Identification of Key Genes and Pathways in Pancreatic Cancer
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Browsing Genes and Genomes with Ensembl
The Bioinformatics Roadshow Copenhagen, Denmark 16 & 17 June 2011 BROWSING GENES AND GENOMES WITH ENSEMBL EXERCISES AND ANSWERS Note: These exercises are based on Ensembl version 62 (13 April 2011). After in future a new version has gone live, version 62 will still be available at http://e62.ensembl.org. If your answer doesn’t correspond with the given answer, please consult the instructor. ______________________________________________________________ BROWSER ______________________________________________________________ Exercise 1 – Exploring a gene (a) Find the human F9 (Coagulation factor IX) gene. On which chromosome and which strand of the genome is this gene located? How many transcripts (splice variants) have been annotated for it? (b) What is the longest transcript? How long is the protein it encodes? How many exons does it have? Are any of the exons completely or partially untranslated? (c) Have a look at the external references for ENST00000218099. What is the function of F9? (d) Is it possible to monitor expression of ENST00000218099 with the CodeLink microarray? If so, can it also be used to monitor expression of the other two transcripts? (e) In which part (i.e. the N-terminal or C-terminal half) of the protein encoded by ENST00000218099 does its peptidase activity, responsible for the cleavage of factor X to yield its active form factor Xa, reside? (f) How many non-synonymous variants have been discovered for the protein encoded by ENST00000218099? (g) Is there a mouse ortholog predicted for the human F9 gene? (h) On which cytogenetic band and on which contig is the F9 gene located? Is there a BAC clone that contains the complete F9 gene? (i) If you have yourself a gene of interest, explore what information Ensembl displays about it! ______________________________________________________________ Answer (a) Go to the Ensembl homepage (http://www.ensembl.org). -

Recombinant Human FLRT1 Catalog Number: 2794-FL
Recombinant Human FLRT1 Catalog Number: 2794-FL DESCRIPTION Source Mouse myeloma cell line, NS0derived human FLRT1 protein Ile21Pro524, with a Cterminal 6His tag Accession # Q9NZU1 Nterminal Sequence Ile21 Analysis Predicted Molecular 56.3 kDa Mass SPECIFICATIONS SDSPAGE 7080 kDa, reducing conditions Activity Measured by the ability of the immobilized protein to support the adhesion of Neuro2A mouse neuroblastoma cells. Recombinant Human FLRT1 immobilized at 2.5 μg/mL, 100 μL/well, will meidate > 50% Neuro2A cell adhesion. Optimal dilutions should be determined by each laboratory for each application. Endotoxin Level <0.10 EU per 1 μg of the protein by the LAL method. Purity >95%, by SDSPAGE under reducing conditions and visualized by silver stain. Formulation Lyophilized from a 0.2 μm filtered solution in PBS. See Certificate of Analysis for details. PREPARATION AND STORAGE Reconstitution Reconstitute at 200 μg/mL in sterile PBS. Shipping The product is shipped at ambient temperature. Upon receipt, store it immediately at the temperature recommended below. Stability & Storage Use a manual defrost freezer and avoid repeated freezethaw cycles. l 12 months from date of receipt, 20 to 70 °C as supplied. l 1 month, 2 to 8 °C under sterile conditions after reconstitution. l 3 months, 20 to 70 °C under sterile conditions after reconstitution. BACKGROUND FLRT1 is one of three FLRT (fibronectin, leucine rich repeat, transmembrane) glycoproteins expressed in distinct areas of the developing brain and other tissues (1, 2). The 90 kDa type I transmembrane (TM) human FLRT1 is synthesized as a 646 amino acid (aa) precursor with a 20 aa signal sequence, a 504 aa extracellular domain (ECD), a 21 aa TM segment and a 101 aa cytoplasmic region. -

University of Groningen Epidermolysis Bullosa Simplex Bolling, Maria
University of Groningen Epidermolysis bullosa simplex Bolling, Maria Caroline IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document version below. Document Version Publisher's PDF, also known as Version of record Publication date: 2010 Link to publication in University of Groningen/UMCG research database Citation for published version (APA): Bolling, M. C. (2010). Epidermolysis bullosa simplex: new insights in desmosomal cardiocutaneous syndromes. s.n. Copyright Other than for strictly personal use, it is not permitted to download or to forward/distribute the text or part of it without the consent of the author(s) and/or copyright holder(s), unless the work is under an open content license (like Creative Commons). The publication may also be distributed here under the terms of Article 25fa of the Dutch Copyright Act, indicated by the “Taverne” license. More information can be found on the University of Groningen website: https://www.rug.nl/library/open-access/self-archiving-pure/taverne- amendment. Take-down policy If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your claim. Downloaded from the University of Groningen/UMCG research database (Pure): http://www.rug.nl/research/portal. For technical reasons the number of authors shown on this cover page is limited to 10 maximum. Download date: 10-10-2021 5 Chromosomal microdeletion explains extracutaneous -

