Phenylpropanoids
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Scopoletin 8-Hydroxylase: a Novel Enzyme Involved in Coumarin
bioRxiv preprint doi: https://doi.org/10.1101/197806; this version posted October 4, 2017. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. 1 Scopoletin 8-hydroxylase: a novel enzyme involved in coumarin biosynthesis and iron- 2 deficiency responses in Arabidopsis 3 4 Running title: At3g12900 encodes a scopoletin 8-hydroxylase 5 6 Joanna Siwinska1, Kinga Wcisla1, Alexandre Olry2,3, Jeremy Grosjean2,3, Alain Hehn2,3, 7 Frederic Bourgaud2,3, Andrew A. Meharg4, Manus Carey4, Ewa Lojkowska1, Anna 8 Ihnatowicz1,* 9 10 1Intercollegiate Faculty of Biotechnology of University of Gdansk and Medical University of 11 Gdansk, Abrahama 58, 80-307 Gdansk, Poland 12 2Université de Lorraine, UMR 1121 Laboratoire Agronomie et Environnement Nancy- 13 Colmar, 2 avenue de la forêt de Haye 54518 Vandœuvre-lès-Nancy, France; 3INRA, UMR 14 1121 Laboratoire Agronomie et Environnement Nancy-Colmar, 2 avenue de la forêt de Haye 15 54518 Vandœuvre-lès-Nancy, France; 16 4Institute for Global Food Security, Queen’s University Belfast, David Keir Building, Malone 17 Road, Belfast, UK; 18 19 [email protected] 20 [email protected] 21 [email protected] 22 [email protected] 23 [email protected] 24 [email protected] 25 [email protected] 26 [email protected] 27 [email protected] 28 *Correspondence: [email protected], +48 58 523 63 30 29 30 The date of submission: 02.10.2017 31 The number of figures: 9 (Fig. -

Biosynthesis of Daphnetin in Daphne Mezereum L.*
Biosynthesis of Daphnetin in Daphne mezereum L.* Stewart A. Brown Department of Chemistry, Trent University, Peterborough, Ont., Canada K9J 7B8 Z. Naturforsch. 41c, 247—252 (1986); received November 4, 1985 Biosynthesis, Coumarins, Daphne mezereum, Daphnetin Shoots of Daphne mezereum synthesized daphnetin (7,8-dihydroxycoumarin) more efficiently from [2-l4C]umbelliferone (7-hydroxycoumarin) than from [2-l4C]p-coumaric acid, and [2-14C]caf- feic acid was more poorly utilized still. These findings do not support the idea of derivation of daphnetin via hydroxylation of the caffeic acid ring at the 2 position, followed by lactone ring formation; instead they are consistent with the concept of daphnetin formation by an additional hydroxylation of umbelliferone at C-8. Umbelliferone was recovered with little l4C dilution from emulsin-hydrolysed extracts of shoots fed labelled umbelliferone, and TLC of extracts from un treated shoots revealed two substances yielding umbelliferone on hydrolysis. Evidence is pre sented from TLC and HPLC analysis that one of these is skimmin (7-O-ß-D-glucosylumbel- liferone), not previously reported from Daphne. The tracer experiments further support the theory that umbelliferone is the general precursor of coumarins bearing two or more hydroxyl functions on the aromatic ring. Introduction (Thymelaeaceae), a hardy perennial shrub, where it Approximately 70 coumarins with the 7,8 oxygen occurs together with its 7-ß-D-glucoside, daphnin ation pattern' have been reported to be naturally (6b) and daphnetin glucoside (6c), the 8-O-ß-D- occurring [1], over half being furanocoumarins de g lu c o s y la te d is o m e r [1]. -

A Detailed Review on Morphotaxonomy And
