory sites and promote cell activa- tion. Complete, functional integrin receptors consist of distinct combinations of integrin chains which are differentially expressed. Integrin alpha M (CD11b) assembles with Integrin beta-2 (CD18) into a receptor known as Macrophage Antigen-1 (Mac-1) or complement receptor type 3 (CR3). This receptor binds and induces intracellular signaling through ICAM-1, ICAM-2, ICAM-3 and ICAM-4 on endothelial cells and can also facilitate removal of iC3b bearing foreign cells.The ICRF44 antibody is widely used as a marker for CD11b expression on macrophages, granulocytes, and subsets of NK cells. It is reported to be cross-reactive with a number of non-human species including Baboon, Chimpanzee, Cynomolgus, Rhesus and Swine. -

Stearoyl-Coa Desaturase 1 As a Therapeutic Target for the Treatment of Cancer
cancers Review Stearoyl-CoA Desaturase 1 as a Therapeutic Target for the Treatment of Cancer Zuzanna Tracz-Gaszewska and Pawel Dobrzyn * Laboratory of Molecular Medical Biochemistry, Nencki Institute of Experimental Biology Polish Academy of Sciences, 02-093 Warsaw, Poland * Correspondence: [email protected] Received: 5 June 2019; Accepted: 3 July 2019; Published: 5 July 2019 Abstract: A distinctive feature of cancer cells of various origins involves alterations of the composition of lipids, with significant enrichment in monounsaturated fatty acids. These molecules, in addition to being structural components of newly formed cell membranes of intensely proliferating cancer cells, support tumorigenic signaling. An increase in the expression of stearoyl-CoA desaturase 1 (SCD1), the enzyme that converts saturated fatty acids to D9-monounsaturated fatty acids, has been observed in a wide range of cancer cells, and this increase is correlated with cancer aggressiveness and poor outcomes for patients. Studies have demonstrated the involvement of SCD1 in the promotion of cancer cell proliferation, migration, metastasis, and tumor growth. Many studies have reported a role for this lipogenic factor in maintaining the characteristics of cancer stem cells (i.e., the population of cells that contributes to cancer progression and resistance to chemotherapy). Importantly, both the products of SCD1 activity and its direct impact on tumorigenic pathways have been demonstrated. Based on these findings, SCD1 appears to be a significant player in the development of malignant disease and may be a promising target for anticancer therapy. Numerous chemical compounds that exert inhibitory effects on SCD1 have been developed and preclinically tested. The present review summarizes our current knowledge of the ways in which SCD1 contributes to the progression of cancer and discusses opportunities and challenges of using SCD1 inhibitors for the treatment of cancer. -

Cells and at Immune-Privileged Sites Dendritic Inhibitor 9, Is Mainly
The Granzyme B Inhibitor, Protease Inhibitor 9, Is Mainly Expressed by Dendritic Cells and at Immune-Privileged Sites1 Bellinda A. Bladergroen,* Merel C. M. Strik,† Niels Bovenschen,* Oskar van Berkum,* George L. Scheffer,* Chris J. L. M. Meijer,* C. Erik Hack,†‡ and J. Alain Kummer2* Granzyme B is released from CTLs and NK cells and an important mediator of CTL/NK-induced apoptosis in target cells. The human intracellular serpin proteinase inhibitor (PI)9 is the only human protein able to inhibit the activity of granzyme B. As a first step to elucidate the physiological role of PI9, PI9 protein expression in various human tissues was studied. A mAb directed against human PI9 was developed, which specifically stained PI9-transfected COS-7 cells, and was used for immunohistochem- istry. Both in primary lymphoid organs and in inflammatory infiltrates, PI9 was present in different subsets of dendritic cells. Also T-lymphocytes in primary and organ-associated lymphoid tissues were PI9 positive. Endothelial cells of small vessels in most organs tested as well as the endothelial layer of large veins and arteries showed strong PI9 staining. Surprisingly, high PI9 protein expression was also found at immune-privileged sites like the placenta, the testis, the ovary, and the eye. These data fit with the hypothesis that PI9 is expressed at sites where degranulation of CTL or NK cells is potentially deleterious. The Journal of Immunology, 2001, 166: 0000–0000. ytotoxic cells such as NK cells and CTL form an impor- phocytes, and Fas-associated death domain-like IL-1-converting tant line of defense against virally infected cells and tu- enzyme-inhibitory protein (3), which directly inhibits the Fas-me- C mor cells.