The Hydrophobic Effect Oil and Water Do Not Mix. This Fact Is So Well
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Heterogeneous Impacts of Protein-Stabilizing Osmolytes On
bioRxiv preprint doi: https://doi.org/10.1101/328922; this version posted May 23, 2018. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Heterogeneous Impacts of Protein-Stabilizing Osmolytes on Hydrophobic Interaction Mrinmoy Mukherjee and Jagannath Mondal⇤ Tata Institute of Fundamental Research Hyderabad, 500107 India E-mail: [email protected],+914020203091 Abstract Osmolytes’ mechanism of protecting proteins against denaturation is a longstanding puzzle, further complicated by the complex diversities inherent in protein sequences. An emergent approach in understanding osmolytes’ mechanism of action towards biopoly- mer has been to investigate osmolytes’ interplay with hydrophobic interaction, the ma- jor driving force of protein folding. However, the crucial question is whether all these protein-stabilizing osmolytes display a single unified mechanism towards hydrophobic interactions. By simulating the hydrophobic collapse of a macromolecule in aqueous solutions of two such osmoprotectants, Glycine and Trimethyl N-oxide (TMAO), both of which are known to stabilize protein’s folded conformation, we here demonstrate that these two osmolytes can impart mutually contrasting effects towards hydropho- bic interaction. While TMAO preserves its protectant nature across diverse range of polymer-osmolyte interactions, glycine is found to display an interesting cross-over from being a protectant at weaker polymer-osmolyte interaction to a denaturant of hydrophobicity at stronger polymer-osmolyte interactions. A preferential-interaction analysis reveals that a subtle balance of conformation-dependent exclusion/binding of ⇤To whom correspondence should be addressed 1 bioRxiv preprint doi: https://doi.org/10.1101/328922; this version posted May 23, 2018. -

Protein Structure & Folding
6 Protein Structure & Folding To understand protein folding In the last chapter we learned that proteins are composed of amino acids Goal as a chemical equilibrium. linked together by peptide bonds. We also learned that the twenty amino acids display a wide range of chemical properties. In this chapter we will see Objectives that how a protein folds is determined by its amino acid sequence and that the three-dimensional shape of a folded protein determines its function by After this chapter, you should be able to: the way it positions these amino acids. Finally, we will see that proteins fold • describe the four levels of protein because doing so minimizes Gibbs free energy and that this minimization structure and the thermodynamic involves both making the most favorable bonds and maximizing disorder. forces that stabilize them. • explain how entropy (S) and enthalpy Proteins exhibit four levels of structure (H) contribute to Gibbs free energy. • use the equation ΔG = ΔH – TΔS The structure of proteins can be broken down into four levels of to determine the dependence of organization. The first is primary structure, the linear sequence of amino the favorability of a reaction on acids in the polypeptide chain. By convention, the primary sequence is temperature. written in order from the amino acid at the N-terminus (by convention • explain the hydrophobic effect and its usually on the left) to the amino acid at the C-terminus. The second level role in protein folding. of protein structure, secondary structure, is the local conformation adopted by stretches of contiguous amino acids. -

