Neuronal Organization in the Inferior Colliculus Revisited with Cell-Type- Dependent Monosynaptic Tracing
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Superior and Inferior Colliculi of the Mole (Scalopus Aquaticus Machxinus)
THE SUPERIOR AND INFERIOR COLLICULI OF THE MOLE (SCALOPUS AQUATICUS MACHXINUS) THOMAS N. JOHNSON' Laboratory of Comparative Neurology, Departmmt of Amtomy, Un&versity of hfiehigan, Ann Arbor INTRODUCTION This investigation is a study of the afferent and efferent connections of the tectum of the midbrain in the mole (Scalo- pus aquaticus machrinus). An attempt is made to correlate these findings with the known habits of the animal. A subterranean animal of the middle western portion of the United States, Scalopus aquaticus machrinus is the largest of the genus Scalopus and its habits have been more thor- oughly studied than those of others of this genus according to Jackson ('15) and Hamilton ('43). This animal prefers a well-drained, loose soil. It usually frequents open fields and pastures but also is found in thin woods and meadows. Following a rain, new superficial burrows just below the surface of the ground are pushed in all directions to facili- tate the capture of worms and other soil life. Ten inches or more below the surface the regular permanent highway is constructed; the mole retreats here during long periods of dry weather or when frost is in the ground. The principal food is earthworms although, under some circumstances, larvae and adult insects are the more usual fare. It has been demonstrated conclusively that, under normal conditions, moles will eat vegetable matter. It seems not improbable that they may take considerable quantities of it at times. A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy in the University of Michigan. -

MR Imaging of Primary Trochlear Nerve Neoplasms
707 MR Imaging of Primary Trochlear Nerve Neoplasms Lindell R. Gentry 1 We present the clinical, anatomic, and MR imaging findings in six patients with seven Rahul C. Mehta 1 primary trochlear nerve neoplasms, as well as the MR and clinical criteria that serve to Richard E. Appen2 establish the diagnosis of these rare cranial nerve neoplasms. Three patients had a Joel M. Weinstein2 history of neurofibromatosis and five patients had clinical evidence of a trochlear nerve palsy. Six of seven neoplasms produced localized, fusiform enlargement of the proximal cisternal segments of the trochlear nerves. The lesions that were visible on noncontrast MR scans (T1-, T2-, and proton density-weighted) had signal intensities that were virtually identical to normal brain parenchyma. All lesions showed intense, homogeneous enhancement on contrast-enhanced scans. Contrast-enhanced imaging was necessary for the detection of five of seven lesions and greatly increased the value of the MR study in all six patients. AJNR 12:707-713, July/August 1991; AJR 157: September 1991 Primary neoplasms arising from pure motor cranial nerves such as the trochlear nerve are rare. To our knowledge just nine primary trochlear nerve neoplasms have been reported in the literature [1-7), only one of which was imaged with MR [1). Over the last 6 years, since we began to use MR as the primary imaging method for evaluating patients with cranial nerve palsies , we have noticed a striking increase in the number of primary trochlear nerve neoplasms over those encountered during the CT era. We believe this is due to a much greater sensitivity of MR in detecting these typically small lesions. -

DR. Sanaa Alshaarawy
By DR. Sanaa Alshaarawy 1 By the end of the lecture, students will be able to : Distinguish the internal structure of the components of the brain stem in different levels and the specific criteria of each level. 1. Medulla oblongata (closed, mid and open medulla) 2. Pons (caudal, mid “Trigeminal level” and rostral). 3. Mid brain ( superior and inferior colliculi). Describe the Reticular formation (structure, function and pathway) being an important content of the brain stem. 2 1. Traversed by the Central Canal. Motor Decussation*. Spinal Nucleus of Trigeminal (Trigeminal sensory nucleus)* : ➢ It is a larger sensory T.S of Caudal part of M.O. nucleus. ➢ It is the brain stem continuation of the Substantia Gelatinosa of spinal cord 3 The Nucleus Extends : Through the whole length of the brain stem and upper segments of spinal cord. It lies in all levels of M.O, medial to the spinal tract of the trigeminal. It receives pain and temperature from face, forehead. Its tract present in all levels of M.O. is formed of descending fibers that terminate in the trigeminal nucleus. 4 It is Motor Decussation. Formed by pyramidal fibers, (75-90%) cross to the opposite side They descend in the Decuss- = crossing lateral white column of the spinal cord as the lateral corticospinal tract. The uncrossed fibers form the ventral corticospinal tract. 5 Traversed by Central Canal. Larger size Gracile & Cuneate nuclei, concerned with proprioceptive deep sensations of the body. Axons of Gracile & Cuneate nuclei form the internal arcuate fibers; decussating forming Sensory Decussation. Pyramids are prominent ventrally. 6 Formed by the crossed internal arcuate fibers Medial Leminiscus: Composed of the ascending internal arcuate fibers after their crossing. -

