Direct Induction of Gj-Specific Transcripts Following Reactivation of the Cdc28 Kinase in the Absence of De Novo Protein Synthesis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Gene Symbol Gene Description ACVR1B Activin a Receptor, Type IB
Table S1. Kinase clones included in human kinase cDNA library for yeast two-hybrid screening Gene Symbol Gene Description ACVR1B activin A receptor, type IB ADCK2 aarF domain containing kinase 2 ADCK4 aarF domain containing kinase 4 AGK multiple substrate lipid kinase;MULK AK1 adenylate kinase 1 AK3 adenylate kinase 3 like 1 AK3L1 adenylate kinase 3 ALDH18A1 aldehyde dehydrogenase 18 family, member A1;ALDH18A1 ALK anaplastic lymphoma kinase (Ki-1) ALPK1 alpha-kinase 1 ALPK2 alpha-kinase 2 AMHR2 anti-Mullerian hormone receptor, type II ARAF v-raf murine sarcoma 3611 viral oncogene homolog 1 ARSG arylsulfatase G;ARSG AURKB aurora kinase B AURKC aurora kinase C BCKDK branched chain alpha-ketoacid dehydrogenase kinase BMPR1A bone morphogenetic protein receptor, type IA BMPR2 bone morphogenetic protein receptor, type II (serine/threonine kinase) BRAF v-raf murine sarcoma viral oncogene homolog B1 BRD3 bromodomain containing 3 BRD4 bromodomain containing 4 BTK Bruton agammaglobulinemia tyrosine kinase BUB1 BUB1 budding uninhibited by benzimidazoles 1 homolog (yeast) BUB1B BUB1 budding uninhibited by benzimidazoles 1 homolog beta (yeast) C9orf98 chromosome 9 open reading frame 98;C9orf98 CABC1 chaperone, ABC1 activity of bc1 complex like (S. pombe) CALM1 calmodulin 1 (phosphorylase kinase, delta) CALM2 calmodulin 2 (phosphorylase kinase, delta) CALM3 calmodulin 3 (phosphorylase kinase, delta) CAMK1 calcium/calmodulin-dependent protein kinase I CAMK2A calcium/calmodulin-dependent protein kinase (CaM kinase) II alpha CAMK2B calcium/calmodulin-dependent -

Enzymatic Manufacture of Deoxythymidine-5'-Triphosphate with Permeable Intact Cells of E. Coli Coexpressing Thymidylate Kinase
J. Microbiol. Biotechnol. (2015), 25(12), 2034–2042 http://dx.doi.org/10.4014/jmb.1506.06020 Research Article Review jmb Enzymatic Manufacture of Deoxythymidine-5’-Triphosphate with Permeable Intact Cells of E. coli Coexpressing Thymidylate Kinase and Acetate Kinase Jiao Zhang, Yahui Qian, Qingbao Ding*, and Ling Ou* State Key Laboratory of Bioreactor Engineering, East China University of Science and Technology, Shanghai 200237, P.R. China Received: June 8, 2015 Revised: July 26, 2015 A one-pot process of enzymatic synthesis of deoxythymidine-5’-triphosphate (5’-dTTP) Accepted: September 11, 2015 employing whole cells of recombinant Escherichia coli coexpressing thymidylate kinase (TMKase) and acetate kinase (ACKase) was developed. Genes tmk and ack from E. coli were First published online cloned and inserted into pET28a(+), and then transduced into E. coli BL21 (DE3) to form September 15, 2015 recombinant strain pTA in which TMKase and ACKase were simultaneously overexpressed. It *Corresponding authors was found that the relative residual specific activities of TMKase and ACKase, in pTA Q.D. pretreated with 20 mM ethylene diamine tetraacetic acid (EDTA) at 25oC for 30 min, were 94% Phone: +86-21-64250676; Fax: +86-21-64250676; and 96%, respectively. The yield of 5’-dTTP reached above 94% from 5 mM deoxythymidine E-mail: [email protected] 5’-monophosphate (5’-dTMP) and 15 mM acetyl phosphate catalyzed with intact cells of pTA L.O. Phone: +86-21-64253257; pretreated with EDTA. The process was so effective that only 0.125 mM adenosine-5’- Fax: +86-21-64253257; triphosphate was sufficient to deliver the phosphate group from acetyl phosphate to dTMP E-mail: [email protected] and dTDP. -

