Certificate of Analysis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Aminophylline Catalog Number A1755 Storage
Aminophylline Catalog Number A1755 Storage Temperature –20 °C Replacement for Catalog Number 216895 CAS RN 317-34-0 Storage/Stability Synonyms: theophylline hemiethylenediamine complex; Aminophylline should be kept tightly closed to prevent 3,7-dihydro-1,3-demethyl-1H-purine-2,6-dione CO2 absorption from the atmosphere, which leads to compound with 1,2-ethanediamine (2:1); formation of theophylline and decreased solubility in 1,2 3 (theophylline)2 • ethylenediamine aqueous solutions. Stock solutions should be protected from light and prevented from contact with Product Description metals.2 Molecular Formula: C7H8N4O2 ·1/2 (C2H8N2) Molecular Weight: 210.3 References 1. The Merck Index, 12th ed., Entry# 485. Aminophylline is a xanthine derivative which is a 2. Martindale: The Extra Pharmacopoeia, 31st ed., combination of theophylline and ethylenediamine that is Reynolds, J. E. F., ed., Royal Pharmaceutical more water soluble than theophylline alone. Society (London, England: 1996), pp. 1651-1652. Aminophylline has been widely used as an inhibitor of 3. Data for Biochemical Research, 3rd ed., Dawson, cAMP phosphodiesterase.3 R. M. C., et al., Oxford University Press (New York, NY: 1986), pp. 316-317. Aminophylline has been shown to limit 4. Pelech, S. L., et al., cAMP analogues inhibit phosphatidylcholine biosynthesis in cultured rat phosphatidylcholine biosynthesis in cultured rat hepatocytes.4 It has been used in studies of acute hepatocytes. J. Biol. Chem., 256(16), 8283-8286 hypoxemia in newborn and older guinea pigs.5 The (1981). effect of various xanthine derivatives, including 5. Crisanti, K. C., and Fewell, J. E., Aminophylline aminophylline, on activation of the cystic fibrosis alters the core temperature response to acute transmembrane conductance regulator (CFTR) chloride hypoxemia in newborn and older guinea pigs. -

Download Product Insert (PDF)
PRODUCT INFORMATION Proxyphylline Item No. 20937 CAS Registry No.: 603-00-9 Formal Name: 3,7-dihydro-7-(2-hydroxypropyl)-1,3- N dimethyl-1H-purine-2,6-dione O N Synonym: NSC 163343 C H N O MF: 10 14 4 3 N FW: 238.2 N Purity: ≥98% O UV/Vis.: λmax: 273, 324 nm Supplied as: A crystalline solid OH Storage: -20°C Stability: As supplied, 2 years from the QC date provided on the Certificate of Analysis, when stored properly Laboratory Procedures Proxyphylline is supplied as a crystalline solid. A stock solution may be made by dissolving the proxyphylline in the solvent of choice. Proxyphylline is soluble in organic solvents such as ethanol, DMSO, and dimethyl formamide (DMF), which should be purged with an inert gas. The solubility of proxyphylline in ethanol is approximately 1 mg/ml and approximately 10 mg/ml in DMSO and DMF. Further dilutions of the stock solution into aqueous buffers or isotonic saline should be made prior to performing biological experiments. Ensure that the residual amount of organic solvent is insignificant, since organic solvents may have physiological effects at low concentrations. Organic solvent-free aqueous solutions of proxyphylline can be prepared by directly dissolving the crystalline solid in aqueous buffers. The solubility of proxyphylline in PBS, pH 7.2, is approximately 1 mg/ml. We do not recommend storing the aqueous solution for more than one day. Description Proxyphylline is a methylxanthine derivative that has bronchodilatory actions.1 It has also been reported 2 to have vasodilatory and cardiac stimulatory effects. -