Analysis of Gene Expression Data for Gene Ontology
ANALYSIS OF GENE EXPRESSION DATA FOR GENE ONTOLOGY BASED PROTEIN FUNCTION PREDICTION A Thesis Presented to The Graduate Faculty of The University of Akron In Partial Fulfillment of the Requirements for the Degree Master of Science Robert Daniel Macholan May 2011 ANALYSIS OF GENE EXPRESSION DATA FOR GENE ONTOLOGY BASED PROTEIN FUNCTION PREDICTION Robert Daniel Macholan Thesis Approved: Accepted: _______________________________ _______________________________ Advisor Department Chair Dr. Zhong-Hui Duan Dr. Chien-Chung Chan _______________________________ _______________________________ Committee Member Dean of the College Dr. Chien-Chung Chan Dr. Chand K. Midha _______________________________ _______________________________ Committee Member Dean of the Graduate School Dr. Yingcai Xiao Dr. George R. Newkome _______________________________ Date ii ABSTRACT A tremendous increase in genomic data has encouraged biologists to turn to bioinformatics in order to assist in its interpretation and processing. One of the present challenges that need to be overcome in order to understand this data more completely is the development of a reliable method to accurately predict the function of a protein from its genomic information. This study focuses on developing an effective algorithm for protein function prediction. The algorithm is based on proteins that have similar expression patterns. The similarity of the expression data is determined using a novel measure, the slope matrix. The slope matrix introduces a normalized method for the comparison of expression levels throughout a proteome. The algorithm is tested using real microarray gene expression data. Their functions are characterized using gene ontology annotations. The results of the case study indicate the protein function prediction algorithm developed is comparable to the prediction algorithms that are based on the annotations of homologous proteins. -

Mouse Net1 Knockout Project (CRISPR/Cas9)
https://www.alphaknockout.com Mouse Net1 Knockout Project (CRISPR/Cas9) Objective: To create a Net1 knockout Mouse model (C57BL/6J) by CRISPR/Cas-mediated genome engineering. Strategy summary: The Net1 gene (NCBI Reference Sequence: NM_019671 ; Ensembl: ENSMUSG00000021215 ) is located on Mouse chromosome 13. 12 exons are identified, with the ATG start codon in exon 1 and the TAA stop codon in exon 12 (Transcript: ENSMUST00000091853). Exon 4~11 will be selected as target site. Cas9 and gRNA will be co-injected into fertilized eggs for KO Mouse production. The pups will be genotyped by PCR followed by sequencing analysis. Note: Mice homozygous for a knock-out allele exhibit delayed mammary gland development during puberty associated with slower ductal extension, reduced ductal branching and epithelial cell proliferation, disorganized myoepithelial and ductal epithelial cells, and increased collagen deposition. Exon 4 starts from about 14.34% of the coding region. Exon 4~11 covers 63.25% of the coding region. The size of effective KO region: ~4176 bp. The KO region does not have any other known gene. Page 1 of 9 https://www.alphaknockout.com Overview of the Targeting Strategy Wildtype allele 5' gRNA region gRNA region 3' 1 4 5 6 7 8 9 10 11 12 Legends Exon of mouse Net1 Knockout region Page 2 of 9 https://www.alphaknockout.com Overview of the Dot Plot (up) Window size: 15 bp Forward Reverse Complement Sequence 12 Note: The 2000 bp section upstream of Exon 4 is aligned with itself to determine if there are tandem repeats. Tandem repeats are found in the dot plot matrix. -

