Commissural Projections of the Nuclei of the Lateral Lemniscus and Keuronal Degeneration Following Midline Transections in the Adult Rat By
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Superior and Inferior Colliculi of the Mole (Scalopus Aquaticus Machxinus)
THE SUPERIOR AND INFERIOR COLLICULI OF THE MOLE (SCALOPUS AQUATICUS MACHXINUS) THOMAS N. JOHNSON' Laboratory of Comparative Neurology, Departmmt of Amtomy, Un&versity of hfiehigan, Ann Arbor INTRODUCTION This investigation is a study of the afferent and efferent connections of the tectum of the midbrain in the mole (Scalo- pus aquaticus machrinus). An attempt is made to correlate these findings with the known habits of the animal. A subterranean animal of the middle western portion of the United States, Scalopus aquaticus machrinus is the largest of the genus Scalopus and its habits have been more thor- oughly studied than those of others of this genus according to Jackson ('15) and Hamilton ('43). This animal prefers a well-drained, loose soil. It usually frequents open fields and pastures but also is found in thin woods and meadows. Following a rain, new superficial burrows just below the surface of the ground are pushed in all directions to facili- tate the capture of worms and other soil life. Ten inches or more below the surface the regular permanent highway is constructed; the mole retreats here during long periods of dry weather or when frost is in the ground. The principal food is earthworms although, under some circumstances, larvae and adult insects are the more usual fare. It has been demonstrated conclusively that, under normal conditions, moles will eat vegetable matter. It seems not improbable that they may take considerable quantities of it at times. A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the University of Michigan. -

Neuronal Organization in the Inferior Colliculus Revisited with Cell-Type- Dependent Monosynaptic Tracing
This Accepted Manuscript has not been copyedited and formatted. The final version may differ from this version. Research Articles: Systems/Circuits Neuronal organization in the inferior colliculus revisited with cell-type- dependent monosynaptic tracing Chenggang Chen1, Mingxiu Cheng1,2, Tetsufumi Ito3 and Sen Song1 1Tsinghua Laboratory of Brain and Intelligence (THBI) and Department of Biomedical Engineering, Beijing Innovation Center for Future Chip, Center for Brain-Inspired Computing Research, McGovern Institute for Brain Research, Tsinghua University, Beijing, 100084, China 2National Institute of Biological Sciences, Beijing, 102206, China 3Anatomy II, School of Medicine, Kanazawa Medical University, Uchinada, Ishikawa, 920-0293, Japan DOI: 10.1523/JNEUROSCI.2173-17.2018 Received: 31 July 2017 Revised: 2 February 2018 Accepted: 7 February 2018 Published: 24 February 2018 Author contributions: C.C., T.I., and S.S. designed research; C.C. and M.C. performed research; C.C. and T.I. analyzed data; C.C. wrote the first draft of the paper; C.C., T.I., and S.S. edited the paper; C.C., T.I., and S.S. wrote the paper. Conflict of Interest: The authors declare no competing financial interests. This work was supported by funding from the National Natural Science Foundation of China (31571095, 91332122, for S.S.), Special Fund of Suzhou-Tsinghua Innovation Leading Action (for S.S.), Beijing Program on the Study of Brain-Inspired Computing System and Related Core Technologies (for S.S.), Beijing Innovation Center for Future Chip (for S.S.), and Chinese Academy of Sciences Institute of Psychology Key Laboratory of Mental Health Open Research Grant (KLMH2012K02, for S.S.), grants from Ministry of Education, Science, and Culture of Japan (KAKENHI grant, Grant numbers 16K07026 and 16H01501; for T.I.), and Takahashi Industrial and Economic Research Foundation (for T.I.). -