Table S1. List of Oligonucleotide Primers Used
Table S1. List of oligonucleotide primers used. Cla4 LF-5' GTAGGATCCGCTCTGTCAAGCCTCCGACC M629Arev CCTCCCTCCATGTACTCcgcGATGACCCAgAGCTCGTTG M629Afwd CAACGAGCTcTGGGTCATCgcgGAGTACATGGAGGGAGG LF-3' GTAGGCCATCTAGGCCGCAATCTCGTCAAGTAAAGTCG RF-5' GTAGGCCTGAGTGGCCCGAGATTGCAACGTGTAACC RF-3' GTAGGATCCCGTACGCTGCGATCGCTTGC Ukc1 LF-5' GCAATATTATGTCTACTTTGAGCG M398Arev CCGCCGGGCAAgAAtTCcgcGAGAAGGTACAGATACGc M398Afwd gCGTATCTGTACCTTCTCgcgGAaTTcTTGCCCGGCGG LF-3' GAGGCCATCTAGGCCATTTACGATGGCAGACAAAGG RF-5' GTGGCCTGAGTGGCCATTGGTTTGGGCGAATGGC RF-3' GCAATATTCGTACGTCAACAGCGCG Nrc2 LF-5' GCAATATTTCGAAAAGGGTCGTTCC M454Grev GCCACCCATGCAGTAcTCgccGCAGAGGTAGAGGTAATC M454Gfwd GATTACCTCTACCTCTGCggcGAgTACTGCATGGGTGGC LF-3' GAGGCCATCTAGGCCGACGAGTGAAGCTTTCGAGCG RF-5' GAGGCCTGAGTGGCCTAAGCATCTTGGCTTCTGC RF-3' GCAATATTCGGTCAACGCTTTTCAGATACC Ipl1 LF-5' GTCAATATTCTACTTTGTGAAGACGCTGC M629Arev GCTCCCCACGACCAGCgAATTCGATagcGAGGAAGACTCGGCCCTCATC M629Afwd GATGAGGGCCGAGTCTTCCTCgctATCGAATTcGCTGGTCGTGGGGAGC LF-3' TGAGGCCATCTAGGCCGGTGCCTTAGATTCCGTATAGC RF-5' CATGGCCTGAGTGGCCGATTCTTCTTCTGTCATCGAC RF-3' GACAATATTGCTGACCTTGTCTACTTGG Ire1 LF-5' GCAATATTAAAGCACAACTCAACGC D1014Arev CCGTAGCCAAGCACCTCGgCCGAtATcGTGAGCGAAG D1014Afwd CTTCGCTCACgATaTCGGcCGAGGTGCTTGGCTACGG LF-3' GAGGCCATCTAGGCCAACTGGGCAAAGGAGATGGA RF-5' GAGGCCTGAGTGGCCGTGCGCCTGTGTATCTCTTTG RF-3' GCAATATTGGCCATCTGAGGGCTGAC Kin28 LF-5' GACAATATTCATCTTTCACCCTTCCAAAG L94Arev TGATGAGTGCTTCTAGATTGGTGTCggcGAAcTCgAGCACCAGGTTG L94Afwd CAACCTGGTGCTcGAgTTCgccGACACCAATCTAGAAGCACTCATCA LF-3' TGAGGCCATCTAGGCCCACAGAGATCCGCTTTAATGC RF-5' CATGGCCTGAGTGGCCAGGGCTAGTACGACCTCG -

Overexpression of DNA Polymerase @Jsensitizes Mammalian Cells to 2',3'
ICANCERRESEARCH57.I10-I 16,JanuaryI. I997J Overexpression of DNA Polymerase @JSensitizesMammalian Cells to 2',3'-Deoxycytidine and 3'-Azido-3' .deoxythymidine' Khalil Bouayadi, Jean-Sébastien Hoffmann, Pascal Fons, Michele Tiraby, Jean-Paul Reynes, and Christophe Cazaux2 Laboratoire de Microbiologic et de Génétique,UniversitéPaul Sabatier, 118 route de Narbonne, 31062 Toulouse cédex(K. B., P. F., C. C.]; Institut de Pharmacologic et Biologic Structurale. Centre National de Ia Recherche ScienujIque, UPR 9062, 205 route de Narbonne, 31077 Toulouse cédexfl. S. H.!; and Laboratoire CAYLA, ZI. Montaudran, 5 rue J. Rodier, 3/400 Toulouse (M. T., J. P. R.J ABSTRACT nator AZT-MP was also observed (10, 11). In vivo, AZT-MP is incorporated into cellular DNA (12), and it has been suggested that Mammalian DNA polymerase @3is a DNA repair enzyme expressed polymerase 13may play a role in this process (1 1). @ constitutively at a low level. In vitro, purified DNA polymerase (Pol) incorporates the nucleotide analogues 2'-3' deoxycytidine (ddC)-triphos AZT and ddC are antiviral agents currently used in the treatment phate and 3'-azido-3'-deoxythymidine (AZT)-triphosphate Into DNA, of AIDS. These chemotherapeutic drugs target HIV reverse tran causing chain termination. We have tested the possibility ofenhancing the scriptase which incorporates them into HIV DNA, terminating cytotoxicity of these chain terminators against mammalian cells by in DNA polymerization (13). In this study, we hypothesized that creasing the level of Pol (I. Chinese hamster ovary AA8 and murine overexpression of Pol f3, a cellular target of ddC and AZT, might melanoma B16 cell lines were stably transfected with rat pol fi cDNA render these analogues effective also against tumor cells. -