Auditory and Vestibular Systems Objective • to Learn the Functional
Auditory and Vestibular Systems Objective • To learn the functional organization of the auditory and vestibular systems • To understand how one can use changes in auditory function following injury to localize the site of a lesion • To begin to learn the vestibular pathways, as a prelude to studying motor pathways controlling balance in a later lab. Ch 7 Key Figs: 7-1; 7-2; 7-4; 7-5 Clinical Case #2 Hearing loss and dizziness; CC4-1 Self evaluation • Be able to identify all structures listed in key terms and describe briefly their principal functions • Use neuroanatomy on the web to test your understanding ************************************************************************************** List of media F-5 Vestibular efferent connections The first order neurons of the vestibular system are bipolar cells whose cell bodies are located in the vestibular ganglion in the internal ear (NTA Fig. 7-3). The distal processes of these cells contact the receptor hair cells located within the ampulae of the semicircular canals and the utricle and saccule. The central processes of the bipolar cells constitute the vestibular portion of the vestibulocochlear (VIIIth cranial) nerve. Most of these primary vestibular afferents enter the ipsilateral brain stem inferior to the inferior cerebellar peduncle to terminate in the vestibular nuclear complex, which is located in the medulla and caudal pons. The vestibular nuclear complex (NTA Figs, 7-2, 7-3), which lies in the floor of the fourth ventricle, contains four nuclei: 1) the superior vestibular nucleus; 2) the inferior vestibular nucleus; 3) the lateral vestibular nucleus; and 4) the medial vestibular nucleus. Vestibular nuclei give rise to secondary fibers that project to the cerebellum, certain motor cranial nerve nuclei, the reticular formation, all spinal levels, and the thalamus. -

Projections of Physiologically Defined Subdivisions of the Inferior Colliculus in the Mustached Bat: Targets in the Medial Geniculate Body and Extrathalamic Nuclei
THE JOURNAL OF COMPARATIVE NEUROLOGY 346207-236 (1994) Projections of Physiologically Defined Subdivisions of the Inferior Colliculus in the Mustached Bat: Targets in the Medial Geniculate Body and Extrathalamic Nuclei JEFFREY J. WENSTRUP, DAVID T. LARUE, AND JEFFERY A. WINER Department of Neurobiology, Northeastern Ohio Universities College of Medicine, Rootstown, Ohio 44272-0095 (J.J.W.) and Division of Neurobiology, Department of Molecular and Cell Biology, University of California at Berkeley, Berkeley, California 94720-3200 (J.J.W.,D.T.L., J.A.W.) ABSTRACT This study examined the output of the central nucleus of the inferior colliculus to the medial geniculate body and other parts of the nervous system in the mustached bat (Pteronotus parnellii). Small deposits of anterograde tracers (horseradish peroxidase, [3Hlleucine, Phaseo- lus uulgaris leucoagglutinin, wheat germ agglutinin conjugated to horseradish peroxidase, or biocytin) were made at physiologically defined sites in the central nucleus representing major components of the bat’s echolocation signal. The topography, frequency specificity, and axonal morphology of these outputs were studied. The medial geniculate body was a major target of inferior collicular neurons, with three distinct input patterns. The projection to the ventral division was tonotopically organized, but had a relatively sparse contribution from neurons representing frequency modulated compo- nents of the biosonar pulse. The second input was to the rostral medial geniculate body, in which projections from inferior collicular neurons representing constant frequency sonar components were separated from those representing frequency modulated components. A third input was to the suprageniculate nucleus, which received strong, topographically arranged projections. Inputs to the dorsal nucleus and medial division were also observed. -
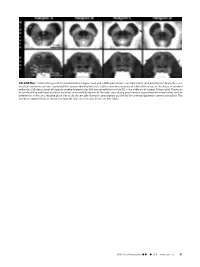
ON-LINE FIG 1. Selected Images of the Caudal Midbrain (Upper Row
ON-LINE FIG 1. Selected images of the caudal midbrain (upper row) and middle pons (lower row) from 4 of 13 total postmortem brains illustrate excellent anatomic contrast reproducibility across individual datasets. Subtle variations are present. Note differences in the shape of cerebral peduncles (24), decussation of superior cerebellar peduncles (25), and spinothalamic tract (12) in the midbrain of subject D (top right). These can be attributed to individual anatomic variation, some mild distortion of the brain stem during procurement at postmortem examination, and/or differences in the axial imaging plane not easily discernable during its prescription parallel to the anterior/posterior commissure plane. The numbers in parentheses in the on-line legends refer to structures in the On-line Table. AJNR Am J Neuroradiol ●:●●2019 www.ajnr.org E1 ON-LINE FIG 3. Demonstration of the dentatorubrothalamic tract within the superior cerebellar peduncle (asterisk) and rostral brain stem. A, Axial caudal midbrain image angled 10° anterosuperior to posteroinferior relative to the ACPC plane demonstrates the tract traveling the midbrain to reach the decussation (25). B, Coronal oblique image that is perpendicular to the long axis of the hippocam- pus (structure not shown) at the level of the ventral superior cerebel- lar decussation shows a component of the dentatorubrothalamic tract arising from the cerebellar dentate nucleus (63), ascending via the superior cerebellar peduncle to the decussation (25), and then enveloping the contralateral red nucleus (3). C, Parasagittal image shows the relatively long anteroposterior dimension of this tract, which becomes less compact and distinct as it ascends toward the thalamus. ON-LINE FIG 2. -

