Minimizing Galvanic Corrosion Tech Data
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Treatise on Combined Metalworking Techniques: Forged Elements and Chased Raised Shapes Bonnie Gallagher
Rochester Institute of Technology RIT Scholar Works Theses Thesis/Dissertation Collections 1972 Treatise on combined metalworking techniques: forged elements and chased raised shapes Bonnie Gallagher Follow this and additional works at: http://scholarworks.rit.edu/theses Recommended Citation Gallagher, Bonnie, "Treatise on combined metalworking techniques: forged elements and chased raised shapes" (1972). Thesis. Rochester Institute of Technology. Accessed from This Thesis is brought to you for free and open access by the Thesis/Dissertation Collections at RIT Scholar Works. It has been accepted for inclusion in Theses by an authorized administrator of RIT Scholar Works. For more information, please contact [email protected]. TREATISE ON COMBINED METALWORKING TECHNIQUES i FORGED ELEMENTS AND CHASED RAISED SHAPES TREATISE ON. COMBINED METALWORKING TECHNIQUES t FORGED ELEMENTS AND CHASED RAISED SHAPES BONNIE JEANNE GALLAGHER CANDIDATE FOR THE MASTER OF FINE ARTS IN THE COLLEGE OF FINE AND APPLIED ARTS OF THE ROCHESTER INSTITUTE OF TECHNOLOGY AUGUST ( 1972 ADVISOR: HANS CHRISTENSEN t " ^ <bV DEDICATION FORM MUST GIVE FORTH THE SPIRIT FORM IS THE MANNER IN WHICH THE SPIRIT IS EXPRESSED ELIEL SAARINAN IN MEMORY OF MY FATHER, WHO LONGED FOR HIS CHILDREN TO HAVE THE OPPORTUNITY TO HAVE THE EDUCATION HE NEVER HAD THE FORTUNE TO OBTAIN. vi PREFACE Although the processes of raising, forging, and chasing of metal have been covered in most technical books, to date there is no major source which deals with the functional and aesthetic requirements -

Repoussé Work for Amateurs
rf Bi oN? ^ ^ iTION av op OCT i 3 f943 2 MAY 8 1933 DEC 3 1938 MAY 6 id i 28 dec j o m? Digitized by the Internet Archive in 2011 with funding from Boston Public Library http://www.archive.org/details/repoussworkforamOOhasl GROUP OF LEAVES. Repousse Work for Amateurs. : REPOUSSE WORK FOR AMATEURS: BEING THE ART OF ORNAMENTING THIN METAL WITH RAISED FIGURES. tfjLd*- 6 By L. L. HASLOPE. ILLUSTRATED. LONDON L. UPCOTT GILL, 170, STRAND, W.C, 1887. PRINTED BY A. BRADLEY, 170, STRAND, LONDON. 3W PREFACE. " JjJjtfN these days, when of making books there is no end," ^*^ and every description of work, whether professional or amateur, has a literature of its own, it is strange that scarcely anything should have been written on the fascinating arts of Chasing and Repousse Work. It is true that a few articles have appeared in various periodicals on the subject, but with scarcely an exception they treated only of Working on Wood, and the directions given were generally crude and imperfect. This is the more surprising when we consider how fashionable Repousse Work has become of late years, both here and in America; indeed, in the latter country, "Do you pound brass ? " is said to be a very common question. I have written the following pages in the hope that they might, in some measure, supply a want, and prove of service to my brother amateurs. It has been hinted to me that some of my chapters are rather "advanced;" in other words, that I have gone farther than amateurs are likely to follow me. -

