Co-Deletion of P18 and P16 /P14 /P15 in Glioblastoma Multiforme
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Expression Profiling of KLF4
Expression Profiling of KLF4 AJCR0000006 Supplemental Data Figure S1. Snapshot of enriched gene sets identified by GSEA in Klf4-null MEFs. Figure S2. Snapshot of enriched gene sets identified by GSEA in wild type MEFs. 98 Am J Cancer Res 2011;1(1):85-97 Table S1: Functional Annotation Clustering of Genes Up-Regulated in Klf4 -Null MEFs ILLUMINA_ID Gene Symbol Gene Name (Description) P -value Fold-Change Cell Cycle 8.00E-03 ILMN_1217331 Mcm6 MINICHROMOSOME MAINTENANCE DEFICIENT 6 40.36 ILMN_2723931 E2f6 E2F TRANSCRIPTION FACTOR 6 26.8 ILMN_2724570 Mapk12 MITOGEN-ACTIVATED PROTEIN KINASE 12 22.19 ILMN_1218470 Cdk2 CYCLIN-DEPENDENT KINASE 2 9.32 ILMN_1234909 Tipin TIMELESS INTERACTING PROTEIN 5.3 ILMN_1212692 Mapk13 SAPK/ERK/KINASE 4 4.96 ILMN_2666690 Cul7 CULLIN 7 2.23 ILMN_2681776 Mapk6 MITOGEN ACTIVATED PROTEIN KINASE 4 2.11 ILMN_2652909 Ddit3 DNA-DAMAGE INDUCIBLE TRANSCRIPT 3 2.07 ILMN_2742152 Gadd45a GROWTH ARREST AND DNA-DAMAGE-INDUCIBLE 45 ALPHA 1.92 ILMN_1212787 Pttg1 PITUITARY TUMOR-TRANSFORMING 1 1.8 ILMN_1216721 Cdk5 CYCLIN-DEPENDENT KINASE 5 1.78 ILMN_1227009 Gas2l1 GROWTH ARREST-SPECIFIC 2 LIKE 1 1.74 ILMN_2663009 Rassf5 RAS ASSOCIATION (RALGDS/AF-6) DOMAIN FAMILY 5 1.64 ILMN_1220454 Anapc13 ANAPHASE PROMOTING COMPLEX SUBUNIT 13 1.61 ILMN_1216213 Incenp INNER CENTROMERE PROTEIN 1.56 ILMN_1256301 Rcc2 REGULATOR OF CHROMOSOME CONDENSATION 2 1.53 Extracellular Matrix 5.80E-06 ILMN_2735184 Col18a1 PROCOLLAGEN, TYPE XVIII, ALPHA 1 51.5 ILMN_1223997 Crtap CARTILAGE ASSOCIATED PROTEIN 32.74 ILMN_2753809 Mmp3 MATRIX METALLOPEPTIDASE -

Charles M. Perou, Phd Associate Professor Departments of Genetics
Charles M. Perou, PhD Associate Professor Departments of Genetics and Pathology Carolina Center for Genome Sciences Lineberger Comprehensive Cancer Center University of North Carolina at Chapel Hill Identification of GBM Subtypes using Gene Expression Profiling Agilent Custom Affymetrix Human Affymetrix HT-HG- 244k Exon 1.0 Array U133A Array Microarray Identify Samples and Genes represented on all 3 platforms Factor Analysis Single unified gene expression measurement for each gene 202 patients 11,681 genes Identification of GBM Subtypes Census Clustering of the 202 samples X 1740 Unsupervised clustering of 1740 genes suggests that 4 subtypes of GBM exist variably expressed genes selected using a unified gene expression measure across 3 expression platforms Core TCGA Samples (173) Gene Ontology/Pathway: with Subtype-defining genes • ProNeural: 1. nervous system development ProNeural Normal-like EGFR Mesenchymal 2. neuron differentiation (SOXs) 3. cell cycle = proliferation 4. cell adhesion molecules 5. ErbB signaling pathway FBXO3 GABRB2 • Normal‐like: SNCG NTSR2 1. nucleotide metabolic process MBP 2. neurological system process DLL3 3. axon NKX2-2, NRXN1, NRXN2 TOP2B, CDC7, MYB 4. neuron projection SOX2, SOX4, SOX10,SOX11 5. synaptic transmission ERBB3, PAK3, PAK7 NCAM1 OLIG2 • EGFR: 1. regulation of transcription 2. cell migration 3. nervous system development FGFR3 4. cell proliferation PDGFRA 5. metal ion binding EGFR AKT2 GLI2 TGFB3 • Mesenchymal: CASP8, CASP5, CASP4, CASP1 1. immune response COL8A2, COL5A1, COL1A1,COL1A2 2. receptor activity ILR4 CHI3L1 3. wound healing TRADD TLR2, TLR4, 4. cytokine and chemokine IGFBI mediated signaling pathway RELB 5. NF‐B Signaling Pathway Correlations between gene expression subtypes and clinical parameters Survival Analysis of Subtypes n=196 Subtypes are correlated with: 1. -