Is Colonic Propionate Delivery a Novel Solution to Improve Metabolism and Inflammation in Overweight Or Obese Subjects?
Commentary in IgG levels in IPE-treated subjects versus Is colonic propionate delivery a novel those receiving cellulose supplementa- tion. This interesting discovery is the Gut: first published as 10.1136/gutjnl-2019-318776 on 26 April 2019. Downloaded from solution to improve metabolism and first evidence in humans that promoting the delivery of propionate in the colon inflammation in overweight or may affect adaptive immunity. It is worth noting that previous preclinical and clin- obese subjects? ical data have shown that supplementation with inulin-type fructans was associated 1,2 with a lower inflammatory tone and a Patrice D Cani reinforcement of the gut barrier.7 8 Never- theless, it remains unknown if these effects Increased intake of dietary fibre has been was the lack of evidence that the observed are directly linked with the production of linked to beneficial impacts on health for effects were due to the presence of inulin propionate, changes in the proportion of decades. Strikingly, the exact mechanisms itself on IPE or the delivery of propionate the overall levels of SCFAs, or the pres- of action are not yet fully understood. into the colon. ence of any other bacterial metabolites. Among the different families of fibres, In GUT, Chambers and colleagues Alongside the changes in the levels prebiotics have gained attention mainly addressed this gap of knowledge and of SCFAs, plasma metabolome analysis because of their capacity to selectively expanded on their previous findings.6 For revealed that each of the supplementa- modulate the gut microbiota composition 42 days, they investigated the impact of tion periods was correlated with different 1 and promote health benefits. -
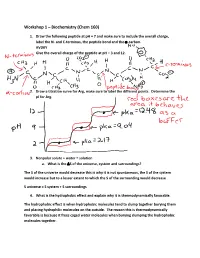
Workshop 1 – Biochemistry (Chem 160)
Workshop 1 – Biochemistry (Chem 160) 1. Draw the following peptide at pH = 7 and make sure to include the overall charge, label the N- and C-terminus, the peptide bond and the -carbon. AVDKY Give the overall charge of the peptide at pH = 3 and 12. 2. Draw a titration curve for Arg, make sure to label the different points. Determine the pI for Arg. 3. Nonpolar solute + water = solution a. What is the S of the universe, system and surroundings? The S of the universe would decrease this is why it is not spontaneous, the S of the system would increase but to a lesser extent to which the S of the surrounding would decrease S universe = S system + S surroundings 4. What is the hydrophobic effect and explain why it is thermodynamically favorable. The hydrophobic effect is when hydrophobic molecules tend to clump together burying them and placing hydrophilic molecules on the outside. The reason this is thermodynamically favorable is because it frees caged water molecules when burying clumping the hydrophobic molecules together. 5. Urea dissolves very readily in water, but the solution becomes very cold as the urea dissolves. How is this possible? Urea dissolves in water because when dissolving there is a net increase in entropy of the universe. The heat exchange, getting colder only reflects the enthalpy (H) component of the total energy change. The entropy change is high enough to offset the enthalpy component and to add up to an overall -G 6. A mutation that changes an alanine residue in the interior of a protein to valine is found to lead to a loss of activity. -

The Lower Critical Solution Temperature (LCST) Transition
Copyright by David Samuel Simmons 2009 The Dissertation Committee for David Samuel Simmons certifies that this is the approved version of the following dissertation: Phase and Conformational Behavior of LCST-Driven Stimuli Responsive Polymers Committee: ______________________________ Isaac Sanchez, Supervisor ______________________________ Nicholas Peppas ______________________________ Krishnendu Roy ______________________________ Venkat Ganesan ______________________________ Thomas Truskett Phase and Conformational Behavior of LCST-Driven Stimuli Responsive Polymers by David Samuel Simmons, B.S. Dissertation Presented to the Faculty of the Graduate School of The University of Texas at Austin in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy The University of Texas at Austin December, 2009 To my grandfather, who made me an engineer before I knew the word and to my wife, Carey, for being my partner on my good days and bad. Acknowledgements I am extraordinarily fortunate in the support I have received on the path to this accomplishment. My adviser, Dr. Isaac Sanchez, has made this publication possible with his advice, support, and willingness to field my ideas at random times in the afternoon; he has my deep appreciation for his outstanding guidance. My thanks also go to the members of my Ph.D. committee for their valuable feedback in improving my research and exploring new directions. I am likewise grateful to the other members of Dr. Sanchez’ research group – Xiaoyan Wang, Yingying Jiang, Xiaochu Wang, and Frank Willmore – who have shared their ideas and provided valuable sounding boards for my mine. I would particularly like to express appreciation for Frank’s donation of his own post-graduation time in assisting my research. -

