MEN4 and CDKN1B Mutations 24:10 T195–T208 Thematic Review
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Cyclin-Dependent Kinase 5 Decreases in Gastric Cancer and Its
Published OnlineFirst January 21, 2015; DOI: 10.1158/1078-0432.CCR-14-1950 Biology of Human Tumors Clinical Cancer Research Cyclin-Dependent Kinase 5 Decreases in Gastric Cancer and Its Nuclear Accumulation Suppresses Gastric Tumorigenesis Longlong Cao1,2, Jiechao Zhou2, Junrong Zhang1,2, Sijin Wu3, Xintao Yang1,2, Xin Zhao2, Huifang Li2, Ming Luo1, Qian Yu1, Guangtan Lin1, Huizhong Lin1, Jianwei Xie1, Ping Li1, Xiaoqing Hu3, Chaohui Zheng1, Guojun Bu2, Yun-wu Zhang2,4, Huaxi Xu2,4,5, Yongliang Yang3, Changming Huang1, and Jie Zhang2,4 Abstract Purpose: As a cyclin-independent atypical CDK, the role of correlated with the severity of gastric cancer based on tumor CDK5 in regulating cell proliferation in gastric cancer remains and lymph node metastasis and patient 5-year fatality rate. unknown. Nuclear localization of CDK5 was found to be significantly Experimental Design: Expression of CDK5 in gastric tumor decreased in tumor tissues and gastric cancer cell lines, and paired adjacent noncancerous tissues from 437 patients was whereas exogenously expression of nucleus-targeted CDK5 measured by Western blotting, immunohistochemistry, and real- inhibited the proliferation and xenograft implantation of time PCR. The subcellular translocation of CDK5 was monitored gastric cancer cells. Treatment with the small molecule during gastric cancer cell proliferation. The role of nuclear CDK5 NS-0011, which increases CDK5 accumulation in the nucleus, in gastric cancer tumorigenic proliferation and ex vivo xenografts suppressed both cancer cell proliferation and xenograft was explored. Furthermore, by screening for compounds in the tumorigenesis. PubChem database that disrupt CDK5 association with its nu- Conclusions: Our results suggest that low CDK5 expression is clear export facilitator, we identified a small molecular (NS-0011) associated with poor overall survival in patients with gastric that inhibits gastric cancer cell growth. -

Molecular Mechanisms of Colon Cancer Progression and Metastasis: Recent Insights and Advancements
International Journal of Molecular Sciences Review Molecular Mechanisms of Colon Cancer Progression and Metastasis: Recent Insights and Advancements Ahmed Malki 1,*, Rasha Abu ElRuz 1, Ishita Gupta 2,3,4, Asma Allouch 1 , Semir Vranic 2,3 and Ala-Eddin Al Moustafa 2,3,4,* 1 Department of Biomedical Science, College of Health Sciences, QU Health, Qatar University, Doha 2713, Qatar; [email protected] (R.A.E.); [email protected] (A.A.) 2 College of Medicine, QU Health, Qatar University, Doha 2713, Qatar; [email protected] (I.G.); [email protected] (S.V.) 3 Biomedical and Pharmaceutical Research Unit, QU Health, Qatar University, Doha 2713, Qatar 4 Biomedical Research Center, QU Health, Qatar University, Doha 2713, Qatar * Correspondence: [email protected] (A.M.); [email protected] (A.-E.A.M.); Tel.: +974-4403-6557 (A.M.); +974-3097-7440 (A.-E.A.M.) Abstract: Colorectal cancer (CRC), the third most common type of cancer, is the second leading cause of cancer-related mortality rates worldwide. Although modern research was able to shed light on the pathogenesis of CRC and provide enhanced screening strategies, the prevalence of CRC is still on the rise. Studies showed several cellular signaling pathways dysregulated in CRC, leading to the onset of malignant phenotypes. Therefore, analyzing signaling pathways involved in CRC metastasis is necessary to elucidate the underlying mechanism of CRC progression and pharmacotherapy. This review focused on target genes as well as various cellular signaling pathways including Wnt/β-catenin, p53, TGF-β/SMAD, NF-κB, Notch, VEGF, and JAKs/STAT3, which are associated with CRC progression and metastasis. -