Brainstem Exclusive of the Pyramids Themselves, Which Were Blood-Supplied Through the Intact Basilar Artery
NOTE ON CONVERGENCE OF PYRAMIDAL AND PRIMARY AFFERENT IMPULSES IN THE SPINAL CORD OF THE CAT BY DAVID P. C. LLOYD THE ROCKEFELLER UNIVERSITY Communicated December 20, 1967 The burden of this note is to present an example of observations long since made but not previously published. The immediate stimulus for presenting them now was provided by the paper by R. Porter' in which a combination of im- pulses of cortical and of lingual nerve origin was shown by convergence to facilitate the response of interneurons in or near the spinal trigeminal nucleus to a level of activity greater than that of the sum of responses elicited by lingual nerve stimu- lation and corticospinal stimulation, respectively, in isolation. Experiments of the sort now to be described concern the convergence upon interneurons in the lumbar spinal enlargement of pyramidal tract impulses and primary afferent im- pulses engendered by stimulation of the seventh lumbar dorsal root. To an ex- tent these experiments are confirmatory, with respect to another location in the neuraxis, of the observations of Porter,' but they demonstrate, in addition, how one pathway by occlusion may pre-empt the interneuron from service to the other pathway. In a prior paper2 convergence at the internuncial level was im- plied by virtue of pyramidal tract facilitatory influence upon disynaptic (three- neuron-arc) reflexes in the absence of any influence upon monosynaptic (two- neuron-arc) reflexes (ref. 2, Figs. 9 and 10). Preparation and procedure were discussed in the previous publication2 and need not be restated here, except to note that the pyramidal tract impulses were "pure" because the bulbar pyramid was stimulated rostral to a lesion that sev- ered the entire brainstem exclusive of the pyramids themselves, which were blood-supplied through the intact basilar artery. -

Imaging of the Confused Patient: Toxic Metabolic Disorders Dara G
Imaging of the Confused Patient: Toxic Metabolic Disorders Dara G. Jamieson, M.D. Weill Cornell Medicine, New York, NY The patient who presents with either acute or subacute confusion, in the absence of a clearly defined speech disorder and focality on neurological examination that would indicate an underlying mass lesion, needs to be evaluated for a multitude of neurological conditions. Many of the conditions that produce the recent onset of alteration in mental status, that ranges from mild confusion to florid delirium, may be due to infectious or inflammatory conditions that warrant acute intervention such as antimicrobial drugs, steroids or plasma exchange. However, some patients with recent onset of confusion have an underlying toxic-metabolic disorders indicating a specific diagnosis with need for appropriate treatment. The clinical presentations of some patients may indicate the diagnosis (e.g. hypoglycemia, chronic alcoholism) while the imaging patterns must be recognized to make the diagnosis in other patients. Toxic-metabolic disorders constitute a group of diseases and syndromes with diverse causes and clinical presentations. Many toxic-metabolic disorders have no specific neuroimaging correlates, either at early clinical stages or when florid symptoms develop. However, some toxic-metabolic disorders have characteristic abnormalities on neuroimaging, as certain areas of the central nervous system appear particularly vulnerable to specific toxins and metabolic perturbations. Areas of particular vulnerability in the brain include: 1) areas of high-oxygen demand (e.g. basal ganglia, cerebellum, hippocampus), 2) the cerebral white matter and 3) the mid-brain. Brain areas of high-oxygen demand are particularly vulnerable to toxins that interfere with cellular respiratory metabolism. -

DR. Sanaa Alshaarawy
By DR. Sanaa Alshaarawy 1 By the end of the lecture, students will be able to : Distinguish the internal structure of the components of the brain stem in different levels and the specific criteria of each level. 1. Medulla oblongata (closed, mid and open medulla) 2. Pons (caudal, mid “Trigeminal level” and rostral). 3. Mid brain ( superior and inferior colliculi). Describe the Reticular formation (structure, function and pathway) being an important content of the brain stem. 2 1. Traversed by the Central Canal. Motor Decussation*. Spinal Nucleus of Trigeminal (Trigeminal sensory nucleus)* : ➢ It is a larger sensory T.S of Caudal part of M.O. nucleus. ➢ It is the brain stem continuation of the Substantia Gelatinosa of spinal cord 3 The Nucleus Extends : Through the whole length of the brain stem and upper segments of spinal cord. It lies in all levels of M.O, medial to the spinal tract of the trigeminal. It receives pain and temperature from face, forehead. Its tract present in all levels of M.O. is formed of descending fibers that terminate in the trigeminal nucleus. 4 It is Motor Decussation. Formed by pyramidal fibers, (75-90%) cross to the opposite side They descend in the Decuss- = crossing lateral white column of the spinal cord as the lateral corticospinal tract. The uncrossed fibers form the ventral corticospinal tract. 5 Traversed by Central Canal. Larger size Gracile & Cuneate nuclei, concerned with proprioceptive deep sensations of the body. Axons of Gracile & Cuneate nuclei form the internal arcuate fibers; decussating forming Sensory Decussation. Pyramids are prominent ventrally. 6 Formed by the crossed internal arcuate fibers Medial Leminiscus: Composed of the ascending internal arcuate fibers after their crossing. -