The Xanthines (Theobromine and Aminophyllin)
effect of this might conceivably escape detection in THE XANTHINES (THEOBROMINE AND individuals engaged in very heavy work. From this AMINOPHYLLINE) IN THE TREAT- point of view our subjects were particularly favorable for this since of as MENT OF CARDIAC PAIN study, most them, shown in the table, were not engaged in any occupation. HARRY M.D. GOLD, GLYCERYL TRINITRATE TEST NATHANIEL T. M.D. KWIT, Early in the course of the study it was believed AND desirable to HAROLD M.D. restrict the selection of patients to those OTTO, who could establish their qualifications for service in NEW YORK such a study as this by their ability to distinguish An endeavor was made in this study to secure evi- between the efficacy of glyceryl trinitrate taken under dence on the question of whether the xanthines relieve the tongue and a soluble placebo tablet taken in the cardiac pain. same manner for relief during attacks of pain. The SELECTION OF PATIENTS discovery of several patients who found the two equally effective those who had suffered an of The were 100 ambulant in attend- among attack subjects patients thrombosis and were to thoracic ance at the cardiac in whom the of coronary subject pain clinic, diagnosis on effort led us to abandon this restriction. arteriosclerotic heart disease with cardiac pain was made, in accordance with the nomenclature and criteria The results obtained in sixty patients in whom the the New York Heart Association.1 glyceryl trinitrate test was made are of some interest. adopted by These received trinitrate were selected from a total case load of patients glyceryl tablets, %0o They approxi- or cr 0.4 which were mately 700 patients, representing an average sample /4so grain (0.6 mg.), they of the cardiac clinic several racial directed to take under the tongue at the onset of an population, comprising attack of In of these of groups, both native and born. -

Chocolate, Theobromine, Dogs, and Other Great Stuff
Nancy Lowry, Professor of Chemistry, Hampshire College, Amherst, MA [email protected] Chocolate, Theobromine, Dogs, and Other Great Stuff. Chocolate is now considered a health food, according to many news reports. It provides a goodly dose of antioxidants, prolongs the lives of Dutch men, contains compounds that chemically echo tetrahydocannabinoid and encourage feelings of love, and it even “may halve the risk of dying,” according to a recent headline in the New Scientist. On the other hand, if chocolate is included in the diet in therapeutic doses, it will also most assuredly lead to obesity. Furthermore, the amounts of anandamide (the THC mimic) and phenylethylamine (the so-called “love” compound) are present in chocolate in very, very low amounts. And finally, we all have a 100% chance of dying at some time, so a headline that talks about cutting our chance of dying in half makes no sense. Nevertheless, chocolate is great stuff. It comes in many varieties. One end of the spectrum is bitter baking chocolate; adding sugar provides chocolate of various degrees of sweetness. Adding milk finally brings us to milk chocolate, which many people consider barely makes it over the line into chocolate. White chocolate is only cocoa butter fat, and really isn’t chocolate at all. Over 600 different molecules contribute to the taste of chocolate. Many people talk about the caffeine in chocolate, but there is relatively very little caffeine in chocolate; the compound that particularly characterizes chocolate is theobromine, a very close relative of caffeine. There is six to ten times more theobromine in chocolate than caffeine. -

Effect of Practical Timing of Dosage on Theophylline Blood Levels In
Arch Dis Child: first published as 10.1136/adc.53.2.167 on 1 February 1978. Downloaded from Archives of Disease in Childhood, 1978, 53, 167-182 Short reports Effect of practical timing of dosage Methods on theophylline blood levels in For the period of the study, the children did not asthmatic children treated with receive tea, which contains theophylline. choline theophyllinate GroupA. These childrenwere given choline theophyll- The bronchodilator effect of theophylline has been inate in a dosage of as near as possible 8 mg/kg per known for over 50 years, but although it has been dose qds (mean 7 9 mg/kg, range 7-2-8 7 mg/kg), widely used both intravenously and rectally in the starting with half the dose on the first day to try and treatment ofacute asthma, its use as an oral broncho- minimise nausea. The doses were given at 0800 h, dilator has been limited by inadequate knowledge of 1300 h, 1700 h, and 2100 h, times considered suitable the correct dosage of the oral preparations. The for administration outside hospital. On the third day therapeutic level of theophylline is generally con- ofthe study serum levels were measured 2 hours after sidered to be between 10 and 20 ,ug/ml (Turner- each dose and before the 0800 h dose the following Warwick, 1957; Jenne et al., 1972), although sub- morning. optimal bronchodilation can be achieved with levels of 5 ,ug/ml or less (Maselli et al., 1970; Nicholson and Group B. These children were given cholinetheophyll- Chick, 1973). Serious toxicity with levels of less than inate in a dosage of as near as possible 10 mg/kg per copyright. -