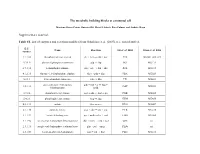
The Metabolic Building Blocks of a Minimal Cell Supplementary
The metabolic building blocks of a minimal cell Mariana Reyes-Prieto, Rosario Gil, Mercè Llabrés, Pere Palmer and Andrés Moya Supplementary material. Table S1. List of enzymes and reactions modified from Gabaldon et. al. (2007). n.i.: non identified. E.C. Name Reaction Gil et. al. 2004 Glass et. al. 2006 number 2.7.1.69 phosphotransferase system glc + pep → g6p + pyr PTS MG041, 069, 429 5.3.1.9 glucose-6-phosphate isomerase g6p ↔ f6p PGI MG111 2.7.1.11 6-phosphofructokinase f6p + atp → fbp + adp PFK MG215 4.1.2.13 fructose-1,6-bisphosphate aldolase fbp ↔ gdp + dhp FBA MG023 5.3.1.1 triose-phosphate isomerase gdp ↔ dhp TPI MG431 glyceraldehyde-3-phosphate gdp + nad + p ↔ bpg + 1.2.1.12 GAP MG301 dehydrogenase nadh 2.7.2.3 phosphoglycerate kinase bpg + adp ↔ 3pg + atp PGK MG300 5.4.2.1 phosphoglycerate mutase 3pg ↔ 2pg GPM MG430 4.2.1.11 enolase 2pg ↔ pep ENO MG407 2.7.1.40 pyruvate kinase pep + adp → pyr + atp PYK MG216 1.1.1.27 lactate dehydrogenase pyr + nadh ↔ lac + nad LDH MG460 1.1.1.94 sn-glycerol-3-phosphate dehydrogenase dhp + nadh → g3p + nad GPS n.i. 2.3.1.15 sn-glycerol-3-phosphate acyltransferase g3p + pal → mag PLSb n.i. 2.3.1.51 1-acyl-sn-glycerol-3-phosphate mag + pal → dag PLSc MG212 acyltransferase 2.7.7.41 phosphatidate cytidyltransferase dag + ctp → cdp-dag + pp CDS MG437 cdp-dag + ser → pser + 2.7.8.8 phosphatidylserine synthase PSS n.i. cmp 4.1.1.65 phosphatidylserine decarboxylase pser → peta PSD n.i. -

A Review of Isozymes in Cancer1
Cancer Research VOLUME31 NOVEMBER 1971 NUMBER11 [CANCER RESEARCH 31, 1523-1542, November 1971] A Review of Isozymes in Cancer1 Wayne E. Criss Department of Obstetrics and Gynecology, University of Florida College of Medicine, Gainesville, Florida 32601 TABLE OF CONTENTS postulated role for that particular isozymic system in cellular metabolism. Summary 1523 Introduction 1523 Normal enzyme differentiation 1523 INTRODUCTION Tumor enzyme differentiation 1524 Isozymes 1524 Normal Enzyme Differentiation DNA polymerase 1524 Enzyme differentiation is the process whereby, during the Hexokinase 1525 Fructose 1,6-diphosphatase 1525 development of an organ in an animal, the organ acquires the quantitative and qualitative adult enzyme patterns (122). Key Aldolase 1526 pathway enzymes in several metabolic processes have been Pyruvate kinase 1527 found to undergo enzymatic differentiation. The enzymes Láclatedehydrogenase 1527 Isocitrate dehydrogenase 1527 involved in nitrogen metabolism, and also in urea cycle Malate dehydrogenase 1528 metabolism (180), are tyrosine aminotransferase (123, 151, Glycerol phosphate dehydrogenase 1529 330, 410), tryptophan pyrrolase (261), serine dehydratase Glutaminase 1529 (123, 410), histidine ammonia lyase (11), and aspartate Aspartate aminotransferase 1530 aminotransferase (337, 388). The enzymes involved in nucleic Adenylate kinase 1531 acid metabolism are DNA polymerase (156, 277) and RNase (52). In glycolysis the enzymes are hexokinase-glucokinase Carbamyl phosphate synthetase 1531 Lactose synthetase 1533 (98, 389), galactokinase 30, aldolase (267, 315), pyruvate Discussion 1533 kinase (73, 386), and lactate dehydrogenase (67, 69). In References 1533 mitochondrial oxidation they are NADH oxidase, succinic oxidase, a-glycero-P oxidase, ATPase, cytochrome oxidase, and flavin content (84, 296). In glycogen metabolism the SUMMARY enzymes involved are UDPG pyrophosphorylase and UDPG glucosyltransferase (19). -