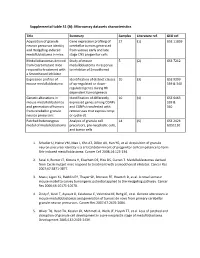
Supplemental Table S1 (A): Microarray Datasets Characteristics
Supplemental table S1 (A): Microarray datasets characteristics Title Summary Samples Literature ref. GEO ref. Acquisition of granule Gene expression profiling of 27 (1) GSE 11859 neuron precursor identity cerebellar tumors generated and Hedgehog‐induced from various early and late medulloblastoma in mice. stage CNS progenitor cells Medulloblastomas derived Study of mouse 5 (2) GSE 7212 from Cxcr6 mutant mice medulloblastoma in response respond to treatment with to inhibitor of Smoothened a Smoothened inhibitor Expression profiles of Identification of distinct classes 10 (3) GSE 9299 mouse medulloblastoma of up‐regulated or down‐ 339 & 340 regulated genes during Hh dependent tumorigenesis Genetic alterations in Identification of differently 10 (4) GSE 6463 mouse medulloblastomas expressed genes among CGNPs 339 & and generation of tumors and CGNPs transfected with 340 from cerebellar granule retroviruses that express nmyc neuron precursors or cyclin‐d1 Patched heterozygous Analysis of granule cell 14 (5) GSE 2426 model of medulloblastoma precursors, pre‐neoplastic cells, GDS1110 and tumor cells 1. Schuller U, Heine VM, Mao J, Kho AT, Dillon AK, Han YG, et al. Acquisition of granule neuron precursor identity is a critical determinant of progenitor cell competence to form Shh‐induced medulloblastoma. Cancer Cell 2008;14:123‐134. 2. Sasai K, Romer JT, Kimura H, Eberhart DE, Rice DS, Curran T. Medulloblastomas derived from Cxcr6 mutant mice respond to treatment with a smoothened inhibitor. Cancer Res 2007;67:3871‐3877. 3. Mao J, Ligon KL, Rakhlin EY, Thayer SP, Bronson RT, Rowitch D, et al. A novel somatic mouse model to survey tumorigenic potential applied to the Hedgehog pathway. Cancer Res 2006;66:10171‐10178. -

Tagging Single Nucleotide Polymorphisms in Cell Cycle Control Genes and Susceptibility to Invasive Epithelial Ovarian Cancer
Research Article Tagging Single Nucleotide Polymorphisms in Cell Cycle Control Genes and Susceptibility to Invasive Epithelial Ovarian Cancer Simon A. Gayther,1 Honglin Song,2 Susan J. Ramus,1 Susan Kru¨ger Kjaer,4 Alice S. Whittemore,5 Lydia Quaye,1 Jonathan Tyrer,2 Danielle Shadforth,2 Estrid Hogdall,4 Claus Hogdall,6 Jan Blaeker,7 Richard DiCioccio,8 Valerie McGuire,5 Penelope M. Webb,9 Jonathan Beesley,9 Adele C. Green,9 David C. Whiteman,9 The Australian Ovarian Cancer Study Group,9 The Australian Cancer Study (Ovarian Cancer),20 Marc T. Goodman,10 Galina Lurie,10 Michael E. Carney,10 Francesmary Modugno,10 Roberta B. Ness,11 Robert P. Edwards,12 Kirsten B. Moysich,7 Ellen L. Goode,13 Fergus J. Couch,13 Julie M. Cunningham,13 Thomas A. Sellers,14 Anna H. Wu,15 Malcolm C. Pike,15 Edwin S. Iversen,16 Jeffrey R. Marks,16 Montserrat Garcia-Closas,17 Louise Brinton,17 Jolanta Lissowska,18 Beata Peplonska,19 Douglas F. Easton,3 Ian Jacobs,1 Bruce A.J. Ponder,2 Joellen Schildkraut,16 C. Leigh Pearce,15 Georgia Chenevix-Trench,9 Andrew Berchuck,16 Paul D.P. Pharoah,2 and on behalf of the Ovarian Cancer Association Consortium 1Translational Research Laboratories, University College London, London, United Kingdom; 2Cancer Research United Kingdom Department of Oncology and 3Cancer Research United Kingdom Genetic Epidemiology Unit, University of Cambridge, Strangeways Research Laboratory, Cambridge, United Kingdom; 4Danish Cancer Society, Copenhagen, Denmark; 5Stanford University School of Medicine, Stanford, California; 6University of Copenhagen, -

