High-Throughput Screening of Mouse Gene Knockouts Identifies Established and Novel Skeletal Phenotypes
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Cytogenomic SNP Microarray - Fetal ARUP Test Code 2002366 Maternal Contamination Study Fetal Spec Fetal Cells
Patient Report |FINAL Client: Example Client ABC123 Patient: Patient, Example 123 Test Drive Salt Lake City, UT 84108 DOB 2/13/1987 UNITED STATES Gender: Female Patient Identifiers: 01234567890ABCD, 012345 Physician: Doctor, Example Visit Number (FIN): 01234567890ABCD Collection Date: 00/00/0000 00:00 Cytogenomic SNP Microarray - Fetal ARUP test code 2002366 Maternal Contamination Study Fetal Spec Fetal Cells Single fetal genotype present; no maternal cells present. Fetal and maternal samples were tested using STR markers to rule out maternal cell contamination. This result has been reviewed and approved by Maternal Specimen Yes Cytogenomic SNP Microarray - Fetal Abnormal * (Ref Interval: Normal) Test Performed: Cytogenomic SNP Microarray- Fetal (ARRAY FE) Specimen Type: Direct (uncultured) villi Indication for Testing: Patient with 46,XX,t(4;13)(p16.3;q12) (Quest: EN935475D) ----------------------------------------------------------------- ----- RESULT SUMMARY Abnormal Microarray Result (Male) Unbalanced Translocation Involving Chromosomes 4 and 13 Classification: Pathogenic 4p Terminal Deletion (Wolf-Hirschhorn syndrome) Copy number change: 4p16.3p16.2 loss Size: 5.1 Mb 13q Proximal Region Deletion Copy number change: 13q11q12.12 loss Size: 6.1 Mb ----------------------------------------------------------------- ----- RESULT DESCRIPTION This analysis showed a terminal deletion (1 copy present) involving chromosome 4 within 4p16.3p16.2 and a proximal interstitial deletion (1 copy present) involving chromosome 13 within 13q11q12.12. This -

Nuclear and Mitochondrial Genome Defects in Autisms
UC Irvine UC Irvine Previously Published Works Title Nuclear and mitochondrial genome defects in autisms. Permalink https://escholarship.org/uc/item/8vq3278q Journal Annals of the New York Academy of Sciences, 1151(1) ISSN 0077-8923 Authors Smith, Moyra Spence, M Anne Flodman, Pamela Publication Date 2009 DOI 10.1111/j.1749-6632.2008.03571.x License https://creativecommons.org/licenses/by/4.0/ 4.0 Peer reviewed eScholarship.org Powered by the California Digital Library University of California THE YEAR IN HUMAN AND MEDICAL GENETICS 2009 Nuclear and Mitochondrial Genome Defects in Autisms Moyra Smith, M. Anne Spence, and Pamela Flodman Department of Pediatrics, University of California, Irvine, California In this review we will evaluate evidence that altered gene dosage and structure im- pacts neurodevelopment and neural connectivity through deleterious effects on synap- tic structure and function, and evidence that the latter are key contributors to the risk for autism. We will review information on alterations of structure of mitochondrial DNA and abnormal mitochondrial function in autism and indications that interactions of the nuclear and mitochondrial genomes may play a role in autism pathogenesis. In a final section we will present data derived using Affymetrixtm SNP 6.0 microar- ray analysis of DNA of a number of subjects and parents recruited to our autism spectrum disorders project. We include data on two sets of monozygotic twins. Col- lectively these data provide additional evidence of nuclear and mitochondrial genome imbalance in autism and evidence of specific candidate genes in autism. We present data on dosage changes in genes that map on the X chromosomes and the Y chro- mosome. -

Diseasespecific and Inflammationindependent Stromal