An EM Study of the Dorsal Nucleus of the Lateral Lemniscus: Inhibitory, Commissural, Synaptic Connections Between Ascending Auditory Pathways
The Journal of Neuroscience, March 1989, g(3): 987-982 An EM Study of the Dorsal Nucleus of the Lateral Lemniscus: Inhibitory, Commissural, Synaptic Connections Between Ascending Auditory Pathways Douglas L. Oliver and Amiram Shneiderman Department of Anatomy, The University of Connecticut Health Center, Farmington, Connecticut 06032 The dorsal nucleus of the lateral lemniscus (DNLL) and its tions between the nuclei. Crossed inhibitory connections in connections constitute one of the ascending auditory path- the DNLL pathway may be important in regulating the flow ways to the inferior colliculus. One notable feature of this of ascending auditory information. nucleus is the heavy commissural connections between DNLL on opposite sides of the midbrain. These commissural con- The dorsal nucleus of the lateral lemniscus (DNLL) holds a nections may have a significant impact on the ascending unique position in the auditory system. Astride the fibers of the pathway. In this study, the fine structure of DNLL in the cat lateral lemniscus,DNLL receives most of the sameprojections and its commissural connections were examined. Both an- that ascendto the inferior colliculus (Glendenning et al., 1981; terograde and retrograde transport methods were used si- Shneiderman et al., 1988); and DNLL also projects bilaterally multaneously at the EM level. Injections of 3H-leucine mixed to the inferior colliculus. Thus, the combined inputs and outputs with WGA-HRP were made in one DNLL. After axonal trans- of DNLL form a multisynaptic pathway, parallel to the other port, EM autoradiographic methods were used to identify the ascending pathways to the inferior colliculus (Goldberg and anterogradely labeled axonal endings from the opposite Moore, 1967; van Noort, 1969; Roth et al., 1978; Adams, 1979; DNLL. -

Spinal Cord Organization
Lecture 4 Spinal Cord Organization The spinal cord . Afferent tract • connects with spinal nerves, through afferent BRAIN neuron & efferent axons in spinal roots; reflex receptor interneuron • communicates with the brain, by means of cell ascending and descending pathways that body form tracts in spinal white matter; and white matter muscle • gives rise to spinal reflexes, pre-determined gray matter Efferent neuron by interneuronal circuits. Spinal Cord Section Gross anatomy of the spinal cord: The spinal cord is a cylinder of CNS. The spinal cord exhibits subtle cervical and lumbar (lumbosacral) enlargements produced by extra neurons in segments that innervate limbs. The region of spinal cord caudal to the lumbar enlargement is conus medullaris. Caudal to this, a terminal filament of (nonfunctional) glial tissue extends into the tail. terminal filament lumbar enlargement conus medullaris cervical enlargement A spinal cord segment = a portion of spinal cord that spinal ganglion gives rise to a pair (right & left) of spinal nerves. Each spinal dorsal nerve is attached to the spinal cord by means of dorsal and spinal ventral roots composed of rootlets. Spinal segments, spinal root (rootlets) nerve roots, and spinal nerves are all identified numerically by th region, e.g., 6 cervical (C6) spinal segment. ventral Sacral and caudal spinal roots (surrounding the conus root medullaris and terminal filament and streaming caudally to (rootlets) reach corresponding intervertebral foramina) collectively constitute the cauda equina. Both the spinal cord (CNS) and spinal roots (PNS) are enveloped by meninges within the vertebral canal. Spinal nerves (which are formed in intervertebral foramina) are covered by connective tissue (epineurium, perineurium, & endoneurium) rather than meninges. -