Dual Proteome-Scale Networks Reveal Cell-Specific Remodeling of the Human Interactome
bioRxiv preprint doi: https://doi.org/10.1101/2020.01.19.905109; this version posted January 19, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Dual Proteome-scale Networks Reveal Cell-specific Remodeling of the Human Interactome Edward L. Huttlin1*, Raphael J. Bruckner1,3, Jose Navarrete-Perea1, Joe R. Cannon1,4, Kurt Baltier1,5, Fana Gebreab1, Melanie P. Gygi1, Alexandra Thornock1, Gabriela Zarraga1,6, Stanley Tam1,7, John Szpyt1, Alexandra Panov1, Hannah Parzen1,8, Sipei Fu1, Arvene Golbazi1, Eila Maenpaa1, Keegan Stricker1, Sanjukta Guha Thakurta1, Ramin Rad1, Joshua Pan2, David P. Nusinow1, Joao A. Paulo1, Devin K. Schweppe1, Laura Pontano Vaites1, J. Wade Harper1*, Steven P. Gygi1*# 1Department of Cell Biology, Harvard Medical School, Boston, MA, 02115, USA. 2Broad Institute, Cambridge, MA, 02142, USA. 3Present address: ICCB-Longwood Screening Facility, Harvard Medical School, Boston, MA, 02115, USA. 4Present address: Merck, West Point, PA, 19486, USA. 5Present address: IQ Proteomics, Cambridge, MA, 02139, USA. 6Present address: Vor Biopharma, Cambridge, MA, 02142, USA. 7Present address: Rubius Therapeutics, Cambridge, MA, 02139, USA. 8Present address: RPS North America, South Kingstown, RI, 02879, USA. *Correspondence: [email protected] (E.L.H.), [email protected] (J.W.H.), [email protected] (S.P.G.) #Lead Contact: [email protected] bioRxiv preprint doi: https://doi.org/10.1101/2020.01.19.905109; this version posted January 19, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. -

Glucose-Induced Changes in Gene Expression in Human Pancreatic Islets: Causes Or Consequences of Chronic Hyperglycemia
Diabetes Volume 66, December 2017 3013 Glucose-Induced Changes in Gene Expression in Human Pancreatic Islets: Causes or Consequences of Chronic Hyperglycemia Emilia Ottosson-Laakso,1 Ulrika Krus,1 Petter Storm,1 Rashmi B. Prasad,1 Nikolay Oskolkov,1 Emma Ahlqvist,1 João Fadista,2 Ola Hansson,1 Leif Groop,1,3 and Petter Vikman1 Diabetes 2017;66:3013–3028 | https://doi.org/10.2337/db17-0311 Dysregulation of gene expression in islets from patients In patients with type 2 diabetes (T2D), islet function de- with type 2 diabetes (T2D) might be causally involved clines progressively. Although the initial pathogenic trigger in the development of hyperglycemia, or it could develop of impaired b-cell function is still unknown, elevated glu- as a consequence of hyperglycemia (i.e., glucotoxicity). cose levels are known to further aggravate b-cell function, a To separate the genes that could be causally involved condition referred to as glucotoxicity, which can stimulate in pathogenesis from those likely to be secondary to hy- apoptosis and lead to reduced b-cell mass (1–5). Prolonged perglycemia, we exposed islets from human donors to exposure to hyperglycemia also can induce endoplasmic re- ISLET STUDIES normal or high glucose concentrations for 24 h and ana- ticulum (ER) stress and production of reactive oxygen spe- fi lyzed gene expression. We compared these ndings with cies (6), which can further impair islet function and thereby gene expression in islets from donors with normal glucose the ability of islets to secrete the insulin needed to meet the tolerance and hyperglycemia (including T2D). The genes increased demands imposed by insulin resistance and obe- whose expression changed in the same direction after sity (7). -
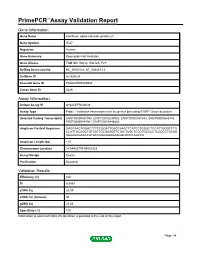
Primepcr™Assay Validation Report
PrimePCR™Assay Validation Report Gene Information Gene Name interferon, alpha-inducible protein 27 Gene Symbol IFI27 Organism Human Gene Summary Description Not Available Gene Aliases FAM14D, ISG12, ISG12A, P27 RefSeq Accession No. NC_000014.8, NT_026437.12 UniGene ID Hs.532634 Ensembl Gene ID ENSG00000165949 Entrez Gene ID 3429 Assay Information Unique Assay ID qHsaCEP0024638 Assay Type Probe - Validation information is for the primer pair using SYBR® Green detection Detected Coding Transcript(s) ENST00000557098, ENST00000298902, ENST00000557634, ENST00000555744, ENST00000444961, ENST00000448882 Amplicon Context Sequence GAGCAACTGGACTCTCCGGATTGACCAAGTTCATCCTGGGCTCCATTGGGTCTG CCATTGCGGCTGTCATTGCGAGGTTCTACTAGCTCCCTGCCCCTCGCCCTGCAG AGAAGAGAACCATGCCAGGGGAGAAGGCACCCAGCCA Amplicon Length (bp) 115 Chromosome Location 14:94582779-94582923 Assay Design Exonic Purification Desalted Validation Results Efficiency (%) 100 R2 0.9997 cDNA Cq 23.09 cDNA Tm (Celsius) 86 gDNA Cq 23.06 Specificity (%) 100 Information to assist with data interpretation is provided at the end of this report. Page 1/4 PrimePCR™Assay Validation Report IFI27, Human Amplification Plot Amplification of cDNA generated from 25 ng of universal reference RNA Melt Peak Melt curve analysis of above amplification Standard Curve Standard curve generated using 20 million copies of template diluted 10-fold to 20 copies Page 2/4 PrimePCR™Assay Validation Report Products used to generate validation data Real-Time PCR Instrument CFX384 Real-Time PCR Detection System Reverse Transcription Reagent iScript™ Advanced cDNA Synthesis Kit for RT-qPCR Real-Time PCR Supermix SsoAdvanced™ SYBR® Green Supermix Experimental Sample qPCR Human Reference Total RNA Data Interpretation Unique Assay ID This is a unique identifier that can be used to identify the assay in the literature and online. Detected Coding Transcript(s) This is a list of the Ensembl transcript ID(s) that this assay will detect. -

