Molecular Interactions of Antibody Drugs Targeting PD-1, PD-L1, and CTLA-4 in Immuno-Oncology
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Fig. L COMPOSITIONS and METHODS to INHIBIT STEM CELL and PROGENITOR CELL BINDING to LYMPHOID TISSUE and for REGENERATING GERMINAL CENTERS in LYMPHATIC TISSUES
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date Χ 23 February 2012 (23.02.2012) WO 2U12/U24519ft ft A2 (51) International Patent Classification: AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, A61K 31/00 (2006.01) CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (21) International Application Number: HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, PCT/US201 1/048297 KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, (22) International Filing Date: ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, 18 August 201 1 (18.08.201 1) NO, NZ, OM, PE, PG, PH, PL, PT, QA, RO, RS, RU, SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, (25) Filing Language: English TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, (26) Publication Language: English ZW. (30) Priority Data: (84) Designated States (unless otherwise indicated, for every 61/374,943 18 August 2010 (18.08.2010) US kind of regional protection available): ARIPO (BW, GH, 61/441,485 10 February 201 1 (10.02.201 1) US GM, KE, LR, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, 61/449,372 4 March 201 1 (04.03.201 1) US ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, DK, (72) Inventor; and EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, ΓΓ, LT, LU, (71) Applicant : DEISHER, Theresa [US/US]; 1420 Fifth LV, MC, MK, MT, NL, NO, PL, PT, RO, RS, SE, SI, SK, Avenue, Seattle, WA 98101 (US). -

Remarkably Similar CTLA-4 Binding Properties of Therapeutic Ipilimumab and Tremelimumab Antibodies
www.impactjournals.com/oncotarget/ Oncotarget, 2017, Vol. 8, (No. 40), pp: 67129-67139 Research Paper Remarkably similar CTLA-4 binding properties of therapeutic ipilimumab and tremelimumab antibodies Mengnan He1,2,*, Yan Chai1,*, Jianxun Qi1, Catherine W.H. Zhang3, Zhou Tong4, Yi Shi1, Jinghua Yan4, Shuguang Tan1 and George F. Gao1,2 1CAS Key Laboratory of Pathogenic Microbiology and Immunology, Institute of Microbiology, Chinese Academy of Sciences, Beijing 100101, China 2University of Chinese Academy of Sciences, Beijing 100049, China 3ImmuFuCell Biotechnology Co., Ltd., Beijing 100102, China 4CAS Key Laboratory of Microbial Physiological and Metabolic Engineering, Institute of Microbiology, Chinese Academy of Sciences, Beijing 100101, China *These authors have contributed equally to this work Correspondence to: George F. Gao, email: [email protected] Shuguang Tan, email: [email protected] Keywords: ipilimumab, CTLA-4, complex structure, tremelimumab Received: April 04, 2017 Accepted: April 21, 2017 Published: May 19, 2017 Copyright: He et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License 3.0 (CC BY 3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. ABSTRACT Monoclonal antibody based immune checkpoint blockade therapies have achieved clinical successes in management of malignant tumors. As the first monoclonal antibody targeting immune checkpoint molecules entered into clinics, the molecular basis of ipilimumab-based anti-CTLA-4 blockade has not yet been fully understood. In the present study, we report the complex structure of ipilimumab and CTLA-4. The complex structure showed similar contributions from VH and VL of ipilimumab in binding to CTLA-4 front β-sheet strands. -

ADC Therapeutics SA (Exact Name of Registrant As Specified in Its Charter)
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 6-K REPORT OF FOREIGN PRIVATE ISSUER PURSUANT TO RULE 13a-16 OR 15d-16 UNDER THE SECURITIES EXCHANGE ACT OF 1934 For the month of July, 2020. Commission File Number: 001-39071 ADC Therapeutics SA (Exact name of registrant as specified in its charter) Biopôle Route de la Corniche 3B 1066 Epalinges Switzerland (Address of principal executive office) Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F: Form 20-F ☒ Form 40-F Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ☐ Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ☐ SIGNATURE Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized. ADC Therapeutics SA Date: July 6, 2020 By: /s/ Dominique Graz Name: Dominique Graz Title: General Counsel EXHIBIT INDEX Exhibit No. Description 99.1 Press release dated July 6, 2020 Exhibit 99.1 ADC Therapeutics Announces U.S. Food and Drug Administration Has Lifted Partial Clinical Hold on Pivotal Phase 2 Clinical Trial of Camidanlumab Tesirine Lausanne, Switzerland — July 6, 2020 — ADC Therapeutics SA (NYSE:ADCT), a clinical-stage oncology-focused biotechnology company leading the development and commercialization of next-generation antibody drug conjugates (ADCs) with highly potent and targeted pyrrolobenzodiazepine (PBD) dimer technology, today announced that the U.S. -