Hydrophobic Effect, Water Structure, and Heat Capacity Changes
J. Phys. Chem. B 1997, 101, 4343-4348 4343 Hydrophobic Effect, Water Structure, and Heat Capacity Changes Kim A. Sharp* and Bhupinder Madan Department of Biochemistry & Biophysics, UniVersity of PennsylVania, 3700 Hamilton Walk, Philadelphia, PennsylVania 19104-6059 ReceiVed: January 16, 1997; In Final Form: April 1, 1997X hyd The hydration heat capacity (∆Cp ) of nine solutes of varying hydrophobicity was studied using a combination of a random network model equation of water and Monte Carlo simulations. Nonpolar solutes cause a concerted decrease in the average length and angle of the water-water hydrogen bonds in the first hydration shell, while polar and ionic solutes have the opposite effect. This is due to changes in the amounts, relative to bulk water, of two populations of hydrogen bonds: one with shorter and more linear bonds and the other with longer and more bent bonds. Heat capacity changes were calculated from these changes in water structure using a random network model equation of state. The calculated changes account for observed hyd differences in ∆Cp for the various solutes. The simulations provide a unified picture of hydrophobic and hyd polar hydration, and both a structural and thermodynamic explanation of the opposite signs of ∆Cp observed for polar and nonpolar solutes. Introduction nonpolar groups means that at higher temperatures (T > 80 °C) their insolubility results from unfavorable changes in enthalpy, In recent years analysis of heat capacity changes has become not entropy. Second, the hydration of a majority of polar and of central importance in understanding the thermodynamics of ionic solutes is also accompanied by a decrease in entropy, and protein folding, protein-protein binding, protein-nucleic acid thus by some kind of structuring of water, but in this case binding, and the hydrophobic effect.1-7 There are several hyd 7,16-20 reasons for this. -

Exercise and Cellular Respiration Lab
California State University of Bakersfield, Department of Chemistry Exercise and Cellular Respiration Lab Standards: MS-LS1-7 Develop a model to describe how food is rearranged through chemical reactions forming new molecules that support growth and/or release energy as this matter moves through an organism. Introduction: I. Background Information. Cellular respiration (see chemical reaction below) is a chemical reaction that occurs in your cells to create energy; when you are exercising your muscle cells are creating ATP to contract. Cellular respiration requires oxygen (which is breathed in) and creates carbon dioxide (which is breathed out). This lab will address how exercise (increased muscle activity) affects the rate of cellular respiration. You will measure 3 different indicators of cellular respiration: breathing rate, heart rate, and carbon dioxide production. You will measure these indicators at rest (with no exercise) and after 1 and 2 minutes of exercise. Breathing rate is measured in breaths per minute, heart rate in beats per minute, and carbon dioxide in the time it takes bromothymol blue to change color. Carbon dioxide production can be measured by breathing through a straw into a solution of bromothymol blue (BTB). BTB is an acid indicator; when it reacts with acid it turns from blue to yellow. When carbon dioxide reacts with water, a weak acid (carbonic acid) is formed (see chemical reaction below). The more carbon dioxide you breathe into the BTB solution, the faster it will change color to yellow. The purpose of this lab activity is to analyze the effect of exercise on cellular respiration. Background: I. -

