LABORATORY #4, 4 MM FROG EMBRYOS Read the Serial Sections on the Slide from Top Left to Bottom Right, Just As You Read the Lines in a Book
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Embryology of Branchial Region
TRANSCRIPTIONS OF NARRATIONS FOR EMBRYOLOGY OF THE BRANCHIAL REGION Branchial Arch Development, slide 2 This is a very familiar picture - a median sagittal section of a four week embryo. I have actually done one thing correctly, I have eliminated the oropharyngeal membrane, which does disappear sometime during the fourth week of development. The cloacal membrane, as you know, doesn't disappear until the seventh week, and therefore it is still intact here, but unlabeled. But, I've labeled a couple of things not mentioned before. First of all, the most cranial part of the foregut, that is, the part that is cranial to the chest region, is called the pharynx. The part of the foregut in the chest region is called the esophagus; you probably knew that. And then, leading to the pharynx from the outside, is an ectodermal inpocketing, which is called the stomodeum. That originally led to the oropharyngeal membrane, but now that the oropharyngeal membrane is ruptured, the stomodeum is a pathway between the amniotic cavity and the lumen of the foregut. The stomodeum is going to become your oral cavity. Branchial Arch Development, slide 3 This is an actual picture of a four-week embryo. It's about 5mm crown-rump length. The stomodeum is labeled - that is the future oral cavity that leads to the pharynx through the ruptured oropharyngeal membrane. And I've also indicated these ridges separated by grooves that lie caudal to the stomodeum and cranial to the heart, which are called branchial arches. Now, if this is a four- week old embryo, clearly these things have developed during the fourth week, and I've never mentioned them before. -

Subject Index
979 Subject Index Acellularity Asymmetry of embryonic starfish mesenchyme cells: KANEKO AND development of left/right asymmetry: BROWN AND OTHERS 129 WOLPERT 1 Acetylcholinesterase Avian tropomyosin/cholinesterase expression in ascidian embryo zygotes: CROWTHER AND OTHERS 953 localization of bFGF: KALCHEIM AND NEUFELD 203 Acrosome Axis reaction anterior-posterior zona receptors on guinea-pig spermatozoa: JONES AND organizer amount and axial pattern in Xenopus: WILLIAMS 41 STEWART AND GERHART 363 Actin Axogenesis effect of stretch on muscle gene expression: LOUGHNA AND Thy-1 expression in murine olfactory bulb: XUE AND OTHERS 217 OTHERS 851 Adult Axolotl rat embryo perinatnl ndu coexistence of O-2A and O-2A " progenitors: extracellular matrix in neural crest pathways: PERRIS WOLSWIJK AND OTHERS 691 AND OTHERS 533 Aggregation Axon pattern guidance of Dictyostelium discoideum: FOERSTER AND OTHERS 11 axon patterning at rat floor plate: BOVOLENTA AND Aging DODD 435 NGF receptor in rat dental tissue: BYERS AND OTHERS 461 outgrowth Agouti growth associated protein in chick visual system: cellular action of the mouse lethal yellow mutation: BARSH SCHLOSSHAUER AND OTHERS 395 AND OTHERS 683 rat Ammonia prenatal Schwann cell development: MIRSKY AND promotes cAMP accumulation in Dictyostelium: RILEY AND OTHERS 105 BARCLAY 715 regeneration AMP effects of protease inhibitors: FAWCETT AND HOUSDEN cyclic 59 ammonia promotes cAMP accumulation in Dictyostelium: RILEY AND BARCLAY 715 prenatal Schwann cell development: MIRSKY AND Back-transplantation OTHERS -