CDKN2C-Null Leiomyosarcoma: a Novel
original reports CDKN2C-Null Leiomyosarcoma: A Novel, Genomically Distinct Class of TP53/ RB1–Wild-Type Tumor With Frequent CIC Genomic Alterations and 1p/19q-Codeletion Erik A. Williams, MD1; Radwa Sharaf, PhD1; Brennan Decker, MD, PhD2; Adrienne J. Werth, MD3; Helen Toma, MD3; Meagan Montesion, PhD1; Ethan S. Sokol, PhD1; Dean C. Pavlick, BS1; Nikunj Shah, BS1; Kevin Jon Williams, MD4; Jeffrey M. Venstrom, MD1; Brian M. Alexander, MD, MPH1; Jeffrey S. Ross, MD1,5; Lee A. Albacker, PhD1; Douglas I. Lin, MD, PhD1; Shakti H. Ramkissoon, MD, PhD1,6; and Julia A. Elvin, MD, PhD1 abstract PURPOSE Leiomyosarcoma (LMS) harbors frequent mutations in TP53 and RB1 but few actionable genomic alterations. Here, we searched for recurrent actionable genomic alterations in LMS that occur in the absence of common untreatable oncogenic drivers. METHODS Tissues from 276,645 unique advanced cancers, including 2,570 uterine and soft tissue LMS, were sequenced by hybrid-capture–based next-generation DNA and RNA sequencing/comprehensive genomic profiling of up to 406 genes. We characterized clinicopathologic features of relevant patient cases. RESULTS Overall, 77 LMS exhibited homozygous copy loss of CDKN2C at chromosome 1p32.3 (3.0% of LMS). Genomic alterations (GAs) in TP53, RB1, and ATRX were rare compared with the remainder of the LMS cohort (11.7% v 73.4%, 0% v 54.5%, 2.6% v 24.5%, respectively; all P , .0001). CDKN2C-null LMS patient cases were significantly enriched for GAs in CIC (40.3% v 1.4%) at 19q13.2, CDKN2A (46.8% v 7.0%), and RAD51B (16.9% v 1.7%; all P , .0001). -

Deletions of CDKN2C in Multiple Myeloma: Biological and Clinical Implications Paola E
Human Cancer Biology Deletions of CDKN2C in Multiple Myeloma: Biological and Clinical Implications Paola E. Leone,1Brian A. Walker,1Matthew W. Jenner,1Laura Chiecchio,2 GianPaolo Dagrada,2 Rebecca K.M. Protheroe,2 David C. Johnson,1Nicholas J. Dickens,1Jose Luis Brito,1Monica Else,1 David Gonzalez,1Fiona M. Ross,2 Selina Chen-Kiang,3 Faith E. Davies,1and Gareth J. Morgan1 Abstract Purpose: Deletions of chromosome 1have been described in 7% to 40% of cases of myeloma with inconsistent clinical consequences. CDKN2C at 1p32.3 has been identified in myeloma cell lines as the potential target of the deletion.We tested the clinical impact of 1p deletion and used high-resolution techniques to define the role of CDKN2C in primary patient material. Experimental Design: We analyzed 515 cases of monoclonal gammopathy of undetermined significance (MGUS), smoldering multiple myeloma (SMM), and newly diagnosed multiple mye- loma using fluorescence in situ hybridization (FISH) for deletions of CDKN2C.In78myeloma cases, we carried out Affymetrix single nucleotide polymorphism mapping and U133 Plus 2.0 ex- pression arrays. In addition, we did mutation, methylation, andWestern blotting analysis. Results: By FISH we identified deletion of 1p32.3 (CDKN2C)in3of66MGUS(4.5%),4of39 SMM (10.3%), and 55 of 369 multiple myeloma cases (15%).We examined the impact of copy number change at CDKN2C on overall survival (OS), and found that the cases with either hemi- zygous or homozygous deletion of CDKN2C had a worse OS compared with cases that were intact at this region (22 months versus 38 months; P = 0.003). Using gene mapping we identified three homozygous deletions at 1p32.3, containing CDKN2C,allofwhichlackedexpressionof CDKN2C. -