Full Length Arthritis & Rheumatism DOI 10.1002/art.37704 Disease-specific and inflammation-independent stromal alterations in spondyloarthritis synovitis Nataliya Yeremenko1,2, Troy Noordenbos1,2, Tineke Cantaert1,3, Melissa van Tok1,2, Marleen van de Sande1, Juan D. Cañete4, Paul P. Tak1,5*, Dominique Baeten1,2 1Department of Clinical Immunology and Rheumatology and 2Department of Experimental Immunology, Academic Medical Center/University of Amsterdam, the Netherlands. 3Department of Immunobiology, Yale University School of Medicine, New Haven, CT, USA. 4Department of Rheumatology, Hospital Clinic de Barcelona and IDIBAPS, Spain. 5Arthrogen B.V., Amsterdam, the Netherlands. *Currently also: GlaxoSmithKline, Stevenage, U.K. Corresponding author: Dominique Baeten, MD, PhD, Department of Clinical Immunology and Rheumatology, F4-105, Academic Medical Center/University of Amsterdam, Meibergdreef 9, 1105 AZ Amsterdam, The Netherlands. E-mail: [email protected] This article has been accepted for publication and undergone full peer review but has not been through the copyediting, typesetting, pagination and proofreading process which may lead to differences between this version and the Version of Record. Please cite this article as an ‘Accepted Article’, doi: 10.1002/art.37704 © 2012 American College of Rheumatology Received: Apr 11, 2012; Revised: Jul 25, 2012; Accepted: Sep 06, 2012 Arthritis & Rheumatism Page 2 of 36 Abstract Objective: The molecular processes driving the distinct patterns of synovial inflammation and tissue remodelling in spondyloarthritis (SpA) versus rheumatoid arthritis (RA) remain largely unknown. Therefore, we aimed to identify novel and unsuspected disease- specific pathways in SpA by a systematic and unbiased synovial gene expression analysis. Methods: Differentially expressed genes were identified by pan-genomic microarray and confirmed by quantitative PCR and immunohistochemistry using synovial tissue biopsies of SpA (n=63), RA (n=28) and gout (n=9) patients. -

Analysis of Gene Expression Data for Gene Ontology
ANALYSIS OF GENE EXPRESSION DATA FOR GENE ONTOLOGY BASED PROTEIN FUNCTION PREDICTION A Thesis Presented to The Graduate Faculty of The University of Akron In Partial Fulfillment of the Requirements for the Degree Master of Science Robert Daniel Macholan May 2011 ANALYSIS OF GENE EXPRESSION DATA FOR GENE ONTOLOGY BASED PROTEIN FUNCTION PREDICTION Robert Daniel Macholan Thesis Approved: Accepted: _______________________________ _______________________________ Advisor Department Chair Dr. Zhong-Hui Duan Dr. Chien-Chung Chan _______________________________ _______________________________ Committee Member Dean of the College Dr. Chien-Chung Chan Dr. Chand K. Midha _______________________________ _______________________________ Committee Member Dean of the Graduate School Dr. Yingcai Xiao Dr. George R. Newkome _______________________________ Date ii ABSTRACT A tremendous increase in genomic data has encouraged biologists to turn to bioinformatics in order to assist in its interpretation and processing. One of the present challenges that need to be overcome in order to understand this data more completely is the development of a reliable method to accurately predict the function of a protein from its genomic information. This study focuses on developing an effective algorithm for protein function prediction. The algorithm is based on proteins that have similar expression patterns. The similarity of the expression data is determined using a novel measure, the slope matrix. The slope matrix introduces a normalized method for the comparison of expression levels throughout a proteome. The algorithm is tested using real microarray gene expression data. Their functions are characterized using gene ontology annotations. The results of the case study indicate the protein function prediction algorithm developed is comparable to the prediction algorithms that are based on the annotations of homologous proteins. -