1 Supporting Information for a Microrna Network Regulates
Supporting Information for A microRNA Network Regulates Expression and Biosynthesis of CFTR and CFTR-ΔF508 Shyam Ramachandrana,b, Philip H. Karpc, Peng Jiangc, Lynda S. Ostedgaardc, Amy E. Walza, John T. Fishere, Shaf Keshavjeeh, Kim A. Lennoxi, Ashley M. Jacobii, Scott D. Rosei, Mark A. Behlkei, Michael J. Welshb,c,d,g, Yi Xingb,c,f, Paul B. McCray Jr.a,b,c Author Affiliations: Department of Pediatricsa, Interdisciplinary Program in Geneticsb, Departments of Internal Medicinec, Molecular Physiology and Biophysicsd, Anatomy and Cell Biologye, Biomedical Engineeringf, Howard Hughes Medical Instituteg, Carver College of Medicine, University of Iowa, Iowa City, IA-52242 Division of Thoracic Surgeryh, Toronto General Hospital, University Health Network, University of Toronto, Toronto, Canada-M5G 2C4 Integrated DNA Technologiesi, Coralville, IA-52241 To whom correspondence should be addressed: Email: [email protected] (M.J.W.); yi- [email protected] (Y.X.); Email: [email protected] (P.B.M.) This PDF file includes: Materials and Methods References Fig. S1. miR-138 regulates SIN3A in a dose-dependent and site-specific manner. Fig. S2. miR-138 regulates endogenous SIN3A protein expression. Fig. S3. miR-138 regulates endogenous CFTR protein expression in Calu-3 cells. Fig. S4. miR-138 regulates endogenous CFTR protein expression in primary human airway epithelia. Fig. S5. miR-138 regulates CFTR expression in HeLa cells. Fig. S6. miR-138 regulates CFTR expression in HEK293T cells. Fig. S7. HeLa cells exhibit CFTR channel activity. Fig. S8. miR-138 improves CFTR processing. Fig. S9. miR-138 improves CFTR-ΔF508 processing. Fig. S10. SIN3A inhibition yields partial rescue of Cl- transport in CF epithelia. -

Reconstructing Cell Cycle Pseudo Time-Series Via Single-Cell Transcriptome Data—Supplement
School of Natural Sciences and Mathematics Reconstructing Cell Cycle Pseudo Time-Series Via Single-Cell Transcriptome Data—Supplement UT Dallas Author(s): Michael Q. Zhang Rights: CC BY 4.0 (Attribution) ©2017 The Authors Citation: Liu, Zehua, Huazhe Lou, Kaikun Xie, Hao Wang, et al. 2017. "Reconstructing cell cycle pseudo time-series via single-cell transcriptome data." Nature Communications 8, doi:10.1038/s41467-017-00039-z This document is being made freely available by the Eugene McDermott Library of the University of Texas at Dallas with permission of the copyright owner. All rights are reserved under United States copyright law unless specified otherwise. File name: Supplementary Information Description: Supplementary figures, supplementary tables, supplementary notes, supplementary methods and supplementary references. CCNE1 CCNE1 CCNE1 CCNE1 36 40 32 34 32 35 30 32 28 30 30 28 28 26 24 25 Normalized Expression Normalized Expression Normalized Expression Normalized Expression 26 G1 S G2/M G1 S G2/M G1 S G2/M G1 S G2/M Cell Cycle Stage Cell Cycle Stage Cell Cycle Stage Cell Cycle Stage CCNE1 CCNE1 CCNE1 CCNE1 40 32 40 40 35 30 38 30 30 28 36 25 26 20 20 34 Normalized Expression Normalized Expression Normalized Expression 24 Normalized Expression G1 S G2/M G1 S G2/M G1 S G2/M G1 S G2/M Cell Cycle Stage Cell Cycle Stage Cell Cycle Stage Cell Cycle Stage Supplementary Figure 1 | High stochasticity of single-cell gene expression means, as demonstrated by relative expression levels of gene Ccne1 using the mESC-SMARTer data. For every panel, 20 sample cells were randomly selected for each of the three stages, followed by plotting the mean expression levels at each stage. -