Autoimmunity in a Mouse Model of Lupus Deficiency Promotes B1a
Cyclin-Dependent Kinase Inhibitor Cdkn2c Deficiency Promotes B1a Cell Expansion and Autoimmunity in a Mouse Model of Lupus This information is current as Hari-Hara S. K. Potula, Zhiwei Xu, Leilani Zeumer, Allison of September 25, 2021. Sang, Byron P. Croker and Laurence Morel J Immunol 2012; 189:2931-2940; Prepublished online 15 August 2012; doi: 10.4049/jimmunol.1200556 http://www.jimmunol.org/content/189/6/2931 Downloaded from Supplementary http://www.jimmunol.org/content/suppl/2012/08/15/jimmunol.120055 Material 6.DC1 http://www.jimmunol.org/ References This article cites 42 articles, 14 of which you can access for free at: http://www.jimmunol.org/content/189/6/2931.full#ref-list-1 Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision by guest on September 25, 2021 • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2012 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Cyclin-Dependent Kinase Inhibitor Cdkn2c Deficiency Promotes B1a Cell Expansion and Autoimmunity in a Mouse Model of Lupus Hari-Hara S. -

Cdkn2a, the Cyclin-Dependent Kinase Inhibitor Encoding P16ink4a and P19arf, Is a Candidate for the Plasmacytoma Susceptibility Locus, Pctr1
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 2429–2434, March 1998 Genetics Cdkn2a, the cyclin-dependent kinase inhibitor encoding p16INK4a and p19ARF, is a candidate for the plasmacytoma susceptibility locus, Pctr1 SHULING ZHANG,EDWARD S. RAMSAY, AND BEVERLY A. MOCK* Laboratory of Genetics, Division of Basic Sciences, National Cancer Institute, National Institutes of Health, Bethesda, MD 20892-4255 Communicated by Michael Potter, National Institutes of Health, Bethesda, MD, December 24, 1997 (received for review November 17, 1997) ABSTRACT Plasma cell tumor induction in mice by cytoma susceptibilityyresistance locus resides. The protein pristane is under multigenic control. BALByc mice are sus- products of these genes bind to the CDKs CDK4 and CDK6 (6, ceptible to tumor development; whereas DBAy2 mice are 7), which, in turn, form complexes with the D-type G1 cyclins resistant. Restriction fragment length polymorphisms be- that phosphorylate pRb to control both cell cycle and tumor tween BALByc and DBAy2 for Cdkn2a(p16) and Cdkn2b(p15), suppression (8, 9). We compared the sequences of p16 and p18 and between BALByc and Mus spretus for Cdkn2c(p18INK4c) between susceptible (BALByc) and resistant (DBAy2N) were used to position these loci with respect to the Pctr1 locus. strains of mice, and found sequence variants in p16 located in These cyclin-dependent kinase (CDK) inhibitors mapped to a two different ankyrin repeat regions of the gene. DBAy2 (wild 6 cM interval of chromosome 4 between Ifna and Tal1. type) and BALByc A134C and G232A variants of the p16 C.D2-Chr 4 congenic strains harboring DBAy2 alleles asso- genes were fused to the glutathione S-transferase (GST) gene, ciated with the Pctr1 locus contained DBAy2 ‘‘resistant’’ and purified GST fusion proteins were tested for their ability alleles of the CDK4yCDK6 inhibitors p16 and p15. -

Inhibition of CDK4/6 As Therapeutic Approach for Ovarian Cancer Patients: Current Evidences and Future Perspectives