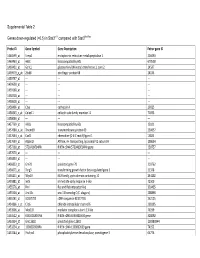
0.5) in Stat3∆/∆ Compared with Stat3flox/Flox
Supplemental Table 2 Genes down-regulated (<0.5) in Stat3∆/∆ compared with Stat3flox/flox Probe ID Gene Symbol Gene Description Entrez gene ID 1460599_at Ermp1 endoplasmic reticulum metallopeptidase 1 226090 1460463_at H60c histocompatibility 60c 670558 1460431_at Gcnt1 glucosaminyl (N-acetyl) transferase 1, core 2 14537 1459979_x_at Zfp68 zinc finger protein 68 24135 1459747_at --- --- --- 1459608_at --- --- --- 1459168_at --- --- --- 1458718_at --- --- --- 1458618_at --- --- --- 1458466_at Ctsa cathepsin A 19025 1458345_s_at Colec11 collectin sub-family member 11 71693 1458046_at --- --- --- 1457769_at H60a histocompatibility 60a 15101 1457680_a_at Tmem69 transmembrane protein 69 230657 1457644_s_at Cxcl1 chemokine (C-X-C motif) ligand 1 14825 1457639_at Atp6v1h ATPase, H+ transporting, lysosomal V1 subunit H 108664 1457260_at 5730409E04Rik RIKEN cDNA 5730409E04Rik gene 230757 1457070_at --- --- --- 1456893_at --- --- --- 1456823_at Gm70 predicted gene 70 210762 1456671_at Tbrg3 transforming growth factor beta regulated gene 3 21378 1456211_at Nlrp10 NLR family, pyrin domain containing 10 244202 1455881_at Ier5l immediate early response 5-like 72500 1455576_at Rinl Ras and Rab interactor-like 320435 1455304_at Unc13c unc-13 homolog C (C. elegans) 208898 1455241_at BC037703 cDNA sequence BC037703 242125 1454866_s_at Clic6 chloride intracellular channel 6 209195 1453906_at Med13l mediator complex subunit 13-like 76199 1453522_at 6530401N04Rik RIKEN cDNA 6530401N04 gene 328092 1453354_at Gm11602 predicted gene 11602 100380944 1453234_at -

Norrin, Frizzled-4, and Lrp5 Signaling in Endothelial Cells Controls a Genetic Program for Retinal Vascularization
Norrin, Frizzled-4, and Lrp5 Signaling in Endothelial Cells Controls a Genetic Program for Retinal Vascularization Xin Ye,1,7 Yanshu Wang,1,4,7 Hugh Cahill,2 Minzhong Yu,5 Tudor C. Badea,1,4 Philip M. Smallwood,1,4 Neal S. Peachey,5,6 and Jeremy Nathans1,2,3,4,* 1Department of Molecular Biology and Genetics 2Department of Neuroscience 3Department of Ophthalmology 4Howard Hughes Medical Institute Johns Hopkins University School of Medicine, Baltimore, MD 21205, USA 5Cole Eye Institute, Cleveland Clinic Foundation, Cleveland, OH 44195, USA 6Research Service, Cleveland VA Medical Center, Cleveland, OH 44106, USA 7These authors contributed equally to this work *Correspondence: [email protected] DOI 10.1016/j.cell.2009.07.047 SUMMARY and structure, multiple cell signaling systems—including the VEGF, Netrin, Ephrin, Notch, and Angiopoietin systems—have Disorders of vascular structure and function play been implicated in coordinating the proliferation, migration, adhe- a central role in a wide variety of CNS diseases. Muta- sion, and differentiation of vascular cells (Adams and Alitalo, 2007). tions in the Frizzled-4 (Fz4) receptor, Lrp5 corecep- The present study focuses on the retinal vasculature. This tor, or Norrin ligand cause retinal hypovasculariza- vasculature plays a central role in a variety of ocular diseases, tion, but the mechanisms by which Norrin/Fz4/Lrp including diabetic retinopathy and retinopathy of prematurity signaling controls vascular development have not (Gariano and Gardner, 2005). The retinal vasculature presents a simple and stereotyped architecture. The major arteries and been defined. Using mouse genetic and cell culture veins reside on the vitreal face of the retina and are oriented radi- models, we show that loss of Fz4 signaling in endo- ally from their origin at the optic disc. -

