Acute Exposure Guideline Levels for Selected Airborne Chemicals
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Problem Formulation of the Risk Evaluation for Perchloroethylene (Ethene, 1,1,2,2-Tetrachloro)
EPA Document# EPA-740-R1-7017 May 2018 DRAFTUnited States Office of Chemical Safety and Environmental Protection Agency Pollution Prevention Problem Formulation of the Risk Evaluation for Perchloroethylene (Ethene, 1,1,2,2-Tetrachloro) CASRN: 127-18-4 May 2018 TABLE OF CONTENTS ABBREVIATIONS ............................................................................................................................ 8 EXECUTIVE SUMMARY .............................................................................................................. 11 1 INTRODUCTION .................................................................................................................... 14 1.1 Regulatory History ..................................................................................................................... 16 1.2 Assessment History .................................................................................................................... 16 1.3 Data and Information Collection ................................................................................................ 18 1.4 Data Screening During Problem Formulation ............................................................................ 19 2 PROBLEM FORMULATION ................................................................................................. 20 2.1 Physical and Chemical Properties .............................................................................................. 20 2.2 Conditions of Use ...................................................................................................................... -

Toxicological Profile for Tetrachloroethylene
TETRACHLOROETHYLENE 263 6. POTENTIAL FOR HUMAN EXPOSURE 6.1 OVERVIEW Tetrachloroethylene has been identified in at least 949 of the 1,854 hazardous waste sites that have been proposed for inclusion on the EPA National Priorities List (NPL) (ATSDR 2017a). However, the number of sites evaluated for tetrachloroethylene is not known. The frequency of these sites can be seen in Figure 6-1. Of these sites, 943 are located within the United States, 3 are located in the Commonwealth of Puerto Rico (not shown), 1 is located in Guam (not shown), and 1 is located in the Virgin Islands (not shown). Tetrachloroethylene is a VOC that is widely distributed in the environment. It is released to the environment via industrial emissions and from building and consumer products. Releases are primarily to the atmosphere. However, the compound is also released to surface water and land in sewage sludges and in other liquid and solid waste, where its high vapor pressure and Henry's law constant usually result in its rapid volatilization to the atmosphere. Tetrachloroethylene has relatively low solubility in water and has medium-to-high mobility in soil; thus, its residence time in surface environments is not expected to be more than a few days. However, it persists in the atmosphere for several months and can last for decades in the groundwater. Background tetrachloroethylene levels in outdoor air are typically <1 μg/m3 (0.15 ppb) for most locations (EPA 2013h, 2018); however, indoor air at source dominant facilities (e.g., dry cleaners) has been shown to have levels >1 mg/m3 (Chiappini et al. -

1,1,1,2-Tetrafluoroethane
This report contains the collective views of an international group of experts and does not necessarily represent the decisions or the stated policy of the United Nations Environment Programme, the International Labour Organisation, or the World Health Organization. Concise International Chemical Assessment Document 11 1,1,1,2-Tetrafluoroethane First draft prepared by Mrs P. Barker and Mr R. Cary, Health and Safety Executive, Liverpool, United Kingdom, and Dr S. Dobson, Institute of Terrestrial Ecology, Huntingdon, United Kingdom Please not that the layout and pagination of this pdf file are not identical to the printed CICAD Published under the joint sponsorship of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization, and produced within the framework of the Inter-Organization Programme for the Sound Management of Chemicals. World Health Organization Geneva, 1998 The International Programme on Chemical Safety (IPCS), established in 1980, is a joint venture of the United Nations Environment Programme (UNEP), the International Labour Organisation (ILO), and the World Health Organization (WHO). The overall objectives of the IPCS are to establish the scientific basis for assessment of the risk to human health and the environment from exposure to chemicals, through international peer review processes, as a prerequisite for the promotion of chemical safety, and to provide technical assistance in strengthening national capacities for the sound management of chemicals. The Inter-Organization -

