Peptides & Proteins
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Heterogeneous Impacts of Protein-Stabilizing Osmolytes On
bioRxiv preprint doi: https://doi.org/10.1101/328922; this version posted May 23, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Heterogeneous Impacts of Protein-Stabilizing Osmolytes on Hydrophobic Interaction Mrinmoy Mukherjee and Jagannath Mondal⇤ Tata Institute of Fundamental Research Hyderabad, 500107 India E-mail: [email protected],+914020203091 Abstract Osmolytes’ mechanism of protecting proteins against denaturation is a longstanding puzzle, further complicated by the complex diversities inherent in protein sequences. An emergent approach in understanding osmolytes’ mechanism of action towards biopoly- mer has been to investigate osmolytes’ interplay with hydrophobic interaction, the ma- jor driving force of protein folding. However, the crucial question is whether all these protein-stabilizing osmolytes display a single unified mechanism towards hydrophobic interactions. By simulating the hydrophobic collapse of a macromolecule in aqueous solutions of two such osmoprotectants, Glycine and Trimethyl N-oxide (TMAO), both of which are known to stabilize protein’s folded conformation, we here demonstrate that these two osmolytes can impart mutually contrasting effects towards hydropho- bic interaction. While TMAO preserves its protectant nature across diverse range of polymer-osmolyte interactions, glycine is found to display an interesting cross-over from being a protectant at weaker polymer-osmolyte interaction to a denaturant of hydrophobicity at stronger polymer-osmolyte interactions. A preferential-interaction analysis reveals that a subtle balance of conformation-dependent exclusion/binding of ⇤To whom correspondence should be addressed 1 bioRxiv preprint doi: https://doi.org/10.1101/328922; this version posted May 23, 2018. -

Protein Structure & Folding
6 Protein Structure & Folding To understand protein folding In the last chapter we learned that proteins are composed of amino acids Goal as a chemical equilibrium. linked together by peptide bonds. We also learned that the twenty amino acids display a wide range of chemical properties. In this chapter we will see Objectives that how a protein folds is determined by its amino acid sequence and that the three-dimensional shape of a folded protein determines its function by After this chapter, you should be able to: the way it positions these amino acids. Finally, we will see that proteins fold • describe the four levels of protein because doing so minimizes Gibbs free energy and that this minimization structure and the thermodynamic involves both making the most favorable bonds and maximizing disorder. forces that stabilize them. • explain how entropy (S) and enthalpy Proteins exhibit four levels of structure (H) contribute to Gibbs free energy. • use the equation ΔG = ΔH – TΔS The structure of proteins can be broken down into four levels of to determine the dependence of organization. The first is primary structure, the linear sequence of amino the favorability of a reaction on acids in the polypeptide chain. By convention, the primary sequence is temperature. written in order from the amino acid at the N-terminus (by convention • explain the hydrophobic effect and its usually on the left) to the amino acid at the C-terminus. The second level role in protein folding. of protein structure, secondary structure, is the local conformation adopted by stretches of contiguous amino acids. -
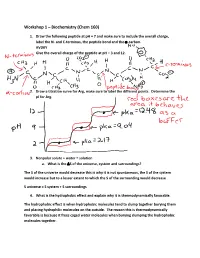
Workshop 1 – Biochemistry (Chem 160)
Workshop 1 – Biochemistry (Chem 160) 1. Draw the following peptide at pH = 7 and make sure to include the overall charge, label the N- and C-terminus, the peptide bond and the -carbon. AVDKY Give the overall charge of the peptide at pH = 3 and 12. 2. Draw a titration curve for Arg, make sure to label the different points. Determine the pI for Arg. 3. Nonpolar solute + water = solution a. What is the S of the universe, system and surroundings? The S of the universe would decrease this is why it is not spontaneous, the S of the system would increase but to a lesser extent to which the S of the surrounding would decrease S universe = S system + S surroundings 4. What is the hydrophobic effect and explain why it is thermodynamically favorable. The hydrophobic effect is when hydrophobic molecules tend to clump together burying them and placing hydrophilic molecules on the outside. The reason this is thermodynamically favorable is because it frees caged water molecules when burying clumping the hydrophobic molecules together. 5. Urea dissolves very readily in water, but the solution becomes very cold as the urea dissolves. How is this possible? Urea dissolves in water because when dissolving there is a net increase in entropy of the universe. The heat exchange, getting colder only reflects the enthalpy (H) component of the total energy change. The entropy change is high enough to offset the enthalpy component and to add up to an overall -G 6. A mutation that changes an alanine residue in the interior of a protein to valine is found to lead to a loss of activity. -