Supplementary Table 1: Adhesion Genes Data Set
Supplementary Table 1: Adhesion genes data set PROBE Entrez Gene ID Celera Gene ID Gene_Symbol Gene_Name 160832 1 hCG201364.3 A1BG alpha-1-B glycoprotein 223658 1 hCG201364.3 A1BG alpha-1-B glycoprotein 212988 102 hCG40040.3 ADAM10 ADAM metallopeptidase domain 10 133411 4185 hCG28232.2 ADAM11 ADAM metallopeptidase domain 11 110695 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 195222 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 165344 8751 hCG20021.3 ADAM15 ADAM metallopeptidase domain 15 (metargidin) 189065 6868 null ADAM17 ADAM metallopeptidase domain 17 (tumor necrosis factor, alpha, converting enzyme) 108119 8728 hCG15398.4 ADAM19 ADAM metallopeptidase domain 19 (meltrin beta) 117763 8748 hCG20675.3 ADAM20 ADAM metallopeptidase domain 20 126448 8747 hCG1785634.2 ADAM21 ADAM metallopeptidase domain 21 208981 8747 hCG1785634.2|hCG2042897 ADAM21 ADAM metallopeptidase domain 21 180903 53616 hCG17212.4 ADAM22 ADAM metallopeptidase domain 22 177272 8745 hCG1811623.1 ADAM23 ADAM metallopeptidase domain 23 102384 10863 hCG1818505.1 ADAM28 ADAM metallopeptidase domain 28 119968 11086 hCG1786734.2 ADAM29 ADAM metallopeptidase domain 29 205542 11085 hCG1997196.1 ADAM30 ADAM metallopeptidase domain 30 148417 80332 hCG39255.4 ADAM33 ADAM metallopeptidase domain 33 140492 8756 hCG1789002.2 ADAM7 ADAM metallopeptidase domain 7 122603 101 hCG1816947.1 ADAM8 ADAM metallopeptidase domain 8 183965 8754 hCG1996391 ADAM9 ADAM metallopeptidase domain 9 (meltrin gamma) 129974 27299 hCG15447.3 ADAMDEC1 ADAM-like, -

Increased Liver Carcinogenesis and Enrichment of Stem Cell Properties in Livers of Dickkopf 2 (Dkk2) Deleted Mice
www.impactjournals.com/oncotarget/ Oncotarget, Vol. 7, No. 20 Increased liver carcinogenesis and enrichment of stem cell properties in livers of Dickkopf 2 (Dkk2) deleted mice Thorsten Maass1, Jens Marquardt2, Ju-Seog Lee3, Markus Krupp4, Peter Scholz-Kreisel2, Carolin Mogler5, Peter Schirmacher5, Martina Müller1, Heiner Westphal6, Peter R. Galle2, Andreas Teufel1 1Department of Internal Medicine I, University of Regensburg, Regensburg, Germany 2I. Department of Medicine, University Medical Center Mainz, Mainz, Germany 3Cancer Biology Program, MD Anderson Cancer Center, Houston, TX, USA 4Department of Informatics, Johannes Gutenberg University Mainz, Mainz, Germany 5Institute of Pathology, University of Heidelberg, Heidelberg, Germany 6 Laboratory of Mammalian Genes and Development, National Institute of Child Health and Human Development, National Institutes of Health, Bethesda, MD, USA Correspondence to: Andreas Teufel, email: [email protected] Keywords: transcriptomics profiling, prognostic signature, genetic signature, Dkk2, stem cells Received: January 13, 2015 Accepted: February 08, 2015 Published: March 16, 2015 ABSTRACT Dkk2 a antagonist of the Wnt/β-catenin-signaling pathway was shown to be silenced in diverse cancers. More recent data indicate that Dkk family members may also possess functions independent of Wnt-signaling during carcinogenesis. The detailed biological function of Dkks and its relevance for liver cancer is unknown. We analyzed the effects of a genetic deletion of Dkk2 (Dkk2−/−) in a hepatocarcinogenesis model using DEN/Phenobarbital. Untreated Dkk2−/− animals, showed considerable atypia with variation of hepatocyte size and chromatin density. In livers of Dkk2−/− mice nodule formation was seen at 9 months of age with focal loss of trabecular architecture and atypical hepatocytes and after DEN induction Dkk2−/− mice developed significantly more liver tumors compared to controls. -