CDK-Independent and PCNA-Dependent Functions of P21 in DNA Replication
G C A T T A C G G C A T genes Review CDK-Independent and PCNA-Dependent Functions of p21 in DNA Replication Sabrina Florencia Mansilla , María Belén De La Vega y, Nicolás Luis Calzetta y, Sebastián Omar Siri y and Vanesa Gottifredi * Cell Cycle and Genomic Stability Laboratory, Fundación Instituto Leloir, IIBBA-CONICET, Av. Patricias Argentinas 435, Buenos Aires 1405, Argentina; [email protected] (S.F.M.); [email protected] (M.B.D.L.V.); [email protected] (N.L.C.); [email protected] (S.O.S.) * Correspondence: [email protected] These authors contributed equally to this work. y Received: 9 April 2020; Accepted: 15 May 2020; Published: 28 May 2020 Abstract: p21Waf/CIP1 is a small unstructured protein that binds and inactivates cyclin-dependent kinases (CDKs). To this end, p21 levels increase following the activation of the p53 tumor suppressor. CDK inhibition by p21 triggers cell-cycle arrest in the G1 and G2 phases of the cell cycle. In the absence of exogenous insults causing replication stress, only residual p21 levels are prevalent that are insufficient to inhibit CDKs. However, research from different laboratories has demonstrated that these residual p21 levels in the S phase control DNA replication speed and origin firing to preserve genomic stability. Such an S-phase function of p21 depends fully on its ability to displace partners from chromatin-bound proliferating cell nuclear antigen (PCNA). Vice versa, PCNA also regulates p21 by preventing its upregulation in the S phase, even in the context of robust p21 induction by γ irradiation. -

The P16 (Cdkn2a/Ink4a) Tumor-Suppressor Gene in Head
The p16 (CDKN2a/INK4a) Tumor-Suppressor Gene in Head and Neck Squamous Cell Carcinoma: A Promoter Methylation and Protein Expression Study in 100 Cases Lingbao Ai, M.D., Krystal K. Stephenson, Wenhua Ling, M.D., Chunlai Zuo, M.D., Perkins Mukunyadzi, M.D., James Y. Suen, M.D., Ehab Hanna, M.D., Chun-Yang Fan, M.D., Ph.D. Departments of Pathology (LA, KKS, CZ, PM, CYF) and Otolaryngology-Head and Neck Surgery (CYF, JYS, EH), University of Arkansas for Medical Sciences; and School of Public Health (LA, WL), Sun-Yat Sen University, Guangzhou, China apparent loss of p16 protein expression appears to The p16 (CDKN2a/INK4a) gene is an important be an independent prognostic factor, although loss tumor-suppressor gene, involved in the p16/cyclin- of p16 protein may be used to predict overall pa- dependent kinase/retinoblastoma gene pathway of tient survival in early-stage head and neck squa- cell cycle control. The p16 protein is considered to mous cell carcinoma. be a negative regulator of the pathway. The gene encodes an inhibitor of cyclin-dependent kinases 4 KEY WORDS: Gene inactivation, Head and and 6, which regulate the phosphorylation of reti- neck squamous cell carcinoma, p16, Promoter noblastoma gene and the G1 to S phase transition of hypermethylation. the cell cycle. In the present study, p16 gene pro- Mod Pathol 2003;16(9):944–950 moter hypermethylation patterns and p16 protein expression were analyzed in 100 consecutive un- The development of head and neck squamous cell treated cases of primary head and neck squamous carcinoma is believed to be a multistep process, in cell carcinoma by methylation-specific PCR and im- which genetic and epigenetic events accumulate as munohistochemical staining. -

P14ARF Inhibits Human Glioblastoma–Induced Angiogenesis by Upregulating the Expression of TIMP3