Amino Acid Building Block Models – in Brief
Amino Acid Building Block Models – In Brief Key Teaching Points for Amino Acid Building Block Models© Overall Student Learning Objective: What Dictates How a Protein Folds? Amino acids are the building blocks of proteins. All amino acids have an identical core structure consisting of an alpha-carbon, carboxyl group, amino group and R-group (sidechain). A linear chain of amino acids is a polypeptide. The primary sequence of a protein is the linear sequence of amino acids in a polypeptide. Proteins are made up of amino acid monomers linked together by peptide bonds. Peptide bond formation between amino acids results in the release of water (dehydration synthesis or condensation reaction). The protein backbone is characterized by the “N-C-C-N-C-C. .” pattern. The “ends” of the protein can be identified by the N-terminus (amino group) end and the C-terminus (carboxyl group) end. For a more complete lesson guide, please visit: http://www.3dmoleculardesigns.com/3DMD-Files/AABB/ContentsandAssembly.pdf Amino Acid Core Structure Build an amino acid according to the diagram to the right: 1. Identify the alpha carbon, amino group, carboxyl group and R-group (sidechain representation) in the structure you have constructed. Two amino acids can be chemically linked by a reaction called “condensation” or “dehydration synthesis” to form a dipeptide bond linking the two amino acids. A chain of amino acid units (monomers) linked together by peptide bonds is called a polypeptide. General Dipeptide Structure Construct a model of a dipeptide using the amino acid models previously built. 2. What are the products of the condensation reaction (dehydration synthesis)? 3. -

Hydrophobic Effect, Water Structure, and Heat Capacity Changes
J. Phys. Chem. B 1997, 101, 4343-4348 4343 Hydrophobic Effect, Water Structure, and Heat Capacity Changes Kim A. Sharp* and Bhupinder Madan Department of Biochemistry & Biophysics, UniVersity of PennsylVania, 3700 Hamilton Walk, Philadelphia, PennsylVania 19104-6059 ReceiVed: January 16, 1997; In Final Form: April 1, 1997X hyd The hydration heat capacity (∆Cp ) of nine solutes of varying hydrophobicity was studied using a combination of a random network model equation of water and Monte Carlo simulations. Nonpolar solutes cause a concerted decrease in the average length and angle of the water-water hydrogen bonds in the first hydration shell, while polar and ionic solutes have the opposite effect. This is due to changes in the amounts, relative to bulk water, of two populations of hydrogen bonds: one with shorter and more linear bonds and the other with longer and more bent bonds. Heat capacity changes were calculated from these changes in water structure using a random network model equation of state. The calculated changes account for observed hyd differences in ∆Cp for the various solutes. The simulations provide a unified picture of hydrophobic and hyd polar hydration, and both a structural and thermodynamic explanation of the opposite signs of ∆Cp observed for polar and nonpolar solutes. Introduction nonpolar groups means that at higher temperatures (T > 80 °C) their insolubility results from unfavorable changes in enthalpy, In recent years analysis of heat capacity changes has become not entropy. Second, the hydration of a majority of polar and of central importance in understanding the thermodynamics of ionic solutes is also accompanied by a decrease in entropy, and protein folding, protein-protein binding, protein-nucleic acid thus by some kind of structuring of water, but in this case binding, and the hydrophobic effect.1-7 There are several hyd 7,16-20 reasons for this. -

Introduction to Proteins and Amino Acids Introduction
Introduction to Proteins and Amino Acids Introduction • Twenty percent of the human body is made up of proteins. Proteins are the large, complex molecules that are critical for normal functioning of cells. • They are essential for the structure, function, and regulation of the body’s tissues and organs. • Proteins are made up of smaller units called amino acids, which are building blocks of proteins. They are attached to one another by peptide bonds forming a long chain of proteins. Amino acid structure and its classification • An amino acid contains both a carboxylic group and an amino group. Amino acids that have an amino group bonded directly to the alpha-carbon are referred to as alpha amino acids. • Every alpha amino acid has a carbon atom, called an alpha carbon, Cα ; bonded to a carboxylic acid, –COOH group; an amino, –NH2 group; a hydrogen atom; and an R group that is unique for every amino acid. Classification of amino acids • There are 20 amino acids. Based on the nature of their ‘R’ group, they are classified based on their polarity as: Classification based on essentiality: Essential amino acids are the amino acids which you need through your diet because your body cannot make them. Whereas non essential amino acids are the amino acids which are not an essential part of your diet because they can be synthesized by your body. Essential Non essential Histidine Alanine Isoleucine Arginine Leucine Aspargine Methionine Aspartate Phenyl alanine Cystine Threonine Glutamic acid Tryptophan Glycine Valine Ornithine Proline Serine Tyrosine Peptide bonds • Amino acids are linked together by ‘amide groups’ called peptide bonds. -