3-Methylxanthine Production Through Biodegradation of Theobromine By
Zhou et al. BMC Microbiology (2020) 20:269 https://doi.org/10.1186/s12866-020-01951-z RESEARCH ARTICLE Open Access 3-Methylxanthine production through biodegradation of theobromine by Aspergillus sydowii PT-2 Binxing Zhou1*† , Cunqiang Ma1,2,3*†, Chengqin Zheng1, Tao Xia4, Bingsong Ma1 and Xiaohui Liu1 Abstract Background: Methylxanthines, including caffeine, theobromine and theophylline, are natural and synthetic compounds in tea, which could be metabolized by certain kinds of bacteria and fungi. Previous studies confirmed that several microbial isolates from Pu-erh tea could degrade and convert caffeine and theophylline. We speculated that these candidate isolates also could degrade and convert theobromine through N-demethylation and oxidation. In this study, seven tea-derived fungal strains were inoculated into various theobromine agar medias and theobromine liquid mediums to assess their capacity in theobromine utilization. Related metabolites with theobromine degradation were detected by using HPLC in the liquid culture to investigate their potential application in the production of 3-methylxanthine. Results: Based on theobromine utilization capacity, Aspergillus niger PT-1, Aspergillus sydowii PT-2, Aspergillus ustus PT-6 and Aspergillus tamarii PT-7 have demonstrated the potential for theobromine biodegradation. Particularly, A. sydowii PT-2 and A. tamarii PT-7 could degrade theobromine significantly (p < 0.05) in all given liquid mediums. 3,7- Dimethyluric acid, 3-methylxanthine, 7-methylxanthine, 3-methyluric acid, xanthine, and uric acid were detected in A. sydowii PT-2 and A. tamarii PT-7 culture, respectively, which confirmed the existence of N-demethylation and oxidation in theobromine catabolism. 3-Methylxanthine was common and main demethylated metabolite of theobromine in the liquid culture. -

Enabling Direct Preferential Crystallization in a Stable Racemic
Enabling Direct Preferential Crystallization in a Stable Racemic Compound System Lina Harfouche, Clément Brandel, Yohann Cartigny, Joop ter Horst, Gérard Coquerel, Samuel Petit To cite this version: Lina Harfouche, Clément Brandel, Yohann Cartigny, Joop ter Horst, Gérard Coquerel, et al.. Enabling Direct Preferential Crystallization in a Stable Racemic Compound System. Molecular Pharmaceutics, American Chemical Society, 2019, 16 (11), pp.4670-4676. 10.1021/acs.molpharmaceut.9b00805. hal- 02331962 HAL Id: hal-02331962 https://hal-normandie-univ.archives-ouvertes.fr/hal-02331962 Submitted on 24 Oct 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Enabling Direct Preferential Crystallization in a Stable Racemic Compound System Lina C. Harfouche,† Clément Brandel,†* Yohann Cartigny,† Joop H. ter Horst,‡ Gérard Coquerel,† and Samuel Petit† † Universite de Rouen Normandie, UFR des Sciences et Techniques, Laboratoire SMS-EA3233, Place Emile Blondel, 76821, Mont-Saint-Aignan, France ‡EPSRC Centre for Innovative Manufacturing in Continuous Manufacturing and Crystallisation (CMAC), Strathclyde Insti- tute of Pharmacy and Biomedical Sciences (SIPBS), Technology and Innovation Centre, University of Strathclyde, 99 George Street, Glasgow G1 1RD (UK) Supporting Information Placeholder 8 ABSTRACT: The preparative resolution by preferential crystal- separation methods, including chiral chromatography and enzy- 9 lization (PC) of proxyphylline has been achieved despite the matic resolution. -
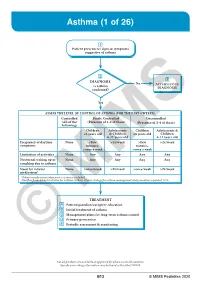
Asthma (1 of 26)
Asthma (1 of 26) 1 Patient presents w/ signs & symptoms suggestive of asthma 2 3 DIAGNOSIS No ALTERNATIVE Is asthma DIAGNOSIS confi rmed? Yes ASSESS THE LEVEL OF CONTROL OF ASTHMA FOR THE PAST 4 WEEKS Controlled Partly Controlled Uncontrolled (All of the (Presence of 1-2 of these) (Presence of 3-4 of these) following) Children Adolescents Children Adolescents & ≤5 years old & Children ≤5 years old Children 6-11 years old 6-11 years old Frequency of daytime None >Few >2x/week >Few >2x/week symptoms minutes, minutes, >once a week >once a week Limitation of activities None Any Any Any Any Nocturnal waking up or None Any Any Any Any coughing due to asthma Need for reliever None >once/week >2x/week >once/week >2x/week medication* *Reliever medications taken prior to exercise excluded. Modified from: Global Initiative for Asthma (GINA). Global strategy for asthma management and prevention: Updated 2020. TREATMENT A Patient/guardian/caregiver education B Initial treatment of asthma C Management plans for long-term asthma control D Primary prevention E © Periodic assessmentMIMS & monitoring Not all products are available or approved for above use in all countries. Specifi c prescribing information may be found in the latest MIMS. B13 © MIMS Pediatrics 2020 Asthma (2 of 26) 1 ASTHMA • A heterogeneous disease w/ chronic infl ammatory disorder of the airways • e most common chronic disease in pediatric age groups that causes signifi cant morbidity • Characterized by history of respiratory symptoms eg wheeze, shortness of breath, chest tightness & cough -