Supplementary Table 1: Adhesion Genes Data Set
Supplementary Table 1: Adhesion genes data set PROBE Entrez Gene ID Celera Gene ID Gene_Symbol Gene_Name 160832 1 hCG201364.3 A1BG alpha-1-B glycoprotein 223658 1 hCG201364.3 A1BG alpha-1-B glycoprotein 212988 102 hCG40040.3 ADAM10 ADAM metallopeptidase domain 10 133411 4185 hCG28232.2 ADAM11 ADAM metallopeptidase domain 11 110695 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 195222 8038 hCG40937.4 ADAM12 ADAM metallopeptidase domain 12 (meltrin alpha) 165344 8751 hCG20021.3 ADAM15 ADAM metallopeptidase domain 15 (metargidin) 189065 6868 null ADAM17 ADAM metallopeptidase domain 17 (tumor necrosis factor, alpha, converting enzyme) 108119 8728 hCG15398.4 ADAM19 ADAM metallopeptidase domain 19 (meltrin beta) 117763 8748 hCG20675.3 ADAM20 ADAM metallopeptidase domain 20 126448 8747 hCG1785634.2 ADAM21 ADAM metallopeptidase domain 21 208981 8747 hCG1785634.2|hCG2042897 ADAM21 ADAM metallopeptidase domain 21 180903 53616 hCG17212.4 ADAM22 ADAM metallopeptidase domain 22 177272 8745 hCG1811623.1 ADAM23 ADAM metallopeptidase domain 23 102384 10863 hCG1818505.1 ADAM28 ADAM metallopeptidase domain 28 119968 11086 hCG1786734.2 ADAM29 ADAM metallopeptidase domain 29 205542 11085 hCG1997196.1 ADAM30 ADAM metallopeptidase domain 30 148417 80332 hCG39255.4 ADAM33 ADAM metallopeptidase domain 33 140492 8756 hCG1789002.2 ADAM7 ADAM metallopeptidase domain 7 122603 101 hCG1816947.1 ADAM8 ADAM metallopeptidase domain 8 183965 8754 hCG1996391 ADAM9 ADAM metallopeptidase domain 9 (meltrin gamma) 129974 27299 hCG15447.3 ADAMDEC1 ADAM-like, -

Novel Pathogenic Mutations of FERMT1 in Two Chinese Kindler Syndrome Families
Novel Pathogenic Mutations of FERMT1 in two Chinese Kindler Syndrome Families Min Li First Aliated Hospital of Soochow University Weisheng Li First Aliated Hospital of Soochow University Dan Zhu First Aliated Hospital of Soochow University Likui Lu First Aliated Hospital of Soochow University Jingliu Liu First Aliated Hospital of Soochow University Yajun Shi First Aliated Hospital of Soochow University Zhimiao Lin Peking University First Hospital Miao Sun ( [email protected] ) First Aliated Hospital of Soochow University https://orcid.org/0000-0002-6414-368X Research Keywords: Kindler syndrome, FERMT1, kindlin-1, nonsense mutation, frame-shift mutation Posted Date: March 31st, 2021 DOI: https://doi.org/10.21203/rs.3.rs-354474/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/13 Abstract Background: Kindler syndrome (KNDLRS) is a very rare autosomal recessive disorder characterized by bullous poikiloderma with photosensitivity. Loss-of-function mutations in FERMT1, which located on chromosome 20p12.3, were responsible for KNDLRS. Numerous mutations in FERMT1 have been reported to be associated with KNDLRS. Results: The present study reported two Chinese KNDLRS families, and affected individuals from both families presented with poikiloderma, palmoplantar hyperkeratosis, and diffuse cigarette paper like atrophy on hands. Skin biopsy of the proband from family 2 showed atrophy of epidermis, hyperkeratosis, dilated blood vessels in upper dermis, and microbubbles at the dermis and epidermis junction. Medical Whole Exome Sequencing V4 combined with Sanger sequencing revealed mutations in FERMT1 with affected individuals. Compound heterozygous nonsense mutations (c.193C>T, c.277C>T) were found with family 1, and a homozygous frameshift mutation (c.220delC) was observed in family 2.