P14ARF inhibits human glioblastoma–induced angiogenesis by upregulating the expression of TIMP3 Abdessamad Zerrouqi, … , Daniel J. Brat, Erwin G. Van Meir J Clin Invest. 2012;122(4):1283-1295. https://doi.org/10.1172/JCI38596. Research Article Oncology Malignant gliomas are the most common and the most lethal primary brain tumors in adults. Among malignant gliomas, 60%–80% show loss of P14ARF tumor suppressor activity due to somatic alterations of the INK4A/ARF genetic locus. The tumor suppressor activity of P14ARF is in part a result of its ability to prevent the degradation of P53 by binding to and sequestering HDM2. However, the subsequent finding of P14ARF loss in conjunction with TP53 gene loss in some tumors suggests the protein may have other P53-independent tumor suppressor functions. Here, we report what we believe to be a novel tumor suppressor function for P14ARF as an inhibitor of tumor-induced angiogenesis. We found that P14ARF mediates antiangiogenic effects by upregulating expression of tissue inhibitor of metalloproteinase–3 (TIMP3) in a P53-independent fashion. Mechanistically, this regulation occurred at the gene transcription level and was controlled by HDM2-SP1 interplay, where P14ARF relieved a dominant negative interaction of HDM2 with SP1. P14ARF-induced expression of TIMP3 inhibited endothelial cell migration and vessel formation in response to angiogenic stimuli produced by cancer cells. The discovery of this angiogenesis regulatory pathway may provide new insights into P53-independent P14ARF tumor-suppressive mechanisms that have implications for the development of novel therapies directed at tumors and other diseases characterized by vascular pathology. Find the latest version: https://jci.me/38596/pdf Research article P14ARF inhibits human glioblastoma–induced angiogenesis by upregulating the expression of TIMP3 Abdessamad Zerrouqi,1 Beata Pyrzynska,1,2 Maria Febbraio,3 Daniel J. -

Expression Profiling of KLF4
Expression Profiling of KLF4 AJCR0000006 Supplemental Data Figure S1. Snapshot of enriched gene sets identified by GSEA in Klf4-null MEFs. Figure S2. Snapshot of enriched gene sets identified by GSEA in wild type MEFs. 98 Am J Cancer Res 2011;1(1):85-97 Table S1: Functional Annotation Clustering of Genes Up-Regulated in Klf4 -Null MEFs ILLUMINA_ID Gene Symbol Gene Name (Description) P -value Fold-Change Cell Cycle 8.00E-03 ILMN_1217331 Mcm6 MINICHROMOSOME MAINTENANCE DEFICIENT 6 40.36 ILMN_2723931 E2f6 E2F TRANSCRIPTION FACTOR 6 26.8 ILMN_2724570 Mapk12 MITOGEN-ACTIVATED PROTEIN KINASE 12 22.19 ILMN_1218470 Cdk2 CYCLIN-DEPENDENT KINASE 2 9.32 ILMN_1234909 Tipin TIMELESS INTERACTING PROTEIN 5.3 ILMN_1212692 Mapk13 SAPK/ERK/KINASE 4 4.96 ILMN_2666690 Cul7 CULLIN 7 2.23 ILMN_2681776 Mapk6 MITOGEN ACTIVATED PROTEIN KINASE 4 2.11 ILMN_2652909 Ddit3 DNA-DAMAGE INDUCIBLE TRANSCRIPT 3 2.07 ILMN_2742152 Gadd45a GROWTH ARREST AND DNA-DAMAGE-INDUCIBLE 45 ALPHA 1.92 ILMN_1212787 Pttg1 PITUITARY TUMOR-TRANSFORMING 1 1.8 ILMN_1216721 Cdk5 CYCLIN-DEPENDENT KINASE 5 1.78 ILMN_1227009 Gas2l1 GROWTH ARREST-SPECIFIC 2 LIKE 1 1.74 ILMN_2663009 Rassf5 RAS ASSOCIATION (RALGDS/AF-6) DOMAIN FAMILY 5 1.64 ILMN_1220454 Anapc13 ANAPHASE PROMOTING COMPLEX SUBUNIT 13 1.61 ILMN_1216213 Incenp INNER CENTROMERE PROTEIN 1.56 ILMN_1256301 Rcc2 REGULATOR OF CHROMOSOME CONDENSATION 2 1.53 Extracellular Matrix 5.80E-06 ILMN_2735184 Col18a1 PROCOLLAGEN, TYPE XVIII, ALPHA 1 51.5 ILMN_1223997 Crtap CARTILAGE ASSOCIATED PROTEIN 32.74 ILMN_2753809 Mmp3 MATRIX METALLOPEPTIDASE -

Involvement of the Cyclin-Dependent Kinase Inhibitor P16 (Ink4a) in Replicative Senescence of Normal Human Fibroblasts