Acta Scientific Microbiology (ISSN: 2581-3226) Review Article Volume 1 Issue 6 June 2018 Skimmia anquetilia N.P. Taylor and Airy Shaw A Detailed Review on Morphotaxonomy and Chemoprofiling of Saduf Nissar1, Neelofar Majid1, Aabid M Rather1*, Irshad A Nawchoo1 and GG Mohi-Ud-Din2 1Plant Reproductive Biology, Genetic Diversity and Phytochemistry Research Laboratory, Department of Botany, University of Kashmir, Srinagar, India 2Department of Botany, Government Degree College for Women, Sopore, Baramullah, India *Corresponding Author: Aabid M Rather, Plant Reproductive Biology, Genetic Diversity and Phytochemistry Research Laboratory, DepartmentReceived: April of Botany, 20, 2018; University Published: of Kashmir, May 28, Srinagar, 2018 India. Abstract - In recent times, medicinal plants have attracted huge attention due to their diverse range of biological and therapeutic proper Skimmia ties. Evidences have been accumulated since ages to demonstrate promising potential of medicinal plants used in various traditional, anquetilia complementary, and alternative systems with the ever-increasing interest of today’s population towards natural products, Rutaceae N.P. Taylor and Airy Shaw emerged out to be one of the most eye-catching plant bearing multiple medicinal properties. It is a perennial aromatic evergreen shrub belonging to family . Pharmacological studies have demonstrated significant action - of different extracts as antimicrobial, anti-inflammatory agents, among others, supporting some of its popular uses. An attempt has S. anquetilia been made in this review article to provide an up-to-date overview of the morphological parameters, taxonomic features, distribu tion pattern, traditional uses, as well as the phytochemistry and biological activities of . The present review provides insights for future research aiming for both ethnopharmacological validation of its popular use and its exploration as a new source ofKeywords herbal drugs: Skimmia and/or anquetilia; bioactive Rutaceaenatural products. -
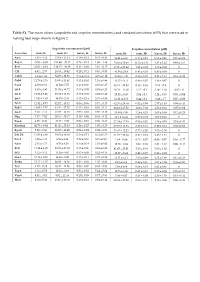
(Scopoletin and Scopolin Concentrations) and Standard Deviations (±SD) That Were Used in Making Heat Maps Shown in Figure 2
Table S1. The mean values (scopoletin and scopolin concentrations) and standard deviations (±SD) that were used in making heat maps shown in Figure 2. Scopoletin concentration [µM] Scopolin concentration [µM] Accession roots, H- roots, H+ leaves, H- leaves, H+ roots, H- roots, H+ leaves, H- leaves, H+ An-1 4.91 ± 4.12 15.19 ± 11.34 0.10 ± 0.12 0.45 ± 0.24 14.81 ± 4.05 0.13 ± 0.12 0.68 ± 0.41 0.05 ± 0.08 Bay-0 41.93 ± 6.58 151.90 ± 30.12 0.29 ± 0.18 1.99 ± 1.46 38.95 ± 10.69 07.15 ± 6.36 3.81 ± 1.22 0.88 ± 1.53 Br-0 35.01 ± 9.48 139.27 ± 49.38 0.31 ± 0.09 2.61 ± 0.27 67.26 ± 23.82 1.22 ± 0.08 1.65 ± 0.08 0 C24 4.92 ± 2.67 16.63 ± 10.42 0.20 ± 0.11 0.83 ± 0.55 41.36 ± 24.8 0.43 ± 0.19 0.85 ± 0.59 0 Can-0 3.14 ± 1.89 64.03 ± 84.91 0.14 ± 0.14 0.37 ± 0.12 34.63 ± 4.85 0.56 ± 0.05 2.23 ± 1.24 0.19 ± 0.33 Col-0 11.79 ± 3.16 29.60 ± 11.41 0.31 ± 0.41 1.16 ± 0.99 11.47 ± 3.41 0.46 ± 0.07 1.48 ± 0.67 0 Cvi-1 4.58 ± 0.62 11.54 ± 7.57 0.19 ± 0.10 0.61 ± 0.27 35.41 ± 32.83 0.35 ± 0.16 2.15 ± 0.8 0 Eil-0 8.03 ± 4.90 21.55 ± 14.72 0.15 ± 0.03 0.53 ± 0.23 28.24 ± 11.10 1.12 ± 0.2 2.19 ± 1.16 0.02 ± 0 Eri-0 12.86 ± 3.96 23.84 ± 13.33 0.13 ± 0.09 0.68 ± 0.48 20.55 ± 8.45 2.04 ± 0.3 2.24 ± 0.93 0.03 ± 0.04 Est-1 14.85 ± 1.63 36.79 ± 2.44 0.42 ± 0.19 2.01 ± 0.86 32.22 ±12.66 0.44 ± 0.2 6.48 ± 3.7 0.07 ± 0.09 Fei-0 12.82 ± 8.73 52.02 ± 15.32 0.09 ± 0.06 0.73 ± 0.37 62.36 ± 28.64 0.52 ± 0.08 2.77 ± 1.53 0.08 ± 0.13 Fuk-1 13.06 ± 7.87 44.36 ± 47.37 0.13 ± 0.01 0.62 ± 0.11 29.23 ± 27.52 8.09 ± 7.02 1.58 ± 0.62 0.05 ± 0.09 Ga-0 3.40 ± 1.11 19.67 ± 12.65 0.03 -