FAM20B (N-14): Sc-164312
SAN TA C RUZ BI OTEC HNOL OG Y, INC . FAM20B (N-14): sc-164312 BACKGROUND SOURCE Chromosome 1 is the largest human chromosome spanning about 260 million FAM20B (N-14) is an affinity purified goat polyclonal antibody raised against base pairs and making up 8% of the human genome. There are about 3,000 a peptide mapping near the N-terminus of FAM20B of human origin. genes on chromosome 1, and considering the great number of genes there are also a large number of diseases associated with chromosome 1. Notably, the PRODUCT rare aging disease Hutchinson-Gilford progeria is associated with the LMNA Each vial contains 200 µg IgG in 1.0 ml of PBS with < 0.1% sodium azide gene which encodes lamin A. When defective, the LMNA gene product can and 0.1% gelatin. build up in the nucleus and cause characteristic nuclear blebs. The mechanism of rapidly enhanced aging is unclear and is a topic of continuing exploration. Blocking peptide available for competition studies, sc-164312 P, (100 µg The MUTYH gene is located on chromosome 1 and is partially responsible for peptide in 0.5 ml PBS containing < 0.1% sodium azide and 0.2% BSA). familial adenomatous polyposis. Stickler syndrome, Parkinsons, Gaucher dis - ease and Usher syndrome are also associated with chromosome 1. A break - APPLICATIONS point has been identified in 1q which disrupts the DISC1 gene and is linked FAM20B (N-14) is recommended for detection of FAM20B of mouse, rat and to schizophrenia. Aberrations in chromosome 1 are found in a variety of can - human origin by Western Blotting (starting dilution 1:200, dilution range cers including head and neck cancer, malignant melanoma and multiple myelo - 1:100-1:1000), immunofluorescence (starting dilution 1:50, dilution range ma. -

High-Throughput Bioinformatics Approaches to Understand Gene Expression Regulation in Head and Neck Tumors
High-throughput bioinformatics approaches to understand gene expression regulation in head and neck tumors by Yanxiao Zhang A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy (Bioinformatics) in The University of Michigan 2016 Doctoral Committee: Associate Professor Maureen A. Sartor, Chair Professor Thomas E. Carey Assistant Professor Hui Jiang Professor Ronald J. Koenig Associate Professor Laura M. Rozek Professor Kerby A. Shedden c Yanxiao Zhang 2016 All Rights Reserved I dedicate this thesis to my family. For their unfailing love, understanding and support. ii ACKNOWLEDGEMENTS I would like to express my gratitude to Dr. Maureen Sartor for her guidance in my research and career development. She is a great mentor. She patiently taught me when I started new in this field, granted me freedom to explore and helped me out when I got lost. Her dedication to work, enthusiasm in teaching, mentoring and communicating science have inspired me to feel the excite- ment of research beyond novel scientific discoveries. I’m also grateful to have an interdisciplinary committee. Their feedback on my research progress and presentation skills is very valuable. In particular, I would like to thank Dr. Thomas Carey and Dr. Laura Rozek for insightful discussions on the biology of head and neck cancers and human papillomavirus, Dr. Ronald Koenig for expert knowledge on thyroid cancers, Dr. Hui Jiang and Dr. Kerby Shedden for feedback on the statistics part of my thesis. I would like to thank all the past and current members of Sartor lab for making the lab such a lovely place to stay and work in. -