Functionalization of Heterocycles: a Metal Catalyzed Approach Via
FUNCTIONALIZATION OF HETEROCYCLES: A METAL CATALYZED APPROACH VIA ALLYLATION AND C-H ACTIVATION A Dissertation Submitted to the Graduate Faculty of the North Dakota State University of Agriculture and Applied Science By Sandeepreddy Vemula In Partial Fulfillment of the Requirements for the Degree of DOCTOR OF PHILOSOPHY Major Department: Chemistry and Biochemistry October 2018 Fargo, North Dakota North Dakota State University Graduate School Title FUNCTIONALIZATION OF HETEROCYCLES: A METAL CATALYZED APPROACH VIA ALLYLATION AND C-H ACTIVATION By Sandeepreddy Vemula The Supervisory Committee certifies that this disquisition complies with North Dakota State University’s regulations and meets the accepted standards for the degree of DOCTOR OF PHILOSOPHY SUPERVISORY COMMITTEE: Prof. Gregory R. Cook Chair Prof. Mukund P. Sibi Prof. Pinjing Zhao Prof. Dean Webster Approved: November 16, 2018 Prof. Gregory R. Cook Date Department Chair ABSTRACT The central core of many biologically active natural products and pharmaceuticals contain N-heterocycles, the installation of simple/complex functional groups using C-H/N-H functionalization methodologies has the potential to dramatically increase the efficiency of synthesis with respect to resources, time and overall steps to key intermediate/products. Transition metal-catalyzed functionalization of N-heterocycles proved as a powerful tool for the construction of C-C and C-heteroatom bonds. The work in this dissertation describes the development of palladium catalyzed allylation, and the transition metal catalyzed C-H activation for selective functionalization of electron deficient N-heterocycles. Chapter 1 A thorough study highlighting the important developments made in transition metal catalyzed approaches for C-C and C-X bond forming reactions is discussed with a focus on allylation, directed indole C-2 substitution and vinylic C-H activation. -

1,2-DICHLOROETHANE 1. Exposure Data
1,2-DICHLOROETHANE Data were last reviewed in IARC (1979) and the compound was classified in IARC Monographs Supplement 7 (1987a). 1. Exposure Data 1.1 Chemical and physical data 1.1.1 Nomenclature Chem. Abstr. Serv. Reg. No.: 107-06-2 Chem. Abstr. Name: 1,2-Dichloroethane IUPAC Systematic Name: 1,2-Dichloroethane Synonym: Ethylene dichloride 1.1.2 Structural and molecular formulae and relative molecular mass Cl CH2 CH2 Cl C2H4Cl2 Relative molecular mass: 98.96 1.1.3 Chemical and physical properties of the pure substance (a) Description: Colourless liquid with a pleasant odour (Budavari, 1996) (b) Boiling-point: 83.5°C (Lide, 1995) (c) Melting-point: –35.5°C (Lide, 1995) (d) Solubility: Slightly soluble in water; miscible with ethanol, chloroform and diethyl ether (Lide, 1995; Budavari, 1996) (e) Vapour pressure: 8 kPa at 20°C (Verschueren, 1996) (f) Flash-point: 18°C, open cup (Budavari, 1996) (g) Conversion factor: mg/m3 = 4.0 × ppm 1.2 Production and use World production capacities in 1988 for 1,2-dichloroethane have been reported as follows (thousand tonnes): North America, 9445; western Europe, 9830; Japan, 3068; and other, 8351 (Snedecor, 1993). Production in the United States has been reported as follows (thousand tonnes): 1983, 5200; 1990, 6300; 1991, 6200; 1992, 6900; 1993, 8100 (United States National Library of Medicine, 1997). The total annual production in Canada in 1990 was estimated to be 922 thousand tonnes; more than 1000 thousand tonnes were produced in the United Kingdom in 1991 (WHO, 1995). –501– 502 IARC MONOGRAPHS VOLUME 71 1,2-Dichloroethane is used primarily in the production of vinyl chloride; 99% of total demand in Canada, 90% in Japan and 88% of total production in the United States are used for this purpose. -