Scopoletin 8-Hydroxylase a Novel Enzyme Involved in Coumarin Biosynthesis and Iron-Deficiency Responses in Arabidopsis
Scopoletin 8-hydroxylase a novel enzyme involved in coumarin biosynthesis and iron-deficiency responses in Arabidopsis Siwinska, J., Siatkowska, K., Olry, A., Grosjean, J., Hehn, A., Bourgaud, F., Meharg, A. A., Carey, M., Lojkowska, E., & Ihnatowicz, A. (2018). Scopoletin 8-hydroxylase a novel enzyme involved in coumarin biosynthesis and iron-deficiency responses in Arabidopsis. Journal of experimental botany, 69(7), 1735-1748. https://doi.org/10.1093/jxb/ery005 Published in: Journal of experimental botany Document Version: Publisher's PDF, also known as Version of record Queen's University Belfast - Research Portal: Link to publication record in Queen's University Belfast Research Portal Publisher rights Copyright the authors 2018. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted reuse, distribution, and reproduction in any medium, provided the original work is properly cited. General rights Copyright for the publications made accessible via the Queen's University Belfast Research Portal is retained by the author(s) and / or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Take down policy The Research Portal is Queen's institutional repository that provides access to Queen's research output. Every effort has been made to ensure that content in the Research Portal does not infringe any person's rights, or applicable UK laws. If you discover content in the Research Portal that you believe breaches copyright or violates any law, please contact [email protected]. -

WO 2018/002916 Al O
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2018/002916 Al 04 January 2018 (04.01.2018) W !P O PCT (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every C08F2/32 (2006.01) C08J 9/00 (2006.01) kind of national protection available): AE, AG, AL, AM, C08G 18/08 (2006.01) AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DJ, DK, DM, DO, (21) International Application Number: DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, HN, PCT/IL20 17/050706 HR, HU, ID, IL, IN, IR, IS, JO, JP, KE, KG, KH, KN, KP, (22) International Filing Date: KR, KW, KZ, LA, LC, LK, LR, LS, LU, LY, MA, MD, ME, 26 June 2017 (26.06.2017) MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, (25) Filing Language: English SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, (26) Publication Language: English TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: (84) Designated States (unless otherwise indicated, for every 246468 26 June 2016 (26.06.2016) IL kind of regional protection available): ARIPO (BW, GH, GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, ST, SZ, TZ, (71) Applicant: TECHNION RESEARCH & DEVEL¬ UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, RU, TJ, OPMENT FOUNDATION LIMITED [IL/IL]; Senate TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, DK, House, Technion City, 3200004 Haifa (IL). -

Identification of Qtls Affecting Scopolin and Scopoletin Biosynthesis in Arabidopsis Thaliana