Identification and Purification of a Factor That Binds to the Mlu I Cell Cycle Box of Yeast DNA Replication Genes
Proc. Nati. Acad. Sci. USA Vol. 88, pp. 7155-7159, August 1991 Biochemistry Identification and purification of a factor that binds to the Mlu I cell cycle box of yeast DNA replication genes (Saccharomyces cerevisiae/G, phase/S phase/periodic transcription/DNA polymerase a) RATI VERMA, ARDEM PATAPOUTIAN, COLIN B. GORDON*, AND JUDITH L. CAMPBELL Braun Laboratories, California Institute of Technology, Pasadena, CA 91125 Communicated by John D. Roberts, May 15, 1991 ABSTRACT In Saccharomyces cerevisiae, the genes encod- that are a mixture of the sequences adjacent to both sites in ing at least 10 enzymes involved in DNA replication are the natural promoter confers periodicity (18). The Mlu I site periodically expressed in the late GI and S phases of the cell is necessary for the periodic expression of the CDC21 and cycle. All of these genes have one copy or more of the sequence CDC9 promoters as well (16, 17), suggesting coordinate ACGCGT, which conforms to the recognition site for the Mlu regulation of the entire set of periodic replication genes I restriction endonuclease. For the CDC21, CDC9, and POLl through Mlu I sites. We have designated the UAS that is genes, the Mlu I site has been shown to be absolutely required involved the Mlu I cell cycle box (MCB). for periodic transcription. Using nuclear extracts fractionated The MCB is a potential target for a trans-acting factor that by conventional and oligonucleotide affinity chromatography, responds either directly or indirectly to Start. Identification we have purified a 17-kDa protein that recognizes the Ml4 I of such a factor in an organism such as yeast should facilitate motif. -

Monitoring of Successive Phosphorylations of Thymidine
Monitoring of successive phosphorylations of thymidine using free and immobilized human nucleoside/nucleotide kinases by Flow Injection Analysis with High-Resolution Mass Spectrometry Justine Ferey, David da Silva, Cyril Colas, Reine Nehmé, Pierre Lafite, Vincent Roy, Philippe Morin, Richard Daniellou, Luigi Agrofoglio, Benoît Maunit To cite this version: Justine Ferey, David da Silva, Cyril Colas, Reine Nehmé, Pierre Lafite, et al.. Monitoring of successive phosphorylations of thymidine using free and immobilized human nucleoside/nucleotide kinases by Flow Injection Analysis with High-Resolution Mass Spectrometry. Analytica Chimica Acta, Elsevier Masson, 2019, 1049, pp.115-122. 10.1016/j.aca.2018.10.032. hal-02090335 HAL Id: hal-02090335 https://hal.archives-ouvertes.fr/hal-02090335 Submitted on 29 Nov 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Monitoring of successive phosphorylations of thymidine using free and immobilized human nucleoside/nucleotide kinases by Flow Injection Analysis with High-Resolution Mass Spectrometry Justine Ferey1, David Da Silva*1, Cyril Colas1,2, Reine Nehmé*1, Pierre Lafite1, Vincent Roy1, Philippe Morin1, Richard Daniellou1, Luigi Agrofoglio1, Benoît Maunit1. 1 Univ. Orléans, CNRS, ICOA, UMR 7311, F-45067 Orléans, France. -

Supplementary Table 1