cancers Review Inhibition of CDK4/6 as Therapeutic Approach for Ovarian Cancer Patients: Current Evidences and Future Perspectives Alessandra Dall’Acqua 1,†, Michele Bartoletti 2,3,† , Nastaran Masoudi-Khoram 1,‡, Roberto Sorio 2, Fabio Puglisi 2,3 , Barbara Belletti 1 and Gustavo Baldassarre 1,* 1 Molecular Oncology Unit, Centro di Riferimento Oncologico di Aviano (CRO), IRCCS, National Cancer Institute, 33081 Aviano, Italy; [email protected] (A.D.); [email protected] (N.M.-K.); [email protected] (B.B.) 2 Medical Oncology and Cancer Prevention Molecular Oncology Unit, Centro di Riferimento Oncologico di Aviano (CRO), IRCCS, National Cancer Institute, 33081 Aviano, Italy; [email protected] (M.B.); [email protected] (R.S.); [email protected] (F.P.) 3 Department of Medicine (DAME), University of Udine, 33100 Udine, Italy * Correspondence: [email protected]; Tel.: +39-0434-659779; Fax: +39-0434-659429 † These authors equally contributed to the work. ‡ Present address: Department of Biophysics, Faculty of Biological Sciences, Tarbiat Modares University, Tehran, Iran. Simple Summary: Altered regulation of the cell cycle is a hallmark of cancer. The recent clinical success of the inhibitors of CDK4 and CDK6 has convincingly demonstrated that targeting cell cycle components may represent an effective anti-cancer strategy, at least in some cancer types. However, possible applications of CDK4/6 inhibitors in patients with ovarian cancer is still under evaluation. Citation: Dall’Acqua, A.; Bartoletti, M.; Masoudi-Khoram, N.; Sorio, R.; Here, we describe the possible biological role of CDK4 and CDK6 complexes in ovarian cancer and Puglisi, F.; Belletti, B.; Baldassarre, G. provide the rationale for the use of CDK4/6 inhibitors in this pathology, alone or in combination Inhibition of CDK4/6 as Therapeutic with other drugs. -

The Role of Cdks and Cdkis in Murine Development
International Journal of Molecular Sciences Review The Role of CDKs and CDKIs in Murine Development Grace Jean Campbell , Emma Langdale Hands and Mathew Van de Pette * Epigenetic Mechanisms of Toxicology Lab, MRC Toxicology Unit, Cambridge University, Cambridge CB2 1QR, UK; [email protected] (G.J.C.); [email protected] (E.L.H.) * Correspondence: [email protected] Received: 8 July 2020; Accepted: 26 July 2020; Published: 28 July 2020 Abstract: Cyclin-dependent kinases (CDKs) and their inhibitors (CDKIs) play pivotal roles in the regulation of the cell cycle. As a result of these functions, it may be extrapolated that they are essential for appropriate embryonic development. The twenty known mouse CDKs and eight CDKIs have been studied to varying degrees in the developing mouse, but only a handful of CDKs and a single CDKI have been shown to be absolutely required for murine embryonic development. What has become apparent, as more studies have shone light on these family members, is that in addition to their primary functional role in regulating the cell cycle, many of these genes are also controlling specific cell fates by directing differentiation in various tissues. Here we review the extensive mouse models that have been generated to study the functions of CDKs and CDKIs, and discuss their varying roles in murine embryonic development, with a particular focus on the brain, pancreas and fertility. Keywords: cyclin-dependent kinase; CDK inhibitors; mouse; development; knock-out models 1. Introduction Cyclin-dependent kinases (CDKs) are proteins that, by definition, require the binding of partner cyclin proteins in order to phosphorylate a series of target proteins. -

MEN4 and CDKN1B Mutations 24:10 T195–T208 Thematic Review