Proc. Natl. Acad. Sci. USA Vol. 93, pp. 13742–13747, November 1996 Biochemistry Involvement of the cyclin-dependent kinase inhibitor p16 (INK4a) in replicative senescence of normal human fibroblasts DAVID A. ALCORTA*†,YUE XIONG‡,DAWN PHELPS‡,GREG HANNON§,DAVID BEACH§, AND J. CARL BARRETT* *Laboratory of Molecular Carcinogenesis, National Institute of Environmental Health Sciences, Research Triangle Park, NC 27709; ‡Lineberger Comprehensive Cancer Center, University of North Carolina, Chapel Hill, NC 27599; and §Howard Hughes Medical Institute, Cold Spring Harbor Laboratories, Cold Spring Harbor, NY 11724 Communicated by Raymond L. Erickson, Harvard University, Cambridge, MA, September 19, 1996 (received for review on May 15, 1996) ABSTRACT Human diploid fibroblasts (HDFs) can be viewed in ref. 5). In senescent fibroblasts, CDK2 is catalytically grown in culture for a finite number of population doublings inactive and the protein down-regulated (7). CDK4 is also before they cease proliferation and enter a growth-arrest state reported to be down-regulated in senescent cells (8), while the termed replicative senescence. The retinoblastoma gene prod- status of CDK6 has not been previously addressed. The uct, Rb, expressed in these cells is hypophosphorylated. To activating cyclins for these CDKs, cyclins D1 and E, are present determine a possible mechanism by which senescent human in senescent cells at similar or elevated levels relative to early fibroblasts maintain a hypophosphorylated Rb, we examined passage cells (8). A role of the CDK inhibitors in senescence the expression levels and interaction of the Rb kinases, CDK4 was revealed by the isolation of a cDNA of a highly expressed and CDK6, and the cyclin-dependent kinase inhibitors p21 message in senescent cells that encoded the CDK inhibitor, p21 and p16 in senescent HDFs. -

Charles M. Perou, Phd Associate Professor Departments of Genetics
Charles M. Perou, PhD Associate Professor Departments of Genetics and Pathology Carolina Center for Genome Sciences Lineberger Comprehensive Cancer Center University of North Carolina at Chapel Hill Identification of GBM Subtypes using Gene Expression Profiling Agilent Custom Affymetrix Human Affymetrix HT-HG- 244k Exon 1.0 Array U133A Array Microarray Identify Samples and Genes represented on all 3 platforms Factor Analysis Single unified gene expression measurement for each gene 202 patients 11,681 genes Identification of GBM Subtypes Census Clustering of the 202 samples X 1740 Unsupervised clustering of 1740 genes suggests that 4 subtypes of GBM exist variably expressed genes selected using a unified gene expression measure across 3 expression platforms Core TCGA Samples (173) Gene Ontology/Pathway: with Subtype-defining genes • ProNeural: 1. nervous system development ProNeural Normal-like EGFR Mesenchymal 2. neuron differentiation (SOXs) 3. cell cycle = proliferation 4. cell adhesion molecules 5. ErbB signaling pathway FBXO3 GABRB2 • Normal‐like: SNCG NTSR2 1. nucleotide metabolic process MBP 2. neurological system process DLL3 3. axon NKX2-2, NRXN1, NRXN2 TOP2B, CDC7, MYB 4. neuron projection SOX2, SOX4, SOX10,SOX11 5. synaptic transmission ERBB3, PAK3, PAK7 NCAM1 OLIG2 • EGFR: 1. regulation of transcription 2. cell migration 3. nervous system development FGFR3 4. cell proliferation PDGFRA 5. metal ion binding EGFR AKT2 GLI2 TGFB3 • Mesenchymal: CASP8, CASP5, CASP4, CASP1 1. immune response COL8A2, COL5A1, COL1A1,COL1A2 2. receptor activity ILR4 CHI3L1 3. wound healing TRADD TLR2, TLR4, 4. cytokine and chemokine IGFBI mediated signaling pathway RELB 5. NF‐B Signaling Pathway Correlations between gene expression subtypes and clinical parameters Survival Analysis of Subtypes n=196 Subtypes are correlated with: 1. -
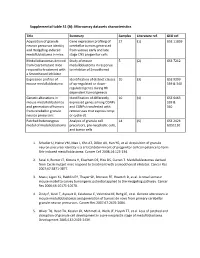