(12) United States Patent (10) Patent No.: US 9,358,393 B1 L0zano (45) Date of Patent: Jun
US00935.8393B1 (12) United States Patent (10) Patent No.: US 9,358,393 B1 L0ZanO (45) Date of Patent: Jun. 7, 2016 (54) STIMULATION METHODS AND SYSTEMS 4,203,440 A 5/1980 Theeuwes FORTREATING AN AUDITORY 4,203.442 A 5/1980 Michaels DYSFUNCTION 4,210,139 A 7/1980 Higuchi (Continued) (76) Inventor: Andres M. Lozano, Toronto (CA) FOREIGN PATENT DOCUMENTS (*) Notice: Subject to any disclaimer, the term of this patent is extended or adjusted under 35 WO WOO1,08617 A1 2/2001 U.S.C. 154(b) by 1826 days. OTHER PUBLICATIONS (21) Appl. No.: 11/271,126 Office action dated Sep. 16, 2009 for related U.S. Appl. No. 1 1/271,688 (Lozano-1), filed Nov. 9, 2005, Inventor: Andres M. (22) Filed: Nov. 9, 2005 Lozano, (14 pages). Related U.S. Application Data (Continued) (60) gynal application No. 60/626,174, filed on Nov. Primary Examiner — Nicole F Lavert s (74) Attorney, Agent, or Firm — Faegre Baker Daniels LLP (51) Int. Cl. A61N L/00 (2006.01) (57) ABSTRACT A6 IB5/02 (2006.01) Methods of treating auditory hallucinations, hyperacusis, A6 IN L/36 (2006.01) Schizophrenia, and/or phonophobia include applying at least A61N L/05 (2006.01) one stimulus to a stimulation site within a patient with an (52) U.S. Cl. implanted stimulator in accordance with one or more stimu CPC .......... A61N I/36132 (2013.01); A61N I/0541 lation parameters. The stimulation site may include, for (2013.01) example, at least one or more of a cochlear nucleus, auditory (58) Field of Classification Search striae, Superior olivary complex, lateral lemniscus, inferior USPC ...................................... -