Biological Membranes
14 Biological Membranes To understand the structure The fundamental unit of life is the cell. All living things are composed of Goal and composition of biological cells, be it a single cell in the case of many microorganisms or a highly membranes. organized ensemble of myriad cell types in the case of multicellular organisms. A defining feature of the cell is a membrane, the cytoplasmic Objectives membrane, that surrounds the cell and separates the inside of the cell, the After this chapter, you should be able to cytoplasm, from other cells and the extracellular milieu. Membranes also • distinguish between cis and trans surround specialized compartments inside of cells known as organelles. unsaturated fatty acids. Whereas cells are typically several microns (μm) in diameter (although • explain why phospholipids some cells can be much larger), the membrane is only about 10 nanometers spontaneously form lipid bilayers and (nm) thick. Yet, and as we will see in subsequent chapters, the membrane is sealed compartments. not simply an ultra-thin, pliable sheet that encases the cytoplasm. Rather, • describe membrane fluidity and how it membranes are dynamic structures that mediate many functions in the is affected by membrane composition life of the cell. In this chapter we examine the composition of membranes, and temperature. their assembly, the forces that stabilize them, and the chemical and physical • explain the role of cholesterol in properties that influence their function. buffering membrane fluidity. The preceding chapters have focused on two kinds of biological molecules, • explain how the polar backbone namely proteins and nucleic acids, that are important in the workings of a membrane protein can be accommodated in a bilayer. -

Brownie's THIRD LUNG
BrMARINEownie GROUP’s Owner’s Manual Variable Speed Hand Carry Hookah Diving System ADVENTURE IS ALWAYS ON THE LINE! VSHCDC Systems This manual is also available online 3001 NW 25th Avenue, Pompano Beach, FL 33069 USA Ph +1.954.462.5570 Fx +1.954.462.6115 www.BrowniesMarineGroup.com CONGRATULATIONS ON YOUR PURCHASE OF A BROWNIE’S SYSTEM You now have in your possession the finest, most reliable, surface supplied breathing air system available. The operation is designed with your safety and convenience in mind, and by carefully reading this brief manual you can be assured of many hours of trouble-free enjoyment. READ ALL SAFETY RULES AND OPERATING INSTRUCTIONS CONTAINED IN THIS MANUAL AND FOLLOW THEM WITH EACH USE OF THIS PRODUCT. MANUAL SAFETY NOTICES Important instructions concerning the endangerment of personnel, technical safety or operator safety will be specially emphasized in this manual by placing the information in the following types of safety notices. DANGER DANGER INDICATES AN IMMINENTLY HAZARDOUS SITUATION WHICH, IF NOT AVOIDED, WILL RESULT IN DEATH OR SERIOUS INJURY. THIS IS LIMITED TO THE MOST EXTREME SITUATIONS. WARNING WARNING INDICATES A POTENTIALLY HAZARDOUS SITUATION WHICH, IF NOT AVOIDED, COULD RESULT IN DEATH OR INJURY. CAUTION CAUTION INDICATES A POTENTIALLY HAZARDOUS SITUATION WHICH, IF NOT AVOIDED, MAY RESULT IN MINOR OR MODERATE INJURY. IT MAY ALSO BE USED TO ALERT AGAINST UNSAFE PRACTICES. NOTE NOTE ADVISE OF TECHNICAL REQUIREMENTS THAT REQUIRE PARTICULAR ATTENTION BY THE OPERATOR OR THE MAINTENANCE TECHNICIAN FOR PROPER MAINTENANCE AND UTILIZATION OF THE EQUIPMENT. REGISTER YOUR PRODUCT ONLINE Go to www.BrowniesMarineGroup.com to register your product. -

Hydrophobic Catalysis and a Potential Biological Role of DNA Unstacking Induced by Environment Effects
Hydrophobic catalysis and a potential biological role of DNA unstacking induced by environment effects Bobo Fenga,1, Robert P. Sosab,c,d, Anna K. F. Mårtenssona, Kai Jiange, Alex Tongf, Kevin D. Dorfmang, Masayuki Takahashih, Per Lincolna, Carlos J. Bustamanteb,c,d,f,i,j,k,l, Fredrik Westerlunde, and Bengt Nordéna,1 aDepartment of Chemistry and Chemical Engineering, Chalmers University of Technology, 412 96 Gothenburg, Sweden; bBiophysics Graduate Group, University of California, Berkeley, CA 94720; cJason L. Choy Laboratory of Single Molecule Biophysics, University of California, Berkeley, CA 94720; dInstitute for Quantitative Biosciences-QB3, University of California, Berkeley, CA 94720; eDepartment of Biology and Biological Engineering, Chalmers University of Technology, 412 96 Gothenburg, Sweden; fDepartment of Chemistry, University of California, Berkeley, CA 94720; gDepartment of Chemical Engineering and Materials Science, University of Minnesota, Twin Cities, Minneapolis, MN 55455; hSchool of Life Science and Technology, Tokyo Institute of Technology, Tokyo 152-8550, Japan; iDepartment of Molecular & Cell Biology, University of California, Berkeley, CA 94720; jDepartment of Physics, University of California, Berkeley, CA 94720; kHoward Hughes Medical Institute, University of California, Berkeley, CA 94720; and lKavli Energy Nanosciences Institute, University of California, Berkeley, CA 94720 Edited by Feng Zhang, Broad Institute, Cambridge, MA, and approved July 22, 2019 (received for review May 27, 2019) Hydrophobic base stacking -