Comparative Effectiveness of Pembrolizumab Vs. Nivolumab In
www.nature.com/scientificreports OPEN Comparative efectiveness of pembrolizumab vs. nivolumab in patients with recurrent or advanced NSCLC Pengfei Cui1,2,4, Ruixin Li2,4, Ziwei Huang3,2,4, Zhaozhen Wu3,2, Haitao Tao2, Sujie Zhang2 & Yi Hu1,2,3* The efcacies of pembrolizumab and nivolumab have never been directly compared in a real-world study. Therefore, we sought to retrospectively evaluate the objective response rate (ORR) and the progression-free survival (PFS) of patients with recurrent or advanced non-small cell lung cancer (NSCLC) in a real-world setting. This study included patients with recurrent or advanced NSCLC diagnosed between September 1, 2015 and August 31, 2019, who were treated with programmed cell death 1 (PD-1) inhibitors at the Cancer Center of the Chinese People’s Liberation Army. PFS was estimated for each treatment group using Kaplan–Meier curves and log-rank tests. The multivariate analysis of PFS was performed with Cox proportional hazards regression models. A total of 255 patients with advanced or recurrent NSCLC treated with PD-1 inhibitors were identifed. The ORR was signifcantly higher in the pembrolizumab group than in the nivolumab group, while PFS was not signifcantly diferent between the two groups. Subgroup analysis showed that the ORR was signifcantly higher for pembrolizumab than for nivolumab in patients in the frst-line therapy subgroup and in those in the combination therapy as frst-line therapy subgroup. Survival analysis of patients receiving combination therapy as second- or further-line therapy showed that nivolumab had better efcacy than pembrolizumab. However, the multivariate analysis revealed no signifcant diference in PFS between patients treated with pembrolizumab and those treated with nivolumab regardless of the subgroup. -

Cutaneous Adverse Effects of Biologic Medications
REVIEW CME MOC Selena R. Pasadyn, BA Daniel Knabel, MD Anthony P. Fernandez, MD, PhD Christine B. Warren, MD, MS Cleveland Clinic Lerner College Department of Pathology Co-Medical Director of Continuing Medical Education; Department of Dermatology, Cleveland Clinic; of Medicine of Case Western and Department of Dermatology, W.D. Steck Chair of Clinical Dermatology; Director of Clinical Assistant Professor, Cleveland Clinic Reserve University, Cleveland, OH Cleveland Clinic Medical and Inpatient Dermatology; Departments of Lerner College of Medicine of Case Western Dermatology and Pathology, Cleveland Clinic; Assistant Reserve University, Cleveland, OH Clinical Professor, Cleveland Clinic Lerner College of Medicine of Case Western Reserve University, Cleveland, OH Cutaneous adverse effects of biologic medications ABSTRACT iologic therapy encompasses an expo- B nentially expanding arena of medicine. Biologic therapies have become widely used but often As the name implies, biologic therapies are de- cause cutaneous adverse effects. The authors discuss the rived from living organisms and consist largely cutaneous adverse effects of tumor necrosis factor (TNF) of proteins, sugars, and nucleic acids. A clas- alpha inhibitors, epidermal growth factor receptor (EGFR) sic example of an early biologic medication is inhibitors, small-molecule tyrosine kinase inhibitors insulin. These therapies have revolutionized (TKIs), and cell surface-targeted monoclonal antibodies, medicine and offer targeted therapy for an including how to manage these reactions -