Siwinska et al. BMC Plant Biology 2014, 14:280 http://www.biomedcentral.com/1471-2229/14/280 RESEARCH ARTICLE Open Access Identification of QTLs affecting scopolin and scopoletin biosynthesis in Arabidopsis thaliana Joanna Siwinska1, Leszek Kadzinski1, Rafal Banasiuk1, Anna Gwizdek-Wisniewska1, Alexandre Olry2,3, Bogdan Banecki1, Ewa Lojkowska1 and Anna Ihnatowicz1* Abstract Background: Scopoletin and its glucoside scopolin are important secondary metabolites synthesized in plants as a defense mechanism against various environmental stresses. They belong to coumarins, a class of phytochemicals with significant biological activities that is widely used in medical application and cosmetics industry. Although numerous studies showed that a variety of coumarins occurs naturally in several plant species, the details of coumarins biosynthesis and its regulation is not well understood. It was shown previously that coumarins (predominantly scopolin and scopoletin) occur in Arabidopsis thaliana (Arabidopsis) roots, but until now nothing is known about natural variation of their accumulation in this model plant. Therefore, the genetic architecture of coumarins biosynthesis in Arabidopsis has not been studied before. Results: Here, the variation in scopolin and scopoletin content was assessed by comparing seven Arabidopsis accessions. Subsequently, a quantitative trait locus (QTL) mapping was performed with an Advanced Intercross Recombinant Inbred Lines (AI-RILs) mapping population EstC (Est-1 × Col). In order to reveal the genetic basis of both scopolin and scopoletin biosynthesis, two sets of methanol extracts were made from Arabidopsis roots and one set was additionally subjected to enzymatic hydrolysis prior to quantification done by high-performance liquid chromatography (HPLC). We identified one QTL for scopolin and five QTLs for scopoletin accumulation. -

Identification of Qtls Affecting Scopolin and Scopoletin
Identification of QTLs affecting scopolin and scopoletin biosynthesis in Arabidopsis thaliana Joanna Siwinska, Leszek Kadzinski, Rafal Banasiuk, Anna Gwizdek-Wisniewska, Alexandre Olry, Bogdan Banecki, Ewa Lojkowska, Anna Ihnatowicz To cite this version: Joanna Siwinska, Leszek Kadzinski, Rafal Banasiuk, Anna Gwizdek-Wisniewska, Alexandre Olry, et al.. Identification of QTLs affecting scopolin and scopoletin biosynthesis in Arabidopsis thaliana. BMC Plant Biology, BioMed Central, 2014, 14, 10.1186/s12870-014-0280-9. hal-01477746 HAL Id: hal-01477746 https://hal.univ-lorraine.fr/hal-01477746 Submitted on 10 May 2017 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Siwinska et al. BMC Plant Biology 2014, 14:280 http://www.biomedcentral.com/1471-2229/14/280 RESEARCH ARTICLE Open Access Identification of QTLs affecting scopolin and scopoletin biosynthesis in Arabidopsis thaliana Joanna Siwinska1, Leszek Kadzinski1, Rafal Banasiuk1, Anna Gwizdek-Wisniewska1, Alexandre Olry2,3, Bogdan Banecki1, Ewa Lojkowska1 and Anna Ihnatowicz1* Abstract Background: Scopoletin and its glucoside scopolin are important secondary metabolites synthesized in plants as a defense mechanism against various environmental stresses. They belong to coumarins, a class of phytochemicals with significant biological activities that is widely used in medical application and cosmetics industry. -

Khellactone Derivatives and Other Phenolics of Phlojodicarpus Sibiricus (Apiaceae): HPLC-DAD-ESI-QQQ-MS/MS and HPLC-UV Profile