Auditory and Vestibular Systems Objective • to Learn the Functional
Auditory and Vestibular Systems Objective • To learn the functional organization of the auditory and vestibular systems • To understand how one can use changes in auditory function following injury to localize the site of a lesion • To begin to learn the vestibular pathways, as a prelude to studying motor pathways controlling balance in a later lab. Ch 7 Key Figs: 7-1; 7-2; 7-4; 7-5 Clinical Case #2 Hearing loss and dizziness; CC4-1 Self evaluation • Be able to identify all structures listed in key terms and describe briefly their principal functions • Use neuroanatomy on the web to test your understanding ************************************************************************************** List of media F-5 Vestibular efferent connections The first order neurons of the vestibular system are bipolar cells whose cell bodies are located in the vestibular ganglion in the internal ear (NTA Fig. 7-3). The distal processes of these cells contact the receptor hair cells located within the ampulae of the semicircular canals and the utricle and saccule. The central processes of the bipolar cells constitute the vestibular portion of the vestibulocochlear (VIIIth cranial) nerve. Most of these primary vestibular afferents enter the ipsilateral brain stem inferior to the inferior cerebellar peduncle to terminate in the vestibular nuclear complex, which is located in the medulla and caudal pons. The vestibular nuclear complex (NTA Figs, 7-2, 7-3), which lies in the floor of the fourth ventricle, contains four nuclei: 1) the superior vestibular nucleus; 2) the inferior vestibular nucleus; 3) the lateral vestibular nucleus; and 4) the medial vestibular nucleus. Vestibular nuclei give rise to secondary fibers that project to the cerebellum, certain motor cranial nerve nuclei, the reticular formation, all spinal levels, and the thalamus. -

Contribution of the Dorsal Nucleus of the Lateral Lemniscus to Binaural Responses in the Inferior Colliculus of the Rat: Interaural Time Delays
The Journal of Neuroscience, November 15, 1996, 16(22):7390–7397 Contribution of the Dorsal Nucleus of the Lateral Lemniscus to Binaural Responses in the Inferior Colliculus of the Rat: Interaural Time Delays Sean A. Kidd and Jack B. Kelly Laboratory of Sensory Neuroscience, Institute of Neuroscience and Department of Psychology, Carleton University, Ottawa, Ontario, Canada K1S 5B6 The contribution of the dorsal nucleus of the lateral lemniscus duced by ipsilateral stimulation. The duration of the inhibitory (DNLL) to binaural responses in the inferior colliculus of the rat effect varied from cell to cell but lasted as long as 20 msec in was determined for a wide range of interaural time differences some cases. Injection of kynurenic acid into the DNLL con- (ITDs). Single-unit action potentials were recorded from the tralateral to the recording site reduced the extent of both short inferior colliculus before and after local injection of the excita- (0–1 msec) and long-lasting (1–20 msec) inhibition in the inferior tory amino acid antagonist kynurenic acid into the DNLL. Bin- colliculus. No effect was seen after injections ipsilateral to the aural properties were determined by manipulating the time recording site. The data demonstrate that the DNLL plays an difference between paired clicks delivered to the ears ipsilateral important role in shaping ITD responses in the inferior colliculus and contralateral to the recording site. The probability of an and contributes to both the short and long-lasting inhibition action potential decreased as contralateral stimulation was produced by stimulation of the ipsilateral ear. delayed, relative to ipsilateral stimulation. -
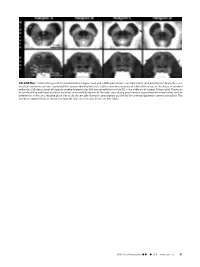
ON-LINE FIG 1. Selected Images of the Caudal Midbrain (Upper Row
ON-LINE FIG 1. Selected images of the caudal midbrain (upper row) and middle pons (lower row) from 4 of 13 total postmortem brains illustrate excellent anatomic contrast reproducibility across individual datasets. Subtle variations are present. Note differences in the shape of cerebral peduncles (24), decussation of superior cerebellar peduncles (25), and spinothalamic tract (12) in the midbrain of subject D (top right). These can be attributed to individual anatomic variation, some mild distortion of the brain stem during procurement at postmortem examination, and/or differences in the axial imaging plane not easily discernable during its prescription parallel to the anterior/posterior commissure plane. The numbers in parentheses in the on-line legends refer to structures in the On-line Table. AJNR Am J Neuroradiol ●:●●2019 www.ajnr.org E1 ON-LINE FIG 3. Demonstration of the dentatorubrothalamic tract within the superior cerebellar peduncle (asterisk) and rostral brain stem. A, Axial caudal midbrain image angled 10° anterosuperior to posteroinferior relative to the ACPC plane demonstrates the tract traveling the midbrain to reach the decussation (25). B, Coronal oblique image that is perpendicular to the long axis of the hippocam- pus (structure not shown) at the level of the ventral superior cerebel- lar decussation shows a component of the dentatorubrothalamic tract arising from the cerebellar dentate nucleus (63), ascending via the superior cerebellar peduncle to the decussation (25), and then enveloping the contralateral red nucleus (3). C, Parasagittal image shows the relatively long anteroposterior dimension of this tract, which becomes less compact and distinct as it ascends toward the thalamus. ON-LINE FIG 2. -