Biological Membranes
14 Biological Membranes To understand the structure The fundamental unit of life is the cell. All living things are composed of Goal and composition of biological cells, be it a single cell in the case of many microorganisms or a highly membranes. organized ensemble of myriad cell types in the case of multicellular organisms. A defining feature of the cell is a membrane, the cytoplasmic Objectives membrane, that surrounds the cell and separates the inside of the cell, the After this chapter, you should be able to cytoplasm, from other cells and the extracellular milieu. Membranes also • distinguish between cis and trans surround specialized compartments inside of cells known as organelles. unsaturated fatty acids. Whereas cells are typically several microns (μm) in diameter (although • explain why phospholipids some cells can be much larger), the membrane is only about 10 nanometers spontaneously form lipid bilayers and (nm) thick. Yet, and as we will see in subsequent chapters, the membrane is sealed compartments. not simply an ultra-thin, pliable sheet that encases the cytoplasm. Rather, • describe membrane fluidity and how it membranes are dynamic structures that mediate many functions in the is affected by membrane composition life of the cell. In this chapter we examine the composition of membranes, and temperature. their assembly, the forces that stabilize them, and the chemical and physical • explain the role of cholesterol in properties that influence their function. buffering membrane fluidity. The preceding chapters have focused on two kinds of biological molecules, • explain how the polar backbone namely proteins and nucleic acids, that are important in the workings of a membrane protein can be accommodated in a bilayer. -

Hydrophobic Catalysis and a Potential Biological Role of DNA Unstacking Induced by Environment Effects
Hydrophobic catalysis and a potential biological role of DNA unstacking induced by environment effects Bobo Fenga,1, Robert P. Sosab,c,d, Anna K. F. Mårtenssona, Kai Jiange, Alex Tongf, Kevin D. Dorfmang, Masayuki Takahashih, Per Lincolna, Carlos J. Bustamanteb,c,d,f,i,j,k,l, Fredrik Westerlunde, and Bengt Nordéna,1 aDepartment of Chemistry and Chemical Engineering, Chalmers University of Technology, 412 96 Gothenburg, Sweden; bBiophysics Graduate Group, University of California, Berkeley, CA 94720; cJason L. Choy Laboratory of Single Molecule Biophysics, University of California, Berkeley, CA 94720; dInstitute for Quantitative Biosciences-QB3, University of California, Berkeley, CA 94720; eDepartment of Biology and Biological Engineering, Chalmers University of Technology, 412 96 Gothenburg, Sweden; fDepartment of Chemistry, University of California, Berkeley, CA 94720; gDepartment of Chemical Engineering and Materials Science, University of Minnesota, Twin Cities, Minneapolis, MN 55455; hSchool of Life Science and Technology, Tokyo Institute of Technology, Tokyo 152-8550, Japan; iDepartment of Molecular & Cell Biology, University of California, Berkeley, CA 94720; jDepartment of Physics, University of California, Berkeley, CA 94720; kHoward Hughes Medical Institute, University of California, Berkeley, CA 94720; and lKavli Energy Nanosciences Institute, University of California, Berkeley, CA 94720 Edited by Feng Zhang, Broad Institute, Cambridge, MA, and approved July 22, 2019 (received for review May 27, 2019) Hydrophobic base stacking -

Introduction 1.1 Post-Translational Modifications
Chapter I: Introduction 1.1 Post-translational modifications Post-translational modifications (PTMs) are responsible for dynamic regulation of protein structure and function (Jensen et al., 2006). One example of the function of post-translational modifications in the dynamic regulation of protein structure and function is signal transduction; post-translational modification of key regulatory proteins in signal transduction pathways controls cellular proliferation, metabolism, gene expression, cytoskeletal organization and apoptosis (Jensen et al., 2006). Post-translational modifications not only change the mass of the protein, they are also often responsible for changes in charge, structure and conformation. This leads to changes in the biological activity of proteins like enzymes, the binding affinity of proteins to receptors and other protein-protein interactions (Hoffmann et al., 2008) (Murray et al., 1998). For example, the interaction of the cytokine TGF-beta with its receptor complex results in the phosphorylation of SMAD family proteins which activates these proteins to regulate target gene expression (Massague et al., 2000). Post-translational modified amino acid residues can act as binding sites on proteins for a specific recognition domain on other proteins. For example, phosphorylated tyrosine residues can bind to SH2 or PTB domains (Pawson et al., 1997). Depending on the type of PTM they can be very abundant and have a large number of target proteins, whereas other PTM`s have only a few target proteins. Moreover, some PTM`s are connected with several different residues and not only with one residue. For example multi-site phosphorylation that primarily targets Serine, Threonine and Tyrosine residues is a crucial mechanism for the regulation of protein localization and functional activity (Cohen et al., 2000). -