Article Khellactone Derivatives and Other Phenolics of Phlojodicarpus sibiricus (Apiaceae): HPLC‐DAD‐ESI‐QQQ‐MS/MS and HPLC‐UV Profile, and Antiobesity Potential of Dihydrosamidin Daniil N. Olennikov 1,*, Innokentii A. Fedorov 2, Nina I. Kashchenko 1, Nadezhda K. Chirikova 3 and Cecile Vennos 4 1 Institute of General and Experimental Biology, Siberian Division, Russian Academy of Science, 6 Sakh’yanovoy Street, Ulan‐Ude 670047, Russia; [email protected] 2 Institute for Biological Problems of Cryolithozone, Siberian Division, Russian Academy of Science, 41 Lenina Street, Yakutsk 677000, Russia; [email protected] 3 Department of Biochemistry and Biotechnology, North‐Eastern Federal University, 58 Belinsky Street, Yakutsk 677027, Russia; [email protected] 4 Regulatory and Medical Scientific Affairs, Padma AG, 1 Underfeldstrasse, Hinwil CH‐8340, Switzerland; [email protected] * Correspondence: [email protected]; Tel.: +7‐902‐160‐0627 Academic Editor: Alessandra Guerrini Received: 27 May 2019; Accepted: 18 June 2019; Published: 19 June 2019 Abstract: With obesity, the consumption of phenolic‐enriched food additives as a part of traditional nutrition avoids the negative implications of eating high‐calorie products. This study investigated the new herbal food additive, Phlojodicarpus sibiricus roots and herb, ubiquitously used in Siberia as a spice. Chromatographic techniques such as HPLC‐DAD‐ESI‐QQQ‐MS/MS and microcolumn HPLC‐UV were the basic instruments for component profiling and quantification, and antiobesity potential was investigated using a differentiated 3T3‐L1 adipocytes assay. We found that the roots and herb of P. sibiricus were high‐coumarin‐containing additives inhibiting triacylglycerol accumulation in 3T3‐L1 preadipocytes. Forty‐one phenolics were detected in P. -

Planta Medica
www.thieme.de/fz/plantamedica | www.thieme-connect.de/ejournals Planta Medica July 2009 · Page 877 – 1094 · Volume 75 9 · 2009 Editorial Poster 877 Editorial 903 Topic A: Lead finding from Nature 928 Topic B: Conservation and biodiversity issues 878 Lectures 939 Topic C: Plants and aging of the population 944 Topic D: Natural products and neglected diseases Workshops 882 WS1 Workshops for Young Researchers: 966 Topic E: Anti-cancer agents Validation of Analytical Methods 988 Topic F: HIV and viral diseases 882 WS2 Workshops for Young Researchers: Cell Culture 991 Topic G: Quality control and safety assessments of phytomedicines 882 WS3 Permanent Committees on Manufacturing and Quality Control of Herbal Remedies and 1007 Topic H: Prevention of metabolic diseases Regulatory Affairs of Herbal Medicinal Products by medicinal plants and nutraceuticals 883 WS4 Permanent Committee on Biological and 1019 Topic I: Cosmetics, flavours and aromas Pharmacological Activity of Natural Products: Phytoestrogens: risks and benefits for human 1029 Topic J: Free Topic health 883 WS5 Permanent Committee on Breeding and 1083 Authors’ Index Cultivation of Medicinal Plants: Genetic Resources, Conservation and Breeding 1094 Masthead 884 Short lectures Editorial 877 57th International Congress and Annual Meeting of the Society for Medicinal Plant and Natural Product Research Date/Place: Geneva, Switzerland, August 16 – 20, 2009 Chairman: Kurt Hostettmann Dear Colleagues, The 57th Congress of the Society of Medicinal Plant and Natural Product research will be held this year in Geneva, Switzerland. The congress venue is going to be at the CICG (Centre International des Confrences Genve) which is very well equipped to host such an important scientific event. -

A Mini Review on the Protective Effect of Lignans for the Treatment of Neurodegenerative Disorders