Commissural Projections of the Nuclei of the Lateral Lemniscus and Keuronal Degeneration Following Midline Transections in the Adult Rat By
Commissural Projections of the Nuclei of the Lateral Lemniscus and Keuronal Degeneration Following Midline Transections in the Adult Rat by Brian Anthony van Adel B .Sc. (Hons) Carleton University, 1995 A thesis subrnitted to the faculty of Graduate Studies and Research in partial fulnllment of the requirements of the degree of Master of Science Specialization in Neuroscience Deparûnent of Biology Ottawa-Carleton Institutes of Biology and Neuroscience Carleton University Ottawa, Ontano May, 1998 O copyright 1998, Brian Anthony van Adel National Library Bibliothèque nationale 1+1 .,nad, du Canada Acquisitions and Acquisitions et Bibliographie Services seMces bibliographiques 395 Wellington Street 395, rue Wellington OttawaON KtAW ûuawaON KtAON4 canada CaMda The author has granted a non- L'auteur a accordé une licence non exclusive licence allowing the exclusive permettant à la National Library of Canada to Bibliothèque nationale du Canada de reproduce, 10- disûiiute or sen reproduire, prêter, distribuer ou copies of this thesis in microform, vendre des copies de cette these sous paper or electronic formats. la forme de microfiche/fïlm, de reproduction sur papier ou sur format électronique. The author retains ownership of the L'auteur conserve la propriété du copyright in this thesis. Neither the droit d'auteur qui protège cette thèse. thesis nor substantial extracts fiom it Ni la thèse ni des extraits substantiels may be printed or otheMrise de celle-ci ne doivent être imprimés reproduced without the author's ou autrement reproduits sans son permission. autorisation. ABSTRACT The normal neuroanatomical organization of the rat's nuclei of the lateral lemniscus (nLL) was investigated (Experiment 1) as a prerequisite for a time course analysis of retrograde changes in commissural projec~glemniscd neurons following midline surgical transection of their axons (Expriment 2). -

THE CEREBELLAR VERMIS ROBUSTLY MODULATES NEURAL ACTIVITY in the INFERIOR COLLICULUS by Richard Jiang Sima
THE CEREBELLAR VERMIS ROBUSTLY MODULATES NEURAL ACTIVITY IN THE INFERIOR COLLICULUS by Richard Jiang Sima A dissertation submitted to Johns Hopkins University in conformity with the requirements for the degree of Doctor of Philosophy Baltimore, Maryland May 2019 © 2019 Richard Jiang Sima All Rights Reserved. Abstract of the Dissertation To survive in an ever-changing world, animals need to rapidly detect and appropriately respond to external stimuli. In the auditory system, the inferior colliculus (IC) is well-positioned as an obligatory auditory hub involved with making acousticomotor responses to initiate these behavioral responses to auditory stimuli. However, it is not well understood how the relevant contextual information needed to detect and respond appropriately to stimuli is conveyed. In this thesis, I investigate the cerebellar vermis as one possible source of this contextual information. My experiments revealed that optogenetic stimulation of the cerebellar vermis robustly modulates the majority of neurons throughout IC in awake mice head-fixed on a treadmill. Because I was to monitor the movement of the treadmill, I also found that vermis stimulation produces a similar behavioral response as auditory stimuli, and that vermis evoked activity in the IC is in part related to this motor response. Furthermore, my results show that animal running modulates IC activity by reducing auditory and vermis responses, while increasing spontaneous activity. Thesis Committee: Professor Sascha du Lac, Thesis Advisor Professor David J. Linden, Chair Professor Jeremiah Y. Cohen, Thesis Reader Professor Amanda M. Lauer Professor Elisabeth B. Glowatzki ii This thesis is dedicated to my mom and dad, Drs. Qing Jiang and Hong Sima, and to my sister, Renee Sima. -