Hercules Jewelry Project
Hercules Knot Jewelry Series I thoroughly enjoyed my wire wrapping jewelry projects, but in the course of researching and building the class, I was inspired to learn real metalsmithing. I’ve been assembling earrings and necklaces for decades, but suddenly I really wanted to solder, and set gems, and fabricate! Fortunately, my good friend Fjorlief InHaga is a brilliant artisan, and she’s been teaching me and letting me use her studio. THE HERCULES KNOT aka HERAKLES, Reef Knot or Square Knot This motif represents strength and came to be associated with marriage. Brides wore their belts tied this way (see my paper on Roman Weddings). It was a common motif in the Greco-Roman world though the Republic and Early – Mid Empire. 1 2 3 Above, gold bracelet and ring from the 1st century BCE – 2nd CE. Silver mirror, 4th century CE. I imagine this would have been a lovely wedding present. Below: Necklaces dated to the 2nd-3rd century, an armband (#6), and rings from 5rd century BCE to 3rd CE . I have included only a small number of the large quantity of eXtant artifacts. Combined with the variations in assembly technique and clasps, the abundance of pieces shows that the Hercules Knot was a style that was widespread and made by many different artisans. 4 5 6 7 8 9 10 For my first project, I wanted to duplicate the look of these early Imperial necklaces of emerald and gold. For cost purposes, I used brass and glass, which was a commonly-used substitute for stones in period. -

Guide to Stainless Steel Finishes
Guide to Stainless Steel Finishes Building Series, Volume 1 GUIDE TO STAINLESS STEEL FINISHES Euro Inox Euro Inox is the European market development associa- Full Members tion for stainless steel. The members of Euro Inox include: Acerinox, •European stainless steel producers www.acerinox.es • National stainless steel development associations Outokumpu, •Development associations of the alloying element www.outokumpu.com industries. ThyssenKrupp Acciai Speciali Terni, A prime objective of Euro Inox is to create awareness of www.acciaiterni.com the unique properties of stainless steels and to further their use in existing applications and in new markets. ThyssenKrupp Nirosta, To assist this purpose, Euro Inox organises conferences www.nirosta.de and seminars, and issues guidance in printed form Ugine & ALZ Belgium and electronic format, to enable architects, designers, Ugine & ALZ France specifiers, fabricators, and end users, to become more Groupe Arcelor, www.ugine-alz.com familiar with the material. Euro Inox also supports technical and market research. Associate Members British Stainless Steel Association (BSSA), www.bssa.org.uk Cedinox, www.cedinox.es Centro Inox, www.centroinox.it Informationsstelle Edelstahl Rostfrei, www.edelstahl-rostfrei.de Informationsstelle für nichtrostende Stähle SWISS INOX, www.swissinox.ch Institut de Développement de l’Inox (I.D.-Inox), www.idinox.com International Chromium Development Association (ICDA), www.chromium-asoc.com International Molybdenum Association (IMOA), www.imoa.info Nickel Institute, www.nickelinstitute.org -

Weights of Various Metals in Pounds Per *Cubic Foot
Weights of Various Metals in Pounds Per *Cubic Foot Aluminum 168.48 Copper 559.87 Manganese 463.10 Silver 654.91 Antimony 419.99 Gold 1206.83 Mercury 849.00 Stainless Steel (18-8) 494.21 Beryllium 113.70 Gun Metal (Aver.) 544.00 Molybdenum 637.63 Steel, Cast/Rolled 490.00 Bismuth 611.00 Iridium 1396.00 Monel Metal 556.00 Tin 455.67 Brass (Approx.) 535.68 Iron 491.09 Nickel 555.72 Titanium 283.39 Bronze, Alum. 481.00 Iron, Cast Grey 442.00 Osmium 1402.00 Tungsten 1204.41 Bronze (Approx.) 541.00 Iron, Wrought 480.00 Palladium 712.00 Vanadium 374.97 Cadmium 540.86 Iron, Slag 172.00 Platinum 1339.20 Zinc 445.30 Chromium 428.00 Lead 707.96 Rhodium 755.00 *1728 CU. IN. PER CU. FT. Cobalt 552.96 Magnesium 108.51 Ruthenium 765.00 Weights of Other Materials in Pounds Per *Cubic Foot Acids, Muriatic, 40% 75.00 Earth, Common Loam 75.00-90.00 Maple, Hard 43.00 Rubber, Goods 94.00 Acids, Nitric, 91% 94.00 Earth, Dry/Loose 76.00 Maple, White 33.00 Sand, Clay & Earth, Dry 100.00 Acids, Sulphuric, 87% 112.00 Earth, Dry/Packed 95.00 Marble 170.00 Sand, Clay & Earth, Wet 120.00 Alcohol, 100% 49.00 Earth, Mud/Packed 115.00 Masonry, Rubble 130.00-150.00 Sand, Pure Quartz, Dry90.00-106.00 Asbestos 153.00 Elm, White 45.00 Masonry, Ashlar 140.00-160.00 Sandstone, Blustone 147.00 Ash, White, Red 40.00 Fats 58.00 Mortar 100.00 Slate 175.00 Asphaltum 81.00 Fir, Douglas 30.00 Mud, River 90.00 Snow, Freshly Fallen 10.00 Basalt 184.00 Fir, Eastern 25.00 Oil, Kerosene 52.00 Snow, Wet 50.00 Brick, Paving 150.00 Flour, Loose 28.00 Oil, Mineral 57.00 Soap Stone, Tall 169.00 Brick, Com. -