Exploring Cocoa Properties: Is Theobromine a Cognitive Modulator?
Psychopharmacology https://doi.org/10.1007/s00213-019-5172-0 REVIEW Exploring cocoa properties: is theobromine a cognitive modulator? Ilaria Cova1 & V. Leta1,2 & C. Mariani2 & L. Pantoni2 & S. Pomati1 Received: 7 May 2018 /Accepted: 16 January 2019 # Springer-Verlag GmbH Germany, part of Springer Nature 2019 Abstract Nutritional qualities of cocoa have been acknowledged by several authors; a particular focus has been placed on its high content of flavanols, known for their excellent antioxidant properties and subsequent protective effect on cardio- and cerebrovascular systems as well as for neuromodulatory and neuroprotective actions. Other active components of cocoa are methylxanthines (caffeine and theobromine). Whereas the effects of caffeine are extensively researched, the same is not the case for theobromine; this review summarizes evidence on the effect of theobromine on cognitive functions. Considering animal studies, it can be asserted that acute exposition to theobromine has a reduced and delayed nootropic effect with respect to caffeine, whereas both animal and human studies suggested a potential neuroprotective action of long-term assumption of theobromine through a reduction of Aβ amyloid pathology, which is commonly observed in Alzheimer’s disease patients’ brains. Hence, the conceiv- able action of theobromine alone and associated with caffeine or other cocoa constituents on cognitive modulation is yet underexplored and future studies are needed to shed light on this promising molecule. Keywords Cocoa . Theobromine . Cognitive modulator . Cognition Introduction predominant fraction of triglyceride molecules species (in particular oleic, stearic, palmitic, and linoleic acid) Theobroma cacao and its products (Pittenauer and Allmaier 2009); proteins contribute to 10–15% of the dry weight of cocoa seeds and consist Cocoa comes from the processing of seeds of a tropical tree, mainly of albumin and globulin fractions (Zak and considered by the Aztecs as a sacred plant. -