Clonal Dispersion During Neural Tube Formation 4097 of Neuromeres
Development 126, 4095-4106 (1999) 4095 Printed in Great Britain © The Company of Biologists Limited 1999 DEV2458 Successive patterns of clonal cell dispersion in relation to neuromeric subdivision in the mouse neuroepithelium Luc Mathis1,*, Johan Sieur1, Octavian Voiculescu2, Patrick Charnay2 and Jean-François Nicolas1,‡ 1Unité de Biologie moléculaire du Développement, Institut Pasteur, 25, rue du Docteur Roux, 75724 Paris Cedex 15, France 2Unité INSERM 368, Ecole Normale Supérieure, 46 rue d’Ulm, 75230 Paris Cedex 05, France *Present address: Beckman Institute (139-74), California Institute of Technology, Pasadena, CA, 91125, USA ‡Author for correspondence (e-mail: [email protected]) Accepted 5 July; published on WWW 23 August 1999 SUMMARY We made use of the laacz procedure of single-cell labelling the AP and DV axis of the neural tube. A similar sequence to visualize clones labelled before neuromere formation, in of AP cell dispersion followed by an arrest of AP cell 12.5-day mouse embryos. This allowed us to deduce two dispersion, a preferential DV cell dispersion and then by a successive phases of cell dispersion in the formation of the coherent neuroepithelial growth, is also observed in the rhombencephalon: an initial anterior-posterior (AP) cell spinal cord and mesencephalon. This demonstrates that a dispersion, followed by an asymmetrical dorsoventral (DV) similar cascade of cell events occurs in these different cell distribution during which AP cell dispersion occurs in domains of the CNS. In the prosencephalon, differences in territories smaller than one rhombomere. We conclude that spatial constraints may explain the variability in the the general arrest of AP cell dispersion precedes the onset orientation of cell clusters. -

Stages of Embryonic Development of the Zebrafish
DEVELOPMENTAL DYNAMICS 2032553’10 (1995) Stages of Embryonic Development of the Zebrafish CHARLES B. KIMMEL, WILLIAM W. BALLARD, SETH R. KIMMEL, BONNIE ULLMANN, AND THOMAS F. SCHILLING Institute of Neuroscience, University of Oregon, Eugene, Oregon 97403-1254 (C.B.K., S.R.K., B.U., T.F.S.); Department of Biology, Dartmouth College, Hanover, NH 03755 (W.W.B.) ABSTRACT We describe a series of stages for Segmentation Period (10-24 h) 274 development of the embryo of the zebrafish, Danio (Brachydanio) rerio. We define seven broad peri- Pharyngula Period (24-48 h) 285 ods of embryogenesis-the zygote, cleavage, blas- Hatching Period (48-72 h) 298 tula, gastrula, segmentation, pharyngula, and hatching periods. These divisions highlight the Early Larval Period 303 changing spectrum of major developmental pro- Acknowledgments 303 cesses that occur during the first 3 days after fer- tilization, and we review some of what is known Glossary 303 about morphogenesis and other significant events that occur during each of the periods. Stages sub- References 309 divide the periods. Stages are named, not num- INTRODUCTION bered as in most other series, providing for flexi- A staging series is a tool that provides accuracy in bility and continued evolution of the staging series developmental studies. This is because different em- as we learn more about development in this spe- bryos, even together within a single clutch, develop at cies. The stages, and their names, are based on slightly different rates. We have seen asynchrony ap- morphological features, generally readily identi- pearing in the development of zebrafish, Danio fied by examination of the live embryo with the (Brachydanio) rerio, embryos fertilized simultaneously dissecting stereomicroscope. -