Up-regulated accession # Development M93275 ADFP, adipose differentiation related protein D43694 MATH-1, homolog of atonal 1 M64068 Bmi-1, zinc finger protein AW124785 Midnolin, midbrain nucleolar protein AI843178 Cla3, Cerebellar ataxia 3 D10712 Nedd1, Neural precursor cell expressed, developmentally down-regulated gene 1 AB011678 Doublecortin, for neurogenesis M57683 mPDGF-alpha-R, PDGF alpha receptor U41626 DSS1, deleted in split hand/split foot 1 homolog (Dss1), for limb development AB010833 PTCH2, patched 2, Mouse homolog of yeast CDC46 NP_034226 Ebf3, early B-cell factor 3 AI846695 Qk, Quaking U63386 Edr1 Early development regulator 1 (homolog of polyhomeotic 1), Mph1 AI043016 Rnf2, Ring finger protein 2 X69942 Enhancer-trap-locus 1, for transcription regulation AF100694 Ruvbl1, Ruv-B like protein 1, DNA helicase AW123618 Fzd2, Frizzled homolog 2 U88566 Sfrp1, secreted frizzled related protein 1 AA681520 Geminin-pending, for embryogenesis and morphogenesis U88567 Sfrp2, secreted frizzled related protein 2 AB025922 Gli1, GLI-Kruppel family member 1 AF089721 Smo, Smoothened X99104 Gli2, GLI-Kruppel family member 2 AF009414 SOX11, SRY-box containing gene 11 U61362 Grg1, groucho-related gene 1, Tle1, transducin-like enhancer of split 1 U85614 SRG3, Smarcc1, SWI/SNF related, matrix associated, action dependent regulator of chromatin, subfamily, member 1 M97506 Hen1, helix-loop-helix protein AI837838 Tmeff1, Transmembrane protein with EGF-like and two follistatin-like domains 1 U79748 Madh4, MAD homolog 4, for transcription regulation -

Genome-Wide Analysis of Vaccinia Virus Protein–Protein Interactions
Genome-wide analysis of vaccinia virus protein–protein interactions Stephen McCraith*, Ted Holtzman†, Bernard Moss‡, and Stanley Fields*§¶ *Departments of Genetics and Medicine, Box 357360, †Locke Computer Center, and §Howard Hughes Medical Institute, University of Washington, Seattle, WA 98195; and ‡Laboratory of Viral Diseases, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD 20892-0445 Contributed by Bernard Moss, February 23, 2000 To detect interactions between proteins of vaccinia virus, we Materials and Methods carried out a comprehensive two-hybrid analysis to assay every Construction of pOBD2-20. A new DNA-binding domain vector was pairwise combination. We constructed an array of yeast transfor- generated containing Gal4 residues 1 to 147 and a small portion mants that contained each of the 266 predicted viral ORFs as Gal4 of the activation domain plasmid pOAD (11) that encodes Gal4 activation domain hybrid proteins. The array was individually mated to transformants containing each ORF as a Gal4–DNA- residues 866 to 881 along with a shared multicloning site (Fig. 1A). PCR was used to amplify the Gal4 DNA-binding domain binding domain hybrid, and diploids expressing the two-hybrid Ј reporter gene were identified. Of the Ϸ70,000 combinations, we from the plasmid pCPL2 (12) by using primer P1 5 - Ј found 37 protein–protein interactions, including 28 that were (GTTCTCGTTCCCTTTCTTCCTTGT)-3 located in the previously unknown. In some cases, e.g., late transcription factors, ADH1 promoter region and primer P2 5Ј-CGATCTCTTTTT- both proteins were known to have related roles although there TTGGGTTTGGTGGGGTATCTTCATCATCGAATAGAT- was no prior evidence of physical associations. -

Periodic Expression of Nuclear and Mitochondrial DNA Replication Genes During the Trypanosomatid Cell Cycle