Inverse Density of Iba1 and Glutamine Synthetase Expressing Glia in Rat Inferior Colliculus
bioRxiv preprint doi: https://doi.org/10.1101/2021.06.30.450495; this version posted July 1, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. Inverse Density of Iba1 and Glutamine Synthetase Expressing Glia in Rat Inferior Colliculus Running title: Iba1 & Glutamine Synthetase in Inferior Colliculi Author names and affiliations: Llwyd David Orton Department of Life Sciences, Manchester Metropolitan University, Manchester, M1 5GD, UK Corresponding author: Llwyd Orton: [email protected]; +44 161 247 2483 Keywords: Auditory, Anatomy, Microglia, Astrocytes, Immunohistochemistry Contributions (CRediT): LDO: Conceptualization, Data curation, Formal analysis, Funding acquisition, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing – original draft, review and editing. Acknowledgements: The author thanks Dave Maskew, Megan Ryan and Rosie Mitchell for their technical support. Conflicting interests: The author declares no competing interests. bioRxiv preprint doi: https://doi.org/10.1101/2021.06.30.450495; this version posted July 1, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. Abstract Microglia and astrocytes undertake numerous essential roles in nervous systems but we know little of their anatomical distribution within numerous nuclei. In the principal nuclei of the mammalian auditory midbrain, the inferior colliculi (IC), the cellular density and relative distribution of glutamate synthetase (GS) expressing astrocytes and ionized calcium-binding adapter molecule 1 (Iba1) expressing microglia is unknown. -

(12) United States Patent (10) Patent No.: US 9,358,393 B1 L0zano (45) Date of Patent: Jun
US00935.8393B1 (12) United States Patent (10) Patent No.: US 9,358,393 B1 L0ZanO (45) Date of Patent: Jun. 7, 2016 (54) STIMULATION METHODS AND SYSTEMS 4,203,440 A 5/1980 Theeuwes FORTREATING AN AUDITORY 4,203.442 A 5/1980 Michaels DYSFUNCTION 4,210,139 A 7/1980 Higuchi (Continued) (76) Inventor: Andres M. Lozano, Toronto (CA) FOREIGN PATENT DOCUMENTS (*) Notice: Subject to any disclaimer, the term of this patent is extended or adjusted under 35 WO WOO1,08617 A1 2/2001 U.S.C. 154(b) by 1826 days. OTHER PUBLICATIONS (21) Appl. No.: 11/271,126 Office action dated Sep. 16, 2009 for related U.S. Appl. No. 1 1/271,688 (Lozano-1), filed Nov. 9, 2005, Inventor: Andres M. (22) Filed: Nov. 9, 2005 Lozano, (14 pages). Related U.S. Application Data (Continued) (60) gynal application No. 60/626,174, filed on Nov. Primary Examiner — Nicole F Lavert s (74) Attorney, Agent, or Firm — Faegre Baker Daniels LLP (51) Int. Cl. A61N L/00 (2006.01) (57) ABSTRACT A6 IB5/02 (2006.01) Methods of treating auditory hallucinations, hyperacusis, A6 IN L/36 (2006.01) Schizophrenia, and/or phonophobia include applying at least A61N L/05 (2006.01) one stimulus to a stimulation site within a patient with an (52) U.S. Cl. implanted stimulator in accordance with one or more stimu CPC .......... A61N I/36132 (2013.01); A61N I/0541 lation parameters. The stimulation site may include, for (2013.01) example, at least one or more of a cochlear nucleus, auditory (58) Field of Classification Search striae, Superior olivary complex, lateral lemniscus, inferior USPC ......................................