Insect Digestion
INSECT DIGESTION Zoo 514 Dr. Reem Alajmi Insect digestive system The alimentary canal is concerned with digestion and absorption of food stuffs. Different parts of the gut are concerned with different aspects of these functions. In fluid feeding insects digestion begin before the food is ingested through the injection or regurgitation of enzymes on to the food . In general it occurs in the mid gut (most of the enzymes are produced). The enzymes break down the complex substances in the food into simple substances which can be absorbed and assimilated. Enzymes function optimally only within limited range of pH and temperature. Absorption is passive process in some cases , but in others active transport occurs. Absorption of water is important in terrestrial insects , rectum remove water from faeces. • Insects of different groups consume astonishing variety of foods, including watery xylem sap, vertebrate blood, dry wood, bacteria and algae and the internal tissues of other insects. • Types of mouth parts correlates with the diets of insect. Also gut structure and function affected by nutrient composition of the food eaten by insect. • Insects that take solid food typically have a wide, straight, short gut with strong musculature and obvious protection from abrasion (especially in the midgut, which has no cuticular lining). • These features are most obvious in solid-feeders with rapid throughput of food, as in many insects and plant-feeding caterpillars In contrast, insects feeding on blood, sap or nectar usually have long, narrow, convoluted guts to allow maximal contact with the liquid food; here, protection from abrasion is unnecessary. The most obvious gut specialization of liquid-feeders is a mechanism for removing excess water to concentrate nutrient substances prior to digestion, as seen in hemipterans. -
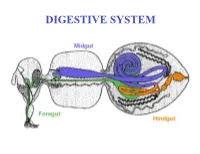
DIGESTIVE SYSTEM Generalized Insect Alimentary Tract the Digestive System Is Just a Tube Within a Surrounding Tube Called the Body
DIGESTIVE SYSTEM Generalized insect alimentary tract The digestive system is just a tube within a surrounding tube called the body. It starts with a mouth and ends with the anus. What goes on in between depends on the insect, its life stage and what it eats. The origin of the digestive tract. At the anterior pole of the embryo an indentation forms that will be the foregut or stomodeum. At the other end a similar thing occurs and the proctodeum or hindgut is formed. Both are lined by cuticle. They both are of ectodermal origin while the midgut is of mesodermal origin and is also called the mesenteron. This different origin of the midgut from the endoderm and not the ecotoderm probably explains why it is not lined with cuticle Anterior midgut invagination. In the bottom photo note the invagination starting forming the ventral furrow lumen (VF) MIDGUT FORMATION IN THE EMBRYO PMG in the above embryo shows the posterior midgut invagination cup where the posterior invagination shown in the drawing on the right will take place. Photo of Drosophila embryo. Hindgut invagination DIGESTIVE SYSTEM The digestive tract not only aids in obtaining, processing and digesting food molecules - It is the largest endocrine tissue in both humans and insects. The digestive system is involved in: 1. Obtaining food 2. Mechanically breaking it down into smaller particles that facilitate digestive enzymes acting on them 3. Enzymatic breakdown of larger food molecules into molecules that can pass through the digestive tract (midgut) and enter the hemolymph 4. Produces molecules or MESSENGERS (eg. -

Expression Patterns of Neural Genes in Euperipatoides Kanangrensis Suggest Divergent Evolution of Onychophoran and Euarthropod Neurogenesis
Expression patterns of neural genes in Euperipatoides kanangrensis suggest divergent evolution of onychophoran and euarthropod neurogenesis Bo Joakim Eriksson and Angelika Stollewerk1 School of Biological and Chemical Sciences, Queen Mary University of London, London E1 4NS, United Kingdom Edited by Thomas C. Kaufman, Indiana University, Bloomington, IN, and approved November 10, 2010 (received for review June 28, 2010) One of the controversial debates on euarthropod relationships pattern of neurogenesis that has been retained in these groups centers on the question as to whether insects, crustaceans, and and thus cannot be used to resolve euarthropod phylogeny? myriapods (Mandibulata) share a common ancestor or whether Analysis of neurogenesis in a closely related group, the Ony- myriapods group with the chelicerates (Myriochelata). The debate chophora, might shed light on this problem. Although the phy- was stimulated recently by studies in chelicerates and myriapods logenetic position of onychophorans is still debated, many that show that neural precursor groups (NPGs) segregate from the phylogenies group them with euarthropods and possibly tardi- neuroectoderm generating the nervous system, whereas in insects grades in the phylum Arthropoda; thus onychophorans share and crustaceans the nervous tissue is produced by stem cells. Do a common ancestor with euarthropods (8, 19–23). Analyses of the shared neural characters of myriapods and chelicerates repre- neurogenesis in onychophorans suggests that, similar to insects sent derived characters that support the Myriochelata grouping? and crustaceans, single neural precursors are formed in the neu- Or do they rather reflect the ancestral pattern? Analyses of neuro- roectoderm, rather than groups of cells as seen in chelicerates and genesis in a group closely related to euarthropods, the onycho- myriapods (24–26). -