Supplemental Table S1 (A): Microarray Datasets Characteristics
Supplemental table S1 (A): Microarray datasets characteristics Title Summary Samples Literature ref. GEO ref. Acquisition of granule Gene expression profiling of 27 (1) GSE 11859 neuron precursor identity cerebellar tumors generated and Hedgehog‐induced from various early and late medulloblastoma in mice. stage CNS progenitor cells Medulloblastomas derived Study of mouse 5 (2) GSE 7212 from Cxcr6 mutant mice medulloblastoma in response respond to treatment with to inhibitor of Smoothened a Smoothened inhibitor Expression profiles of Identification of distinct classes 10 (3) GSE 9299 mouse medulloblastoma of up‐regulated or down‐ 339 & 340 regulated genes during Hh dependent tumorigenesis Genetic alterations in Identification of differently 10 (4) GSE 6463 mouse medulloblastomas expressed genes among CGNPs 339 & and generation of tumors and CGNPs transfected with 340 from cerebellar granule retroviruses that express nmyc neuron precursors or cyclin‐d1 Patched heterozygous Analysis of granule cell 14 (5) GSE 2426 model of medulloblastoma precursors, pre‐neoplastic cells, GDS1110 and tumor cells 1. Schuller U, Heine VM, Mao J, Kho AT, Dillon AK, Han YG, et al. Acquisition of granule neuron precursor identity is a critical determinant of progenitor cell competence to form Shh‐induced medulloblastoma. Cancer Cell 2008;14:123‐134. 2. Sasai K, Romer JT, Kimura H, Eberhart DE, Rice DS, Curran T. Medulloblastomas derived from Cxcr6 mutant mice respond to treatment with a smoothened inhibitor. Cancer Res 2007;67:3871‐3877. 3. Mao J, Ligon KL, Rakhlin EY, Thayer SP, Bronson RT, Rowitch D, et al. A novel somatic mouse model to survey tumorigenic potential applied to the Hedgehog pathway. Cancer Res 2006;66:10171‐10178. -

Transcriptional Regulation of the P16 Tumor Suppressor Gene
ANTICANCER RESEARCH 35: 4397-4402 (2015) Review Transcriptional Regulation of the p16 Tumor Suppressor Gene YOJIRO KOTAKE, MADOKA NAEMURA, CHIHIRO MURASAKI, YASUTOSHI INOUE and HARUNA OKAMOTO Department of Biological and Environmental Chemistry, Faculty of Humanity-Oriented Science and Engineering, Kinki University, Fukuoka, Japan Abstract. The p16 tumor suppressor gene encodes a specifically bind to and inhibit the activity of cyclin-CDK specific inhibitor of cyclin-dependent kinase (CDK) 4 and 6 complexes, thus preventing G1-to-S progression (4, 5). and is found altered in a wide range of human cancers. p16 Among these CKIs, p16 plays a pivotal role in the regulation plays a pivotal role in tumor suppressor networks through of cellular senescence through inhibition of CDK4/6 activity inducing cellular senescence that acts as a barrier to (6, 7). Cellular senescence acts as a barrier to oncogenic cellular transformation by oncogenic signals. p16 protein is transformation induced by oncogenic signals, such as relatively stable and its expression is primary regulated by activating RAS mutations, and is achieved by accumulation transcriptional control. Polycomb group (PcG) proteins of p16 (Figure 1) (8-10). The loss of p16 function is, associate with the p16 locus in a long non-coding RNA, therefore, thought to lead to carcinogenesis. Indeed, many ANRIL-dependent manner, leading to repression of p16 studies have shown that the p16 gene is frequently mutated transcription. YB1, a transcription factor, also represses the or silenced in various human cancers (11-14). p16 transcription through direct association with its Although many studies have led to a deeper understanding promoter region. -

The P16 Status of Tumor Cell Lines Identifies Small Molecule Inhibitors Specific for Cyclin-Dependent Kinase 41