Das M and Devi KP. J Nutr Food Lipid Sci 2019(1): 40-53. https://doi.org/10.33513/NFLS/1901-06 OCIMUM Journal of Nutrition, Food and Lipid Science Review Article A Mini Review on the Protective Effect of Lignans for the Treatment of Neurodegenerative Disorders Abstract Nature is a rich source of numerous bioactive compounds that are categorized as secondary metabolites. Lignans are group of such compounds, generally called phytoestrogens widely present in many plants and vegetables, grains, seeds, nuts and tea. They have been used as folk medicine for the treatment of several clinical conditions like asthma, cardiovascular diseases, arthrosclerosis, colitis Mamali Das and Kasi Pandima Devi* and many more. Structurally, lignans are characterized by two phenylpropane Department of Biotechnology, Alagappa University, groups attached by a carbon bond. They have been divided in to several types Tamil Nadu, India on the basis of structure of their carbon skeleton, the way of cyclization and oxygen incorporation in the skeleton. Lignans from numerous plant species Received Date: 22 December 2018 such as Kadsura polysperma, Kadsura ananosma, Schisandra wilsoniana, Accepted Date: 28 January 2019 Schisandra chinensis, Schisandra arisanensis, Manglietiastrum sinicum, Published Date: 19 February 2019 Pycnanthus angolensis, Cleistanthus indochinensis, Sargentodoxa cuneata, Tabebuia chrysotricha, Lindera glauca, Tilia amurensis and many more have Citation been found to be beneficial for cancer, hepatitis, microbial and fungal infection. Neurodegenerative Diseases (NDD) represent a class of disorder each of which Das M, Devi KP (2019) A Mini Review on the corresponds to a specific pathological condition while their molecular pathways Protective Effect of Lignans for the Treatment of have been found to be interlinked. -

Two Glucosides of Coumarin Derivatives in Sweet Potato Roots Infected by Ceratocystis Fimbriata by Takao MINAMIKAWA, TAKASHI
[Agr. Biol. Chem., Vol. 28, No. 4, p. 230•`233, 1964] Two Glucosides of Coumarin Derivatives in Sweet Potato Roots Infected by Ceratocystis Fimbriata By Takao MINAMIKAWA,TAKASHI AKAZAWA and IKUZO URITANI Laboratoryof BiochemistryNagoya University, Anjo, Aichi ReceivedJanuary 13, 1964 Two glucosides of coumarin derivatives were separated from sweet potato roots with black rot, by the combination of silica gel-coated thin layer chromatography and paper chromatography, and identified with skimmin and scopolin. The magnitude of synthesis of both bound coumarins was lower than that of the corresponding free coumarins, whereas they were detectable in neither cut nor fresh root tissues. sues7). During the course of this work, we INTRODUCTION also tried to find out whether or not the Several workers have reported the produc fungus infection would induce the produc tion of coumarins in plant tissues infected by tion of the bound forms of the respective pathogenic microorganisms1•`4) Post-infec coumarins in the root tissues. Hughes and tion synthesis of umbelliferone (7-hydroxy- Swain4) reported that about 25-fold increase coumarin) and scopoletin (6-methoxy-7-hy of scopolin (scopoletin-7-ƒÀ-glucoside) was in droxycoumarin) in sweet potato roots attack duced by the infection of Phytophthora in ed by the black rot fungus, Ceratocystis festans in potato tubers, whereas the chloro fimbriata, was reported by Uritani and Hoshi genic acid increase was only 2- to 5-fold in - ya5), and recently the third coumarin com the infected region. pound, esculetin (6,7-dihydroxycoumarin), This paper describes the chromatographic has been added to the list6). We have also identification of skimmin (umbelliferone-7-ƒÀ- carried out an analytical study on the synthe sis of the former two coumarins in the in glucoside) and scopolin in the sweet potato tissue with black rot.