Chelsea & Westminster Healthcare Trust Drugs Committee
Chelsea and Westminster Hospital NHS Foundation Trust Trust Medicines Group Summary of Main Points from the Meeting held on Monday 12th December 2016 2. Minutes and Summary Notes from last meeting The Minutes and Summary notes from the November 2016 Medicines Group meeting were approved and will be circulated. 3. Matters Arising The Group noted the matters arising from the previous meeting. 4. Business to be transacted by the Medicines Group 4.1 Formulary Applications Ex-panel Deferasirox (Exjade®) 90mg, 180mg and 360mg Tablets (Novartis) Exjade® film coated tablets have been introduced in November 2016. These will take the place of the dispersible tabs which will be phased out and no longer available from June 2017. Costs are equivalent to the dispersible tablets. The new film coated tablets have a number of advantages over the dispersible tablets including: - Can be swallowed whole - Lactose free - No food restrictions - No mixing/preparation required. Decision: Approved DuoResp Spiromax® 160 micrograms / 4.5 micrograms (Budesonide & Formoterol) Inhalation Powder (Teva) Request by the Respiratory Team for the management of asthma in line with the BTS guidelines where a ® combination of an inhaled long acting B2 agonist and corticosteroid is indicated. Adding DuoResp Spiromax to the formulary will provide an increased choice of devices available to prescribe. Symbicort Turbohaler® is already included on the formulary. DuoResp Spiromax® is more cost-effective than Symbicort Turbohaler® and is currently included on the NWLIF and GPs have already started to prescribe it. Decision: Approved Sodium Valpoate (Epilim Chronosphere®) 50mg and 100mg Modified Release Granules (Sanofi) Requested by the Paediatric Neurology Team for paediatric patients requiring sodium valpoate in very low doses. -

New Biological Therapies: Introduction to the Basis of the Risk of Infection
New biological therapies: introduction to the basis of the risk of infection Mario FERNÁNDEZ RUIZ, MD, PhD Unit of Infectious Diseases Hospital Universitario “12 de Octubre”, Madrid ESCMIDInstituto de Investigación eLibraryHospital “12 de Octubre” (i+12) © by author Transparency Declaration Over the last 24 months I have received honoraria for talks on behalf of • Astellas Pharma • Gillead Sciences • Roche • Sanofi • Qiagen Infections and biologicals: a real concern? (two-hour symposium): New biological therapies: introduction to the ESCMIDbasis of the risk of infection eLibrary © by author Paul Ehrlich (1854-1915) • “side-chain” theory (1897) • receptor-ligand concept (1900) • “magic bullet” theory • foundation for specific chemotherapy (1906) • Nobel Prize in Physiology and Medicine (1908) (together with Metchnikoff) Infections and biologicals: a real concern? (two-hour symposium): New biological therapies: introduction to the ESCMIDbasis of the risk of infection eLibrary © by author 1981: B-1 antibody (tositumomab) anti-CD20 monoclonal antibody 1997: FDA approval of rituximab for the treatment of relapsed or refractory CD20-positive NHL 2001: FDA approval of imatinib for the treatment of chronic myelogenous leukemia Infections and biologicals: a real concern? (two-hour symposium): New biological therapies: introduction to the ESCMIDbasis of the risk of infection eLibrary © by author Functional classification of targeted (biological) agents • Agents targeting soluble immune effector molecules • Agents targeting cell surface receptors -

Actemra® (Tocilizumab)
Actemra® (tocilizumab) (Intravenous) Document Number: MODA-0002 Last Review Date: 10/26/2020 Date of Origin: 09/21/2010 Dates Reviewed: 12/2010, 03/2011, 05/2011, 06/2011, 09/2011, 12/2011, 03/2012, 06/2012, 09/2012, 09/2012, 11/2012, 12/2012, 03/2013, 06/2013, 09/2013, 11/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 05/2015, 09/2015, 12/0215, 03/2016, 06/2016, 09/2016, 12/2016, 03/2017, 05/2017, 09/2017, 12/2017, 03/2018, 06/2018, 10/2018, 10/2019, 10/2020, 11/2020 I. Length of Authorization Coverage will be provided as follows: o Castleman’s Disease: 4 months and may be renewed o Cytokine Release Syndrome: 4 doses only and may not be renewed o Immune Checkpoint Inhibitor related arthritis: 1 dose and may not be renewed o All other indications: 6 months and may be renewed. II. Dosing Limits A. Quantity Limit (max daily dose) [NDC Unit]: o Actemra 80 mg/4 mL vial: 1 vial per 14 days o Actemra 200 mg/10 mL vial: 1 vial per 14 days o Actemra 400 mg/20 mL vial: 2 vials per 14 days B. Max Units (per dose and over time) [HCPCS Unit]: Diagnosis Billable Units Interval (days) Rheumatoid Arthritis & Polyarticular Juvenile Idiopathic 800 28 Arthritis, NMOSD Systemic Juvenile Idiopathic Arthritis, Castleman’s Disease (NHL) & Acute Graft Versus Host Disease 800 14 (aGVHD) Cytokine Release Syndrome (CRS) 3200 1 course of therapy only Immune Checkpoint Inhibitor related arthritis 800 1 course of therapy only III. -