Journal of Cell Science 107, 3515-3520 (1994) 3515 Printed in Great Britain © The Company of Biologists Limited 1994 Periodic expression of nuclear and mitochondrial DNA replication genes during the trypanosomatid cell cycle Sally G. Pasion, Grant W. Brown, Lisa M. Brown and Dan S. Ray* Molecular Biology Institute and Department of Biology, University of California, Los Angeles, California 90024, USA *Author for correspondence SUMMARY In trypanosomatids, DNA replication in the nucleus and in drial topoisomerase, was disrupted by the insertion of a the single mitochondrion (or kinetoplast) initiates nearly NEO drug-resistance cassette was found to express both a simultaneously, suggesting that the DNA synthesis (S) truncated TOP2 mRNA and a truncated topoisomerase phases of the nucleus and the mitochondrion are coordi- polypeptide. The truncated mRNA was also expressed peri- nately regulated. To investigate the basis for the temporal odically coordinate with the expression of the endogenous link between nuclear and mitochondrial DNA synthesis TOP2 mRNA indicating that cis elements necessary for phases the expression of the genes encoding DNA ligase I, periodic expression are contained within cloned sequences. the 51 and 28 kDa subunits of replication protein A, dihy- The expression of both TOP2 and nuclear DNA replication drofolate reductase and the mitochondrial type II topoiso- genes at the G1/S boundary suggests that regulated merase were analyzed during the cell cycle progression of expression of these genes may play a role in coordinating synchronous cultures of Crithidia fasciculata. These DNA nuclear and mitochondrial S phases in trypanosomatids. replication genes were all expressed periodically, with peak mRNA levels occurring just prior to or at the peak of DNA synthesis in the synchronized cultures. -

A Method and Kit for Determination of Thymidine
(19) & (11) EP 1 856 275 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: C12Q 1/48 (2006.01) 12.08.2009 Bulletin 2009/33 (86) International application number: (21) Application number: 06716936.7 PCT/SE2006/000246 (22) Date of filing: 24.02.2006 (87) International publication number: WO 2006/091158 (31.08.2006 Gazette 2006/35) (54) A METHOD AND KIT FOR DETERMINATION OF THYMIDINE KINASE ACTIVITY AND USE THEREOF VERFAHREN UND KIT ZUR BESTIMMUNG VON THYMIDINKINASE-AKTIVITÄT UND VERWENDUNG DAVON MÉTHODE ET TROUSSE PERMETTANT DE DÉTERMINER L’ACTIVITÉ DE LA THYMIDINE KINASE, ET LEUR MODE D’UTILISATION (84) Designated Contracting States: (56) References cited: AT BE BG CH CY CZ DE DK EE ES FI FR GB GR EP-A2- 0 094 923 WO-A1-90/06320 HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI WO-A1-20/04100760 WO-A2-20/04011627 SK TR Designated Extension States: • ARMSTRONG B ET AL: "SELECTIVE INHIBITION AL BA HR MK YU OF THYMIDINE KINASE ISOENZYMES BY (E)- 5-(2-BROMOVINYL)-2-DEOXYURIDINE" (30) Priority: 25.02.2005 SE 0500432 BIOCHEMICAL SOCIETY TRANSACTIONS, COLCHESTER, ESSEX, GB, vol. 18, no. 2, 1990, (43) Date of publication of application: page 270, XP001070196 ISSN: 0300-5127 21.11.2007 Bulletin 2007/47 • OHRVIK ANDERS ET AL: "Sensitive nonradiometric method for determining (73) Proprietor: Rönnerbol International AB thymidine kinase I activity" CLINICAL 752 39 Uppsala (SE) CHEMISTRY, vol. 50, no. 9, September 2004 (2004-09), pages 1597-1606, XP002470247 ISSN: (72) Inventor: GRONOWITZ, Simon 0009-9147 S-752 39 Uppsala (SE) • WICKREMASINGHE R.G.