Prepared by 3M
DRAFT ECOTOXICOLOGY AND ENVIRONMENTAL FATE TESTING OF SHORT CHAIN PERFLUOROALKYL COMPOUNDS RELATED TO 3M CHEMISTRIES XXX XX, 2008 Prepared by 3M Page 1 CONFIDENTIAL - SUBJECT TO A PROTECTIVE ORDER ENTERED IN 3M MN01537089 HENNEPIN COUNTY DISTRICT COURT, NO. 27-CV-10-28862 2231.0001 DRAFT TABLE OF CONTENTS Page Introduction (describes propose, goals, document organization .... ) 3 1) Degradation modes 4-27 Basic degradation pathways, Conversion routes from polymer to monomer & degradants or residual intermediates to degradants) 2) Degradants & Intermediates - Phys. Chem properties and test data by section & chapter i) Fluorinated Sulfonic acids and derivatives a) PFBS (PBSK) 28-42 b) PFBSI 43-44 ii) Fluorinated Carboxylates a) TFA 46-55 b) PFPA 56-59 c) PFBA (APFB) 60-65 d) MeFBSAA (MeFBSE Acid, M370) 66-68 iii) Fluorinated Non-ionics a) MeFBSE 70-74 b) MeFBSA 75-80 c) FBSE 81-84 d) FBSA 85-88 e) HxFBSA 89-91 iv) Fluorinated Inerts & volatiles a) PBSF 94-97 b) NFB, C4 hydride, CF3CF2CF2CF2-H 98-99 3) Assessments X Glossary 100-102 References/Endnotes 105- Page 2 CONFIDENTIAL - SUBJECT TO A PROTECTIVE ORDER ENTERED IN 3M MN01537090 HENNEPIN COUNTY DISTRICT COURT, NO. 27-CV-10-28862 2231.0002 DRAFT Introduction to Environmental White Papers on Perfluoroalkyl acids related to 3M chemistries Perfluorochemicals have been commonplace in chemical industry over 50 years but until recently there has been little information on environmental fate and effects avialble in open literature. The following chapters summarize the findings of"list specific C4 intermediates PFBS, PFBSI, PFBA, PFPA, MeFBSAA, TFA, MeFBSE, FBSA, HxFBSA, FBSE, PBSF, NFB " As background, 3M announced on May 16, 2000 the voluntary manufacturing phase out of perfluorooctanyl chemicals which included perfluorooctanoic acid (PFOA), perfluorooctane sulfonate (PFOS), and PFOS-related chemistries. -

Tetrachloroethylene Facts
TETRACHLOROETHYLENE CAS # 127-18-4 Agency for Toxic Substances and Disease Registry ToxFAQs September 1997 This fact sheet answers the most frequently asked health questions (FAQs) about tetrachloroethylene. For more information, call the ATSDR Information Center at 1-888-422-8737. This fact sheet is one in a series of summaries about hazardous substances and their health effects. It’s important you understand this information because this substance may harm you. The effects of exposure to any hazardous substance depend on the dose, the duration, how you are exposed, personal traits and habits, and whether other chemicals are present. HIGHLIGHTS: Tetrachloroethylene is a manufactured chemical used for dry cleaning and metal degreasing. Exposure to very high concentrations of tetrachloroethylene can cause dizziness, headaches, sleepiness, confusion, nausea, difficulty in speaking and walking, unconsciousness, and death. Tetrachloroethylene has been found in at least 771 of the 1,430 National Priorities List sites identified by the Environmental Protection Agency (EPA). What is tetrachloroethylene? How might I be exposed to tetrachloroethylene? (Pronounced tµt”r…-klôr“ ½-µth“…-l¶n”) q When you bring clothes from the dry cleaners, they will release small amounts of tetrachloroethylene into the air. Tetrachloroethylene is a manufactured chemical that is q widely used for dry cleaning of fabrics and for metal-degreas- When you drink water containing tetrachloroethylene, you are exposed to it. ing. It is also used to make other chemicals and is used in some consumer products. Other names for tetrachloroethylene include perchloroet- How can tetrachloroethylene affect my health? hylene, PCE, and tetrachloroethene. It is a nonflammable High concentrations of tetrachloroethylene (particularly liquid at room temperature. -