Affinity of Small-Molecule Solutes to Hydrophobic, Hydrophilic, and Chemically Patterned Interfaces in Aqueous Solution
Affinity of small-molecule solutes to hydrophobic, hydrophilic, and chemically patterned interfaces in aqueous solution Jacob I. Monroea, Sally Jiaoa, R. Justin Davisb, Dennis Robinson Browna, Lynn E. Katzb, and M. Scott Shella,1 aDepartment of Chemical Engineering, University of California, Santa Barbara, CA 93106; and bDepartment of Civil, Architectural and Environmental Engineering, University of Texas at Austin, Austin, TX 78712 Edited by Peter J. Rossky, Rice University, Houston, TX, and approved November 17, 2020 (received for review September 30, 2020) Performance of membranes for water purification is highly influ- However, a molecular understanding that links membrane enced by the interactions of solvated species with membrane surface chemistry to solute affinity and hence membrane func- surfaces, including surface adsorption of solutes upon fouling. tional properties remains incomplete, due in part to the complex Current efforts toward fouling-resistant membranes often pursue interplay among specific interactions (e.g., hydrogen bonds, surface hydrophilization, frequently motivated by macroscopic electrostatics, dispersion) and molecular morphology (e.g., sur- measures of hydrophilicity, because hydrophobicity is thought to face and polymer configurations) that are difficult to disentangle increase solute–surface affinity. While this heuristic has driven di- (11–14). Chemically heterogeneous surfaces are even less un- verse membrane functionalization strategies, here we build on derstood but can affect fouling in complex ways. -

Peptides & Proteins
Peptides & Proteins (thanks to Hans Börner) 1 Proteins & Peptides Proteuos: Proteus (Gr. mythological figure who could change form) proteuo: „"first, ref. the basic constituents of all living cells” peptos: „Cooked referring to digestion” Proteins essential for: Structure, metabolism & cell functions 2 Bacterium Proteins (15 weight%) 50% (without water) • Construction materials Collagen Spider silk proteins 3 Proteins Structural proteins structural role & mechanical support "cell skeleton“: complex network of protein filaments. muscle contraction results from action of large protein assemblies Myosin und Myogen. Other organic material (hair and bone) are also based on proteins. Collagen is found in all multi cellular animals, occurring in almost every tissue. • It is the most abundant vertebrate protein • approximately a quarter of mammalian protein is collagen 4 Proteins Storage Various ions, small molecules and other metabolites are stored by complexation with proteins • hemoglobin stores oxygen (free O2 in the blood would be toxic) • iron is stored by ferritin Transport Proteins are involved in the transportation of particles ranging from electrons to macromolecules. • Iron is transported by transferrin • Oxygen via hemoglobin. • Some proteins form pores in cellular membranes through which ions pass; the transport of proteins themselves across membranes also depends on other proteins. Beside: Regulation, enzymes, defense, functional properties 5 Our universal container system: Albumines 6 zones: IA, IB, IIA, IIB, IIIA, IIIB; IIB and