ESCMID Online Lecture Library @ by Author
The diverse monoclonal antibodies in immunology and medical oncology: relevance for infectious diseases Dra. Isabel Ruiz Camps ESCMIDHospital Online Universitari Lecture Vall d’Hebron Library @ by authorBarcelona Disclosure • Astellas • Gilead Sciences • MSD • Novartis • Pfizer ESCMID Online Lecture Library @ by author A huge topic for such a ESCMIDshort Online time Lecture Library @ by author natalizumab antiTNF antiCD20: rituximab, obinutuzumab, ofatumumab gemtuzumab (antiCD33) alemtuzumab (antiCD52) daratumumab (antiCD38) Inotuzumab (antiCD22) Brentuximab (CD30) Seculinumab (anti IL-17) Tocilizumab (antiIL6) PI3K inhibitors PARP inh: olaparib, Guselkumab (anti IL12/23) rucaparib (copanlisib, more ... Roxulitinib, tofacitinib (JAK inh) Urelumab (CD137 R) Immunotherapy: ipilimumab TK inhibitors : Imatinib, dasatinib, Belimumab (antiBAFF) (CTLA-4), tremelimumab (CTLA-4), masitinib, bosutinib, nilotinib, fostamatinib nivolimumab (PD1/PDL1), (spleen), ibrutinib (BTK), alisertib (ATK), Pembrolizumab (PD1), more...... afatinib Cabozantinib: MET, RET, VEGFR2 HER2/neu:ESCMID trastuzumab, lapatinib Online VEGFR: bevacizumabLecture, LibrarymTOR: temsirolimus sorafenib, sunitinib EGFR: cetuximab, panitumumab, MAPK inh: dabrafenib, vemurafenib erlotinib, gefitinib Selinexor (XPO1 antagonist) @ by author Trametinib (MEK inh) Index • Background • Biological therapies for immunological diseases and risk of infection • Biological therapies for cancer – Target pathways – Risk of infection • Prevention ESCMID Online Lecture Library @ by author What are -

Actemra (Tocilizumab) NON-ONCOLOGY POLICY (Intravenous) Department: PHA
Policy Title: Actemra (tocilizumab) NON-ONCOLOGY POLICY (Intravenous) Department: PHA Effective Date: 01/01/2020 Review Date: 09/25/2019, 12/18/2019, 1/22/2020, 8/3/2020 Revision Date: 09/25/2019, 12/18/2019, 1/22/2020, 8/3/2020 Purpose: To support safe, effective and appropriate use of Actemra (tocilizumab). Scope: Medicaid, Commerical, Medicare-Medicaid Plan (MMP) Policy Statement: Actemra (tocilizumab) is covered under the Medical Benefit when used within the following guidelines for non-oncology indications. Use outside of these guidelines may result in non-payment unless approved under an exception process. For oncology indications, please refer to NHPRI Oncology Policy Procedure: Coverage of Actemra (tocilizumab) will be reviewed prospectively via the prior authorization process based on criteria below. Initial Criteria: Patient has been evaluated and screened for the presence of latent TB infection prior to initiating treatment; AND Patient does not have an active infection, including clinically important localized infections; AND Must not be administered concurrently with live vaccines; AND Patient is not on concurrent treatment with another TNF-inhibitor, biologic response modifier or other non-biologic agent (i.e., apremilast, tofacitinib, baricitinib); MMP members who have previously received this medication within the past 365 days are not subject to Step Therapy Requirements Rheumatoid Arthritis Patient is 18 years or older; AND Physician has assessed baseline disease severity utilizing an objective measure/tool; AND -