2410 R Alrezk et al. MEN4 and CDKN1B mutations 24:10 T195–T208 Thematic Review MEN4 and CDKN1B mutations: the latest of the MEN syndromes Rami Alrezk1, Fady Hannah-Shmouni2 and Constantine A Stratakis2 1 The National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, Correspondence Maryland, USA should be addressed 2 Section on Endocrinology & Genetics, the Eunice Kennedy Shriver National Institute of Child Health and Human to C A Stratakis Development, NIH, Bethesda, Maryland, USA Email [email protected] Abstract Multiple endocrine neoplasia (MEN) refers to a group of autosomal dominant disorders Key Words with generally high penetrance that lead to the development of a wide spectrum of f multiple endocrine endocrine and non-endocrine manifestations. The most frequent among these conditions neoplasia is MEN type 1 (MEN1), which is caused by germline heterozygous loss-of-function f MEN4 mutations in the tumor suppressor gene MEN1. MEN1 is characterized by primary f MEN1 hyperparathyroidism (PHPT) and functional or nonfunctional pancreatic neuroendocrine f neuroendocrine tumors tumors and pituitary adenomas. Approximately 10% of patients with familial or sporadic f CDKN1B MEN1-like phenotype do not have MEN1 mutations or deletions. A novel MEN syndrome f p27 was discovered, initially in rats (MENX), and later in humans (MEN4), which is caused by germline mutations in the putative tumor suppressor CDKN1B. The most common phenotype of the 19 established cases of MEN4 that have been described to date is PHPT Endocrine-Related Cancer Endocrine-Related followed by pituitary adenomas. Recently, somatic or germline mutations in CDKN1B were also identified in patients with sporadic PHPT, small intestinal neuroendocrine tumors, lymphoma and breast cancer, demonstrating a novel role for CDKN1B as a tumor susceptibility gene for other neoplasms. -

Mutations in CDKN2C (P18) and CDKN2D (P19) May Cause Sporadic Parathyroid Adenoma
T Gluick et al. CDKI in parathyroid adenoma 20:6 L27–L29 Research Letter Mutations in CDKN2C (p18) and CDKN2D (p19) may cause sporadic parathyroid adenoma Dear Editor Hyperparathyroidism (HPT) can arise from germline 2008, van Veelen et al. 2009). CDKN2D encodes p19, mutation of multiple endocrine neoplasia type 1 which is closely related to p18 but not previously (MEN1), CASR,orHRPT (HRPT1; Marx 2011). Recent considered as a tumor suppressor. studies also suggested that germline mutation of several One parathyroid tumor showed a deletion in the cyclin-dependent kinase inhibitors (CDKIs) is an uncom- p18 gene and one showed an insertion in p18; mon cause of HPT (Pellegata et al. 2006, Agarwal et al. neither change was found in the patient’s germline. 2009). A gene that predisposes to tumor via germline In the first (p18F71Ter), a 25-nucleotide deletion mutation may also predispose to similar tumor by somatic at c.1427_1454del causes a stop codon at Phe 71. In mutation; in fact, the MEN1 gene is mutated in about 30% the second (p18V118fs*6), a two-nucleotide insertion of sporadic parathyroid tumors (Marx 2011). Screening a c.1567_1568insGG causes a frameshift, followed by small number of sporadic parathyroid tumors by others, new sequence GWSSWWstop, also predicting a shortened using whole exome sequencing analysis, did not show protein. The chromatograms in Fig. 1AandBshow any CDKI gene mutation (Cromer et al. 2012, Newey et al. overlapping WT and mutant sequences, indicating hetero- 2012). In a recent study, all seven CDKI genes were zygosity or normal admixture. Western blots show stable sequenced in 81 sporadic parathyroid adenomas.