Tonotopic Organization in the Depth of Human Inferior Colliculus
ORIGINAL RESEARCH ARTICLE published: 19 September 2013 HUMAN NEUROSCIENCE doi: 10.3389/fnhum.2013.00586 Tonotopic organization in the depth of human inferior colliculus David Ress 1* and Bharath Chandrasekaran2 1 Neuroscience Department, Center for Perceptual Systems, Imaging Research Center, Institute for Neuroscience, The University of Texas at Austin, Austin TX, USA 2 Department of Communication Sciences and Disorders, Center for Perceptual Systems, Institute for Neuroscience, The University of Texas at Austin, Austin, TX, USA Edited by: Experiments in animal models indicate that inferior colliculus (IC), the primary auditory mid- Florian Beissner, Martinos Center for brain structure, represents sound frequency in a particular spatial organization, a tonotopy, Biomedical Imaging, USA that proceeds from dorsal and superficial to ventral and deeper tissue. Experiments are Reviewed by: Elia Formisano, Maastricht University, presented that use high-resolution, sparse-sampling functional magnetic resonance imag- Netherlands ing (fMRI) at 3T to determine if tonotopic gradients can be reliably measured in human Marc Schönwiesner, University of IC using high-resolution fMRI. Stimuli were sequences of bandpass-filtered noise with dif- Montreal, Canada ferent center frequencies, presented sequentially while fMRI data were collected. Four *Correspondence: subjects performed an adaptive frequency-discrimination task throughout the experiment. David Ress, Neuroscience Results show statistically significant tonotopic gradients within both ICs of all subjects. Department, Baylor College of Medicine, 1 Baylor Plaza, S104N MS Frequency gradients as a function of depth were measured using surface-based analysis BCM240, Houston, TX 77030, USA methods that make virtual penetrations into the IC tissue. This organization was evident e-mail: [email protected] over substantial portions of the IC, at locations that are consistent with the expected loca- tion of the central nucleus of IC.The results confirm a laminar tonotopy in the human IC at 3T, but with a heterogeneous, patchy character. -

The Nuclear Pattern of the Nok-Tectal Portions of the Midbrain and Isthmus in the Opossum
THE NUCLEAR PATTERN OF THE NOK-TECTAL PORTIONS OF THE MIDBRAIN AND ISTHMUS IN THE OPOSSUM RUSSELL T. WOODBURNE Department of Anatomy, Uniwersity of Yichigan SIX PLATES (TWELVE FIGURES) INTRODUCTION It is logical that the present series of descriptions of the nuclear pattern of the midbrain tegmentum in mammals should begin with the account of this region in marsupials, since the American opossum presents a simplified and generalized type of mammalian midbrain. The material employed in the present study consists of toluidin blue series, cut in various planes, of the brain of the American opossum, Didelphis virginiana. These preparations are a part of the Huber Neurological Collection of the Department of Anatomy of the University of Michigan. The literature particularly pertinent to specific nuclear de- scriptions will be discussed in connection with such descrip- tions and the general literature dealing with other than marsupial forms is dealt with in other sections of this series of papers and complete reference made in the comprehensive bibliography. There are, however, certain papers of which some mention should be made. The series of papers by Castaldi ('23, '24, '26) gave the basis for the nomenclature and the general pattern of subdivision followed here. Tsai's ('25) account of portions of the marsupial midbrain, although con- cerned primarily with tectal and pretectal areas, gave some aid in orientation. Certain of the pretectal regions were con- sidered in the light of earlier accounts of Chu ( '32) and Bodian ('40). The text of Ariens Kappers, Huber and Crosby ('36) was used for general orientation and comparative information. -

A. Diencephalon B. Telencephalon C. Metencephalon D
SAMPLE TEST QUESTIONS Select the one best answer. l. The olfactory nerve is attached to the: A. Diencephalon B. Telencephalon C. Metencephalon D. Mesencephalon E. Myelencephalon 2. Following complete transection of a peripheral nerve, all the following may occur except: A. Chromatoloysis in the cell body. B. Degeneration of peripheral myelin distal to the site of injury. C. Degeneration of the axon distal to the site of injury. D. Immediate restoration of normal function following prompt surgical repair. E. Outgrowth of axonal sprouting from the proximal nerve stump .. 3. A lesion of the left hypoglossal nerve causes: A. Loss of taste sensation on the left side of the tongue. B. Deviation of the tongue to the right side, upon protrusion. C. Total inability to protrude the tongue. D. Deviation of the tongue to the left, upon protrusion. E. Hoarseness. 4. In the brain stem, the principle sensory decussation f0r general sensation ca~ried in the posterior columns of the spinal cord forms the: A. Medial lemniscus. B. Lateral lemniscus. C. Central tegmental fasciculus D. Medial longitudinal fasciculus E. Spinal lemniscus. 5. All are true of the lateral spinothalamic tract except: A. It caries sensations of pain and thermal sense. B. Fibers from sacral origin are located in the ventro-medial portion of the tract. C. Decussation occurs within one or two spinal segments of its origin. D. Fibers of cervical origin are located in the ventro-medial portion of the tract. E. It forms part of the brain stem lemniscal systems. 81 6. A unilateral lesion of the internal capsule involving the genu and the posterior limb, would cause: A.