Cephalic Neurulation in the Mouse Embryo Analyzed by SEM and Morphometry
THE ANATOMICAL RECORD 203:375-396 (1982) Cephalic Neurulation in the Mouse Embryo Analyzed by SEM and Morphometry ANTONE G. JACOBSON AND PATRICK P.L. TAM Department of Zoology. Uniuersity of Texas, Austin, TX 78712 (A.G.J.) and Department of Anatomy, (‘hinese University of Hong Kong, Shatin, N.T., Hong Kong IP.PL.T) ABSTRACT A detailed account of mouse neurulation is given based mostly on SEM analysis over 20 hr of development. Many observations and measure- ments were made on staged living embryos and on embryos prepared for scanning and light microscopy to help deduce what mechanisms may contribute to neural tube formation. Each lateral half of the early cephalic neural plate makes a convex bulge, opposite to the way it must fold to form a tube. Underlying mesenchyme and matrix are reported to have a role in forming these bulges. Processes that form the tube must overcome this opposed folding and the forces that produce it. Crani- al flexure begins long before tube formation. The flexure commences at the rostra1 tip of the cephalic neural plate, then the apex of the flexure migrates caudally to the mesencephalic region. Early appearance of this flexure imposes a mechanical impediment to tube closure in forebrain and midbrain regions. Tube closure begins in the cervical region exactly where the neural plate is reflected dorsally by a bend in the embryo. This bend may mechanically assist closure in this region. Cells of the mouse neural plate are reported to contain organized microfilaments and mi- crotubules, and the plate cells appear to change shape (reduce apical area and in- crease cell height) in the same manner as that suggested in embryos of some other species to contribute to neural tube formation. -

R-Cadherin Expression During Nucleus Formation in Chicken Forebrain Neuromeres
The Journal of Neuroscience, June 1995, 15(6): 4157-4172 R-Cadherin Expression during Nucleus Formation in Chicken Forebrain Neuromeres Susanne I. I. GBnzler and Christoph Redies Department of Biochemistry, Max Planck Institute for Developmental Biology, D-72072 Tiibingen, Germany The primordial neuroepithelium of the vertebrate forebrait dahl, 1924; Vaage, 1969; Kuhlenbeck, 1973; Puelleset al., 1987; consists of transverse and longitudinal morphogenetic Puelles and Rubenstein, 1993) that represent independentmor- compartments (“neuromeres”). During development, neu- phogenetic fields (Bergquist and KallCn, 1953a,b, 1954; Keyser, rons born in the ventricular zone of each neuromere mi- 1972; Puelleset al., 1987). The boundariesbetween neuromeres grate outward to the mantle zone. Here, neuroblasts grad- often coincide with primordial fiber tracts and restrict cell lin- ually accumulate and aggregate either into sheets (“lami- eage and migration. Generally, they coincide with the borders nae”) or into roundish structures (“nuclei”). As brain ar- of expressionof generegulatory proteins (reviewed in Lumsden, chitecture matures, sets of nuclei and laminae derived from 1990, 1993; Figdor and Stern, 1993; Krumlauf et al., 1993; Puel- several neuromeres become connected by fiber tracts to les and Rubenstein, 1993; Wilson et al., 1993). form functional circuits. We show by immunostaining and During development, a percentageof cells born in the prolif- in situ hybridization techniques that, in the E3-E5 chicken erative (ventricular) zone of each neuromerebecome postmitotic embryo, the cell adhesion molecule R-cadherin is ex- and migrate as neuroblastsoutward into the mantle zone (Sauer, pressed in several stripes and patches in the forebrain neu- 1935; Fujita, 1964; Morest, 1970) in two or three sustained roepithelium. -

Homeotic Gene Action in Embryonic Brain Development of Drosophila
Development 125, 1579-1589 (1998) 1579 Printed in Great Britain © The Company of Biologists Limited 1998 DEV1254 Homeotic gene action in embryonic brain development of Drosophila Frank Hirth, Beate Hartmann and Heinrich Reichert* Institute of Zoology, University of Basel, Rheinsprung 9, CH-4051 Basel, Switzerland *Author for correspondence (e-mail: [email protected]) Accepted 18 February; published on WWW 1 April 1998 SUMMARY Studies in vertebrates show that homeotic genes are absence of labial, mutant cells are generated and positioned involved in axial patterning and in specifying segmental correctly in the brain, but these cells do not extend axons. identity of the embryonic hindbrain and spinal cord. To Additionally, extending axons of neighboring wild-type gain further insights into homeotic gene action during CNS neurons stop at the mutant domains or project ectopically, development, we here characterize the role of the homeotic and defective commissural and longitudinal pathways genes in embryonic brain development of Drosophila. We result. Immunocytochemical analysis demonstrates that first use neuroanatomical techniques to map the entire cells in the mutant domains do not express neuronal anteroposterior order of homeotic gene expression in the markers, indicating a complete lack of neuronal identity. Drosophila CNS, and demonstrate that this order is An alternative glial identity is not adopted by these mutant virtually identical in the CNS of Drosophila and mammals. cells. Comparable effects are seen in Deformed mutants but We then carry out a genetic analysis of the labial gene in not in other homeotic gene mutants. Our findings embryonic brain development. Our analysis shows that demonstrate that the action of the homeotic genes labial loss-of-function mutation and ubiquitous overexpression of and Deformed are required for neuronal differentiation in labial results in ectopic expression of neighboring the developing brain of Drosophila. -