On the Horizon: Immuno-Oncology (I-O) Combinations
Immuno-Oncology (I-O) Combinations • Jeffrey A. Sosman, MD • Robert H. Lurie Comprehensive Cancer Center of Northwestern University The Cancer–Immunity Cycle Daniel Chen and Ira Mellman Immunity, Volume 39, Issue 1, 2013, 1 - 10 The Cancer–Immunity Cycle Daniel Chen and Ira Mellman Immunity, Volume 39, Issue 1, 2013, 1 - 10 Stimulatory and Inhibitory Factors in the Cancer-Immunity Cycle Each step of the Cancer-Immunity Cycle requires the coordination of numerous factors, both stimulatory and inhibitory in nature. Stimulatory factors shown in green promote immunity, ... Where will Improvements come from? • Combinations: – Based on Template: anti-PD-1/PD-L1 or with anti-PD- 1/anti-CTLA-4 • Block other co-inhibitory: LAG3, TIM3, KIR, VISTA • Activate co-stimulatory: 4-1BB, OX-40, GITR, CD27, ICOS • Block inhibitory molecules- IDOi, TGFbi, CSF1Ri, anti-IL-6 or anti- IL-10 • Effect trafficking- anti-VEGF, CCL5, CXCR4i • Vaccines- TVEC- oncolytic virus, Neoantigen, other cellular • Adoptive Cellular therapy- TIL, CAR-T cells, TCR T-cells Where will Improvements come from? • Combinations: – Based on Template: anti-PD-1/PD-L1 or with anti-PD- 1/anti-CTLA-4 • Signal Inhibition, BRAF directed (BRAFi+MEKi), MEKi, PI3K inhibition (PTEN effects) • Cytokines- IL-2, IFN a,b,g,, Directed cytokines (FAP-IL-2v or CEA-IL-2v) • Epigenetic modulation- gene expression and EVR expression • Microbiome modification- fecal transplants • Chemotherapy other cytotoxics • Localized Irradiation SBRT, SRS T cells in Tumors Express Multiple Immunoinhibitory Receptors -
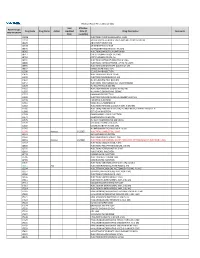
2021 Prior Authorization List Part B Appendix a (PDF)
Medicare Part B PA List Effective 2021 Last Effective Part B Drugs: Drug Code Drug Name Action Updated Date (if Drug Description Comments STEP THERAPY Date available) C9050 INJECTION, EMAPALUMAB-LZSG, 1 MG C9122 MOMETASONE FUROATE SINUS IMPLANT 10 MCG SINUVA J0129 ABATACEPT INJECTION J0178 AFLIBERCEPT INJECTION J0570 BUPRENORPHINE IMPLANT 74.2MG J0585 INJECTION,ONABOTULINUMTOXINA J0717 CERTOLIZUMAB PEGOL INJ 1MG J0718 CERTOLIZUMAB PEGOL INJ J0791 INJECTION CRIZANLIZUMAB-TMCA 5 MG J0800 INJECTION, CORTICOTROPIN, UP TO 40 UNITS J0896 INJECTION LUSPATERCEPT-AAMT 0.25 MG J0897 DENOSUMAB INJECTION J1300 ECULIZUMAB INJECTION J1428 INJECTION ETEPLIRSEN 10 MG J1429 INJECTION GOLODIRSEN 10 MG J1442 INJ FILGRASTIM EXCL BIOSIMIL J1447 INJECTION, TBO-FILGRASTIM, 1 MICROGRAM J1459 INJ IVIG PRIVIGEN 500 MG J1555 INJECTION IMMUNE GLOBULIN 100 MG J1556 INJ, IMM GLOB BIVIGAM, 500MG J1557 GAMMAPLEX INJECTION J1558 INJECTION IMMUNE GLOBULIN XEMBIFY 100 MG J1559 HIZENTRA INJECTION J1561 GAMUNEX-C/GAMMAKED J1562 INJECTION; IMMUNE GLOBULIN 10%, 5 GRAMS J1566 INJECTION, IMMUNE GLOBULIN, INTRAVENOUS, LYOPHILIZED (E.G. P J1568 OCTAGAM INJECTION J1569 GAMMAGARD LIQUID INJECTION J1572 FLEBOGAMMA INJECTION J1575 INJ IG/HYALURONIDASE 100 MG IG J1599 IVIG NON-LYOPHILIZED, NOS J1602 GOLIMUMAB FOR IV USE 1MG J1745 INJ INFLIXIMAB EXCL BIOSIMILR 10 MG J1930 Remove 1/1/2021 INJECTION, LANREOTIDE, 1 MG J2323 NATALIZUMAB INJECTION J2350 INJECTION OCRELIZUMAB 1 MG J2353 Remove 1/1/2021 INJECTION, OCTREOTIDE, DEPOT FORM FOR INTRAMUSCULAR INJECTION, 1 MG J2357 INJECTION, OMALIZUMAB,