Caffeine and Theobromine Identifications in Post
MEDICINE—EXERCISE Caffeine and Theobromine Identifications in Post- Race Urines: Threshold Levels and Regulatory Significance of Such Identifications Amit Budhraja, BVSc, AH; Fernanda C. Camargo, DVM, PhD; Charlie Hughes, BS, MS; Andreas F. Lehner, BS, MS, PhD; Kent Stirling, BBA; Noel Brennan, BSc; Mark Dowling, BSc; and Thomas Tobin, MVB, MSc, PhD, MRCVS, Diplomate ABT* Caffeine is a widespread environmental substance, and testing for caffeine is now highly sensi- tive. Currently, many authorities in North America are not concerned about plasma caffeine concentrations of Ͻ100 ng/ml, equivalent to ϳ300 ng/ml in urine. This is because such low caffeine concentrations are likely to be associated with environmental exposure to caffeine and are also unlikely to be associated with pharmacological responses. Authors’ addresses: Gradu- ate Center for Toxicology, Health Sciences Research Building, University of Kentucky, Lexington, KY 40536 (Budhraja); Maxwell Gluck Equine Research Center, Farm Lane and Nicholasville Road, University of Kentucky, Lexington, KY 40546 (Camargo, Hughes, Tobin); U.K. Livestock Disease Diagnostic Center, 1490 Bull Lea Road, Lexington, KY 40512 (Lehner); Florida Horse- men’s Benevolent and Protective Association, PO Box 1808 Calder race course, Opa-Locka, FL 33056 (Stirling); and Connolly’s Redmills, Goresbridge, Kilkenny, Ireland (Brennan, Dowling); e-mail: [email protected] (Tobin). © 2007 AAEP. *Presenting author. 1. Introduction In horses, caffeine is metabolized to theophylline, Caffeine and theobromine are commonly found in theobromine, and paraxanthine. These metabo- equine feeds and environments. They are well lites are excreted at high concentrations in equine absorbed and may appear at microgram concen- urine. Caffeine identifications in the absence of trations in equine blood and urine; enzyme-linked these metabolites suggest post-collection entry of immunosorbent assay (ELISA) tests can detect caffeine into the sample. -

High Throughput Separation of Xanthines by Rapid Resolution HPLC Application Note
High Throughput Separation of Xanthines by Rapid Resolution HPLC Application Note Biochemistry, Food and Beverage, Pharmaceutical Author from 8 minutes to 1.5 minutes without a major loss in resolution. A simple isocratic HPLC method was used John W. Henderson and Ronald E. Majors and described to analyze theobromine, theophylline and Agilent Technologies, Inc. caffeine in liquid refreshments (tea, chocolate syrup, and 2850 Centerville Road cocoa). Wilmington, DE 19808-1610 USA Introduction Abstract Xanthines are a group of alkaloids with the most common xanthine being caffeine, found in foods such as coffee Xanthines were found to be optimally separated by beans, tea, kola nuts, and in small amounts in cacao reversed-phase HPLC on a C18 column. By a reduction in beans. The chemical structures of these xanthines and column length and particle size, the separation time for some of their metabolites are depicted in Figure 1. a mixture of the xanthines investigated was reduced H O H O O HC O CH3 3 CH H HC H N N 3 N N 3 N N N N N O N N O O N N N O CH3 H CH3 CH3 Theophylline Basic xanthine Caffeine Theobromine (1,3-dimethylxanthine) structure (1,3,7-trimethylxanthine) (3,7-dimethylxanthine) O H O O H CH3 HC N HC HC 3 N 3 N 3 N O O N N O N N O N N H CH3 H H 1,3-dimethyluric acid 1,7-dimethylxanthine 1-methylxanthine (theophylline metabolite) (caffeine metabolite) (theophylline metabolite) Figure 1. Structures of selected xanthines and metabolites used in this study. -

Xanthine Urolithiasis: Inhibitors of Xanthine Crystallization
bioRxiv preprint doi: https://doi.org/10.1101/335364; this version posted May 31, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. Xanthine urolithiasis: Inhibitors of xanthine crystallization Authors: Felix Grases, Antonia Costa-Bauza, Joan Roig, Adrian Rodriguez Laboratory of Renal Lithiasis Research, Faculty of Sciences, University Institute of Health Sciences Research (IUNICS-IdISBa), University of Balearic Islands, Ctra. de Valldemossa, km 7.5, 07122 Palma de Mallorca, Spain. Corresponding author: Felix Grases. Laboratory of Renal Lithiasis Research, Faculty of Sciences, University Institute of Health Sciences Research (IUNICS), University of Balearic Islands, Ctra. de Valldemossa, km 7.5, 07122 Palma de Mallorca, Spain. Telephone number: +34 971 17 32 57 e-mail: [email protected] Key words: xanthine lithiasis, 7-Methylxanthine, 3-Methylxanthine, Theobromine. 1 bioRxiv preprint doi: https://doi.org/10.1101/335364; this version posted May 31, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. Abstract OBJECTIVE. To identify in vitro inhibitors of xanthine crystallization that have potential for inhibiting the formation of xanthine crystals in urine and preventing the development of the renal calculi in patients with xanthinuria. METHODS. The formation of xanthine crystals in synthetic urine and the effects of 10 potential crystallization inhibitors were assessed using a kinetic turbidimetric system with a photometer.