Embryology and Teratology in the Curricula of Healthcare Courses
ANATOMICAL EDUCATION Eur. J. Anat. 21 (1): 77-91 (2017) Embryology and Teratology in the Curricula of Healthcare Courses Bernard J. Moxham 1, Hana Brichova 2, Elpida Emmanouil-Nikoloussi 3, Andy R.M. Chirculescu 4 1Cardiff School of Biosciences, Cardiff University, Museum Avenue, Cardiff CF10 3AX, Wales, United Kingdom and Department of Anatomy, St. George’s University, St George, Grenada, 2First Faculty of Medicine, Institute of Histology and Embryology, Charles University Prague, Albertov 4, 128 01 Prague 2, Czech Republic and Second Medical Facul- ty, Institute of Histology and Embryology, Charles University Prague, V Úvalu 84, 150 00 Prague 5 , Czech Republic, 3The School of Medicine, European University Cyprus, 6 Diogenous str, 2404 Engomi, P.O.Box 22006, 1516 Nicosia, Cyprus , 4Department of Morphological Sciences, Division of Anatomy, Faculty of Medicine, C. Davila University, Bucharest, Romania SUMMARY Key words: Anatomy – Embryology – Education – Syllabus – Medical – Dental – Healthcare Significant changes are occurring worldwide in courses for healthcare studies, including medicine INTRODUCTION and dentistry. Critical evaluation of the place, tim- ing, and content of components that can be collec- Embryology is a sub-discipline of developmental tively grouped as the anatomical sciences has biology that relates to life before birth. Teratology however yet to be adequately undertaken. Surveys (τέρατος (teratos) meaning ‘monster’ or ‘marvel’) of teaching hours for embryology in US and UK relates to abnormal development and congenital medical courses clearly demonstrate that a dra- abnormalities (i.e. morphofunctional impairments). matic decline in the importance of the subject is in Embryological studies are concerned essentially progress, in terms of both a decrease in the num- with the laws and mechanisms associated with ber of hours allocated within the medical course normal development (ontogenesis) from the stage and in relation to changes in pedagogic methodol- of the ovum until parturition and the end of intra- ogies. -

Boundary Formation and Compartition in the Avian Diencephalon
The Journal of Neuroscience, July 1, 2001, 21(13):4699–4711 Boundary Formation and Compartition in the Avian Diencephalon Camilla W. Larsen, Lori M. Zeltser, and Andrew Lumsden Medical Research Council Centre for Developmental Neurobiology, King’s College London, London SE1 1UL, England The diencephalon comprises three functionally distinct regions: boundary does not. Similarly, there is no lineage restriction synencephalon, dorsal thalamus, and ventral thalamus. Pat- between the parencephalon and the more rostral forebrain terning of the diencephalon has been proposed to involve (secondary prosencephalon). Subdivision of the parencephalon subdivision of its anteroposterior axis into segments, neuro- into ventral and dorsal thalamus involves the formation of a meres or prosomeres (Bergquist and Kallen, 1954; Vaage, 1969; narrow intraparencephalic territory, the zona limitans intrathal- Figdor and Stern, 1993; Rubenstein et al., 1994; Redies et al., amica (zli). This is correlated with the acquisition of cell lineage 2000; Yoon et al., 2000). However, the number and sequence of restriction at both anterior and posterior borders of the zli, the diencephalic neuromeres, or even their existence, are uncer- appearance of boundary-specific properties, and Gbx2 and tain. We have examined the proposed subdivisions by morphol- Dlx2 expression in dorsal thalamic and ventral thalamic territo- ogy, gene expression, acquisition of boundary-specific pheno- ries, respectively. At stage 22, the synencephalon is divided into types, and cell lineage restriction. We find that at stage 16 in two domains, distinguished by differential gene expression and chick the diencephalon is divided into synencephalon and tissue morphology, but associated with neither a boundary parencephalon. The synencephalon exhibits neuromeric mor- phenotype nor cell lineage restriction.