Poly(Limonene Carbonate): a Bio-Based & Versatile High-Performance Thermoplastic
Poly(limonene carbonate): a bio-based & versatile high-performance thermoplastic Dissertation zur Erlangung des akademischen Grades eines Doktors der Naturwissenschaften (Dr. rer. Nat.) an der Bayreuther Graduiertenschule für Mathematik und Naturwissenschaften (BayNAT) der Universität Bayreuth vorgelegt von Oliver Hauenstein aus Dortmund Bayreuth 2016 Die vorliegende Arbeit wurde in der Zeit von November 2012 bis Juni 2016 in Bayreuth am Lehrstuhl Makromolekulare Chemie II unter Betreuung von Herrn Professor Dr. Andreas Greiner angefertigt. Vollständiger Abdruck der von der Bayreuther Graduiertenschule für Mathematik und Naturwissenschaften (BayNAT) der Universität Bayreuth genehmigten Dissertation zur Erlangung des akademischen Grades eines Doktors der Naturwissenschaften (Dr. rer. nat.). Dissertation eingereicht am: 13.07.2016 Zulassung durch das Leitungsgremium: 26.07.2016 Wissenschaftliches Kolloquium: 01.02.2017 Amtierender Direktor: Prof. Dr. Stephan Kümmel Prüfungsausschuss: Prof. Dr. Andreas Greiner (Erstgutachter) Prof. Dr. Hans-Werner Schmidt (Zweitgutachter) Prof. Dr. Carlo Unverzagt. (Vorsitz) Prof. Dr. Jürgen Senker Drittgutachter: Prof. Dr. Volker Abetz Für meine Familie & Hui Contents Abbreviations & symbols ......................................................................................................................... i List of publications ................................................................................................................................... v Abstract .................................................................................................................................................... -
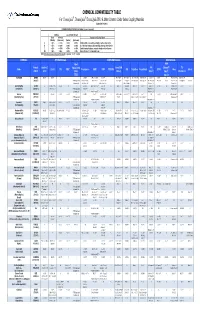
CPC Chemical Compatibility Chart
CHEMICAL COMPATIBILITY TABLE For ChemQuik®, DrumQuik®, DrumQuik PRO & Other Common Colder Series Coupling Materials (Updated 01/14/2010) INTERPRETATION OF TEST DATA (In 30 days to 1 year of exposure) Swelling Loss of Tensile Strength Linear Volumetric Description of Chemical Attack (Plastics) (Elastomers) (Plastics) (Elastomers) A < 10% <= 15% < 15% <=15% Excellent, little or no swelling, softening or surface deterioration B < 15% <= 30% < 30% <= 30% Good chemical resistance, minor swelling, softening or deterioration C < 20% <= 50% < 50% <= 60% Limited chemical resistance, moderate attack, conditional service NR > 20% > 50% > 50% > 60% Severe attack, not recommended for use NOTE: All temperatures are in degrees Fahrenheit. Conversion: °C = (°F - 32)/1.8 CHEMICAL SPRING Materials COUPLING Materials SEAL Materials Teflon® FFKM ® Formula Hastelloy C Encapsulated Acetal/POM FKM (Chemraz / TPO Name 316 SS PPS PEEK™ Polypropylene HDPE PVDF PTFE/PFA ABS Polysulfone Polycarbonate EPDM Buna Silicone (CAS #) (276) 316SS (Celcon) (Viton®) Simriz® / (Santoprene) (TESS) Kalrez®) Acetic Acid C2H4O2 A to 212° 212° A to 212° 212° A A A A to 140° 140° AB to 100% to 70° 70° A to 122° 122° A AAto5%to70 to 5% to 70°° AB 10% to 70° 70° A to 100% to 70° 70° A to 50% to 70° 70° A 10% to 70° 70° AAto70 to 70°° A B to 30% at 70° 70° A to 30% to 70° 70° A (64-19-7) (PTFE Encapsulated AB 50-100% to 160° AB 60% to 180° A to 10% to 225° BC 10% @ 70° C 20% @ 70° A to 20% to 140° B to 50% @ 122° B 10-25% to 100° AB to 200° A to 70° B to 20% to 185° C 50% @ 70° A to -

Chemicals Used in Drycleaning Operations
Chemicals Used In Drycleaning Operations January, 2002 Revised July 2009 Chemicals Search Menu The following resource was developed for the State Coalition for Remediation of Drycleaners (SCRD) using material safety data sheets (MSDS) and other sources. The report was prepared by Bill Linn, Florida Department of Environmental Protection (FDEP). Scott Stupak, North Carolina Superfund Section, provided technical support for database development. INTRODUCTION A wide variety of chemicals has been used and is currently utilized in drycleaning operations. Using material safety data sheets (MSDS) and other sources; a drycleaning chemical data base was developed that includes many of the chemicals that have been used in drycleaning operations. These data and the accompanying text are intended to aid those engaged in the assessment and remediation of contaminated drycleaning sites and to assist regulators conducting compliance inspections at drycleaning facilities. Some of the chemicals/products listed on the spreadsheet are no longer manufactured, marketed or used in drycleaning operations. Drycleaning Chemical Data Spread Sheet The spread sheet is divided into the following categories: • Chemical product or trade name (as listed on the MSDS) • Chemical manufacturer or distributor • Use or function of the product • Additional information • Chemical constituent(s) as listed on the MSDS • Chemical Abstract Numbers (CAS #s) for listed constituents • Relative concentration of the constituent in the product All product ingredients or constituents that appear on the MSDS were listed on the spread sheet – both hazardous and non-hazardous. The manner in which product constituents are reported on MSDS varies widely. Many manufacturers/vendors simply list all ingredients as being proprietary (trade secrets). -