Vol. 5, 4279–4286, December 1999 Clinical Cancer Research 4279 The p16 Status of Tumor Cell Lines Identifies Small Molecule Inhibitors Specific for Cyclin-dependent Kinase 41 Akihito Kubo,2 Kazuhiko Nakagawa,2, 3 CDK4 kinase inhibitors that may selectively induce growth Ravi K. Varma, Nicholas K. Conrad, inhibition of p16-altered tumors. Jin Quan Cheng, Wen-Ching Lee, INTRODUCTION Joseph R. Testa, Bruce E. Johnson, INK4A 4 The p16 gene (also known as CDKN2A) encodes p16 , Frederic J. Kaye, and Michael J. Kelley which inhibits the CDK45:cyclin D and CDK6:cyclin D com- Medicine Branch [A. K., K. N., N. K. C., F. J. K., B. E. J.] and plexes (1). These complexes mediate phosphorylation of the Rb Developmental Therapeutics Program [R. K. V.], National Cancer Institute, Bethesda, Maryland 20889; Department of Medical protein and allow cell cycle progression beyond the G1-S-phase Oncology, Fox Chase Cancer Center, Philadelphia, Pennsylvania checkpoint (2). Alterations of p16 have been described in a wide 19111 [J. Q. C., W-C. L., J. R. T.]; and Department of Medicine, variety of histological types of human cancers including astro- Duke University Medical Center, Durham, North Carolina 27710 cytoma, melanoma, leukemia, breast cancer, head and neck [M. J. K.] squamous cell carcinoma, malignant mesothelioma, and lung cancer. Alterations of p16 can occur through homozygous de- ABSTRACT letion, point mutation, and transcriptional suppression associ- ated with hypermethylation in cancer cell lines and primary Loss of p16 functional activity leading to disruption of tumors (reviewed in Refs. 3–5). the p16/cyclin-dependent kinase (CDK) 4:cyclin D/retino- Whereas the Rb gene is inactivated in a narrow range of blastoma pathway is the most common event in human tumor cells, the pattern of mutational inactivation of Rb is tumorigenesis, suggesting that compounds with CDK4 ki- inversely correlated with p16 alterations (6–8), suggesting that nase inhibitory activity may be useful to regulate cancer cell a single defect in the p16/CDK4:cyclin D/Rb pathway is suffi- growth. -

Cyclin E-CDK2 Is a Regulator of P27 Kip1
Downloaded from genesdev.cshlp.org on October 4, 2021 - Published by Cold Spring Harbor Laboratory Press Cyclin E-CDK2 is a regulator of p27 Kip1 Robert J. Sheaff, 1 Mark Groudine, 1'4 Matthew Gordon, 1 James M. Roberts, 1's and Bruce E. Clurman 1-3,5 Divisions of 1Basic Sciences and 2Clinical Research, Fred Hutchinson Cancer Research Center (FHCRC), Seattle, Washington 98104; Departments of 3Medicine and 4Radiation Oncology, University of Washington, Seattle, Washington 98104 CDK inhibitors are thought to prevent cell proliferation by negatively regulating cyclin-CDK complexes. We propose that the opposite is also true, that cyclin-CDK complexes in mammmalian cells can promote cell cycle progression by directly down-regulating CDK inhibitors. We show that expression of cyclin E-CDK2 in murine fibroblasts causes phosphorylation of the CDK inhibitor p27 Kip1 on T187, and that cyclin E-CDK2 can directly phosphorylate p27 T187 in vitro. We further show that cyclin E-CDK2-dependent phosphorylation of p27 results in elimination of p27 from the cell, allowing cells to transit from G1 to S phase. Moreover, mutation of T187 in p27 to alanine creates a p27 protein that causes a G1 block resistant to cyclin E and whose level of expression is not modulated by cyclin E. A kinetic analysis of the interaction between p27 and cyclin E-CDK2 explains how p27 can be regulated by the same enzyme it targets for inhibition. We show that p27 interacts with cyclin E-CDK2 in at least two distinct ways: one resulting in p27 phosphorylation and release, the other in tight binding and cyclin E-CDK2 inhibition.