Predicting Hydrophobic Alkyl-Alkyl and Perfluoroalkyl-Perfluoroalkyl Van
Predicting hydrophobic alkyl-alkyl and perfluoroalkyl-perfluoroalkyl van der Waals dispersion interactions and their potential involvement in protein folding and the binding of drugs in hydrophobic pockets Clifford Fong To cite this version: Clifford Fong. Predicting hydrophobic alkyl-alkyl and perfluoroalkyl-perfluoroalkyl van der Waals dispersion interactions and their potential involvement in protein folding and the binding of drugs in hydrophobic pockets. [Research Report] Eigenenergy, Adelaide, Australia. 2019. hal-02176023v2 HAL Id: hal-02176023 https://hal.archives-ouvertes.fr/hal-02176023v2 Submitted on 5 Jan 2020 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Predicting hydrophobic alkyl-alkyl and perfluoroalkyl-perfluoroalkyl van der Waals dispersion interactions and their potential involvement in protein folding and the binding of drugs in hydrophobic pockets Clifford W. Fong Eigenenergy, Adelaide, South Australia. Email: [email protected] Abstract It has been shown that a model compound which has previously allowed the experimental determination of the hydrophobic van der Waals dispersion alkyl-alkyl folding free energy interaction in a wide range of polar and non-polar solvents is strongly correlated with the cavitation, dispersion, solvent-structure (CDS) solvation free energy and to a lesser extent the molecular polarizability of the foldamers. -

Probing the Base Stacking Contributions During
PROBING THE BASE STACKING CONTRIBUTIONS DURING TRANSLESION DNA SYNTHESIS by BABHO DEVADOSS Submitted in partial fulfillment of the requirements For the degree of Doctor of Philosophy Thesis Adviser: Dr. Irene Lee Department of Chemistry CASE WESTERN RESERVE UNIVERSITY January, 2009 CASE WESTERN RESERVE UNIVERSITY SCHOOL OF GRADUATE STUDIES We hereby approve the thesis/dissertation of _____________________________________________________ candidate for the ______________________degree *. (signed)_______________________________________________ (chair of the committee) ________________________________________________ ________________________________________________ ________________________________________________ ________________________________________________ ________________________________________________ (date) _______________________ *We also certify that written approval has been obtained for any proprietary material contained therein. ii TABLE OF CONTENTS Title Page…………………………………………………………………………………..i Committee Sign-off Sheet………………………………………………………………...ii Table of Contents…………………………………………………………………………iii List of Tables…………………………………………………………………………….vii List of Figures…………………………………………………………………….………ix Acknowledgements…………………………………………………………….………..xiv List of Abbreviations…………………………………………………………….………xv Abstract………………………………………………………………………………….xix CHAPTER 1 Introduction…………………………………………………………………1 1.1 The Chemistry and Biology of DNA…………………………………………2 1.2 DNA Synthesis……………………………………………………………….6 1.3 Structural Features of DNA Polymerases…………………………………...11 -

Structure and Synthesis of Antifungal Disulfide
microorganisms Review Structure and Synthesis of Antifungal Disulfide β-Strand Proteins from Filamentous Fungi Györgyi Váradi 1,*, Gábor K. Tóth 1,2 and Gyula Batta 3 1 Department of Medical Chemistry, Faculty of Medicine, University of Szeged, H-6720 Szeged, Hungary; [email protected] 2 MTA-SZTE Biomimetic Systems Research Group, University of Szeged, H-6720 Szeged, Hungary 3 Department of Organic Chemistry, University of Debrecen, H-4032 Debrecen, Hungary; [email protected] * Correspondence: [email protected]; Tel.: +36-62-545-142; Fax: +36-62-545-971 Received: 20 November 2018; Accepted: 24 December 2018; Published: 27 December 2018 Abstract: The discovery and understanding of the mode of action of new antimicrobial agents is extremely urgent, since fungal infections cause 1.5 million deaths annually. Antifungal peptides and proteins represent a significant group of compounds that are able to kill pathogenic fungi. Based on phylogenetic analyses the ascomycetous, cysteine-rich antifungal proteins can be divided into three different groups: Penicillium chrysogenum antifungal protein (PAF), Neosartorya fischeri antifungal protein 2 (NFAP2) and “bubble-proteins” (BP) produced, for example, by P. brevicompactum. They all dominantly have β-strand secondary structures that are stabilized by several disulfide bonds. The PAF group (AFP antifungal protein from Aspergillus giganteus, PAF and PAFB from P. chrysogenum, Neosartorya fischeri antifungal protein (NFAP)) is the best characterized with their common β-barrel tertiary structure. These proteins and variants can efficiently be obtained either from fungi production or by recombinant expression. However, chemical synthesis may be a complementary aid for preparing unusual modifications, e.g., the incorporation of non-coded amino acids, fluorophores, or even unnatural disulfide bonds.