Laboratory and Modelling Study Evaluating Thermal Remediation of Tetrachloroethene and Multi- Component Napl Impacted Soil
LABORATORY AND MODELLING STUDY EVALUATING THERMAL REMEDIATION OF TETRACHLOROETHENE AND MULTI- COMPONENT NAPL IMPACTED SOIL by Chen Zhao A thesis submitted to the Department of Civil Engineering In conformity with the requirements for Master of Applied Science Queen’s University Kingston, Ontario, Canada (September 2013) Copyright © Chen Zhao, 2013 Abstract Dense non-aqueous phase liquids (DNAPLs) such as chlorinated solvents, coal tar and creosote are some of the most problematic groundwater and soil contaminants in industrialized countries throughout the world. In Situ Thermal Treatment (ISTT) is a candidate remediation technology for this class of contaminants. However, the relationships between gas production, gas flow, and contaminant mass removal during ISTT are not fully understood. A laboratory study was conducted to assess the degree of mass removal, as well as the gas generation rate and the composition of the gas phase as a function of different heating times and initial DNAPL saturations. Experiments consisted of a soil, water, and DNAPL mixture packed in a 1 L jar that was heated in an oven. Gas produced during each experiment, which consisted of steam and volatile organic compounds (VOCs), was collected and condensed to quantify the gas generation rate. The temperature of the contaminated soil was measured continuously using a thermocouple embedded in the source zone, and was used to identify periods of heating, co-boiling and boiling. Samples were collected from the aqueous and DNAPL phase of the condensate, as well as from the source soil, at different heating times, and analyzed by gas chromatography/mass spectrometry. In addition to laboratory experiments, a mathematical model was developed to predict the co-boiling temperature and transient composition of the gas phase during heating of a uniform source. -

1,1,1,2-Tetrafluoroethane (HFC-134A) (CAS No. 811-97-2) (Second Edition)
1,1,1,2-Tetrafluoroethane (HFC-134a) (CAS No. 811-97-2) (Second Edition) JACC No. 50 ISSN-0773-6339-50 Brussels, January 2006 1,1,1,2-Tetrafluoroethane (HFC-134a) (CAS No. 811-97-2) (Second Edition) ECETOC JACC REPORT No. 50 © Copyright – ECETOC AISBL European Centre for Ecotoxicology and Toxicology of Chemicals 4 Avenue E. Van Nieuwenhuyse (Bte 6), B-1160 Brussels, Belgium. All rights reserved. No part of this publication may be reproduced, copied, stored in a retrieval system or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise without the prior written permission of the copyright holder. Applications to reproduce, store, copy or translate should be made to the Secretary General. ECETOC welcomes such applications. Reference to the document, its title and summary may be copied or abstracted in data retrieval systems without subsequent reference. The content of this document has been prepared and reviewed by experts on behalf of ECETOC with all possible care and from the available scientific information. It is provided for information only. ECETOC cannot accept any responsibility or liability and does not provide a warranty for any use or interpretation of the material contained in the publication. ECETOC JACC No. 50 1,1,1,2-Tetrafluoroethane (HFC-134a) (CAS No. 811-97-2) (Second Edition) 1,1,1,2-Tetrafluoroethane (HFC-134a) (CAS No. 811-97-2) CONTENTS EXECUTIVE SUMMARY 1 THE ECETOC SCHEME FOR THE JOINT ASSESSMENT OF COMMODITY CHEMICALS 3 1. SUMMARY AND CONCLUSIONS 4 2. IDENTITY, PHYSICAL AND CHEMICAL PROPERTIES, ANALYTICAL METHODS 6 2.1 Identity 6 2.2 EU classification and labelling 6 2.3 Physical and chemical properties 6 2.4 Conversion factors 8 2.5 Analytical methods 8 3.