Supporting Online Material
1 Conserved Transcriptomic Profiles Underpin Monogamy across 2 Vertebrates 3 4 Rebecca L. Younga,b,1, Michael H. Ferkinc, Nina F. Ockendon-Powelld, Veronica N. Orre, 5 Steven M. Phelpsa,f, Ákos Pogányg, Corinne L. Richards-Zawackih, Kyle Summersi, 6 Tamás Székelyd,j,k, Brian C. Trainore, Araxi O. Urrutiaj,l, Gergely Zacharm, Lauren A. 7 O’Connelln, and Hans A. Hofmanna,b,f,1 8 9 1correspondence to: [email protected]; [email protected] 10 1 www .pnas.org/cgi/doi/10.1073/pnas.1813775115 11 Materials and Methods 12 Sample collection and RNA extraction 13 Reproductive males of each focal species were sacrificed and brains were rapidly 14 dissected and stored to preserve RNA (species-specific details provided below). All animal 15 care and use practices were approved by the respective institutions. For each species, 16 RNA from three individuals was pooled to create an aggregate sample for transcriptome 17 comparison. The focus of this study is to characterize similarity among species with 18 independent species-level transitions to a monogamous mating system rather than to 19 characterize individual-level variation in gene expression. Pooled samples are reflective 20 of species-level gene expression variation of each species and limit potentially 21 confounding individual variation for species-level comparisons (1, 2). While exploration of 22 individual variation is critical to identify mechanisms underlying differences in behavioral 23 expression, high levels of variation between two pooled samples of conspecifics could 24 obscure more general species-specific gene expression patterns. Note that two pooled 25 replicates per species would not be sufficiently large for estimating within species 26 variance, and the effect of an outlier within a pool of two individuals would be considerable. -

DIPPER, a Spatiotemporal Proteomics Atlas of Human Intervertebral Discs
TOOLS AND RESOURCES DIPPER, a spatiotemporal proteomics atlas of human intervertebral discs for exploring ageing and degeneration dynamics Vivian Tam1,2†, Peikai Chen1†‡, Anita Yee1, Nestor Solis3, Theo Klein3§, Mateusz Kudelko1, Rakesh Sharma4, Wilson CW Chan1,2,5, Christopher M Overall3, Lisbet Haglund6, Pak C Sham7, Kathryn Song Eng Cheah1, Danny Chan1,2* 1School of Biomedical Sciences, , The University of Hong Kong, Hong Kong; 2The University of Hong Kong Shenzhen of Research Institute and Innovation (HKU-SIRI), Shenzhen, China; 3Centre for Blood Research, Faculty of Dentistry, University of British Columbia, Vancouver, Canada; 4Proteomics and Metabolomics Core Facility, The University of Hong Kong, Hong Kong; 5Department of Orthopaedics Surgery and Traumatology, HKU-Shenzhen Hospital, Shenzhen, China; 6Department of Surgery, McGill University, Montreal, Canada; 7Centre for PanorOmic Sciences (CPOS), The University of Hong Kong, Hong Kong Abstract The spatiotemporal proteome of the intervertebral disc (IVD) underpins its integrity *For correspondence: and function. We present DIPPER, a deep and comprehensive IVD proteomic resource comprising [email protected] 94 genome-wide profiles from 17 individuals. To begin with, protein modules defining key †These authors contributed directional trends spanning the lateral and anteroposterior axes were derived from high-resolution equally to this work spatial proteomes of intact young cadaveric lumbar IVDs. They revealed novel region-specific Present address: ‡Department profiles of regulatory activities