6 W Elding Accessories Tungsten Electrodes Magnesium Aluminum
Sylvania Tungsten Vendor Code: SYL Tungsten Electrodes Magnesium Magnesium alloys are in 3 groups. They are: (1) aluminum-zinc-magnesium, (2) aluminum-magnesium and (3) manganese-magnesium. Since magnesium will absorb a number of harmful ingredients and oxidize rapidly when subjected to welding heat, TIG welding in an inert gas atmosphere is distinctly advantageous. The welding of magnesium is similar, in many respects, to the welding of aluminum. Magnesium was one of the first metals to be welded commercially by the inert-gas nonconsumable process (TIG). Accessories Welding Aluminum The use of TIG welding for aluminum has many advantages for both manual and automatic processes. Filler metal can be either wire or rod and should be compatible with the base alloy. Filler metal must be Ground Dia. Length Electrodes dry, free of oxides, grease or other foreign matter. If filler metal becomes damp, heat for 2 hours at Part No. (inches) (inches) 250˚ F before using. Although AC high-frequency stabilized current is recommended, DC reverse polarity 0407G .040 7 has been successfully used for thicknesses up to 3/32". 1167G 1/16 7 Stainless Steel Pure 3327G 3/32 7 In TIG welding of stainless steel, welding rods having the AWS-ASTM prefixes of E or ER can be used as 187G 1/8 7 filler rods. However, only bare uncoated rods should be used. Stainless steel can be welded using AC high frequency stabilized current, however, for DC straight polarity current recommendations must be increased 5327G 5/32 7 6 25%. Light gauge metal less than 1/16" thick should always be welded with DC straight polarity using 0407GL .040 7 argon gas. -

Basic Facts About Stainless Steel
What is stainless steel ? Stainless steel is the generic name for a number of different steels used primarily for their resistance to corrosion. The one key element they all share is a certain minimum percentage (by mass) of chromium: 10.5%. Although other elements, particularly nickel and molybdenum, are added to improve corrosion resistance, chromium is always the deciding factor. The vast majority of steel produced in the world is carbon and alloy steel, with the more expensive stainless steels representing a small, but valuable niche market. What causes corrosion? Only metals such as gold and platinum are found naturally in a pure form - normal metals only exist in nature combined with other elements. Corrosion is therefore a natural phenomena, as nature seeks to combine together elements which man has produced in a pure form for his own use. Iron occurs naturally as iron ore. Pure iron is therefore unstable and wants to "rust"; that is, to combine with oxygen in the presence of water. Trains blown up in the Arabian desert in the First World War are still almost intact because of the dry rainless conditions. Iron ships sunk at very great depths rust at a very slow rate because of the low oxygen content of the sea water . The same ships wrecked on the beach, covered at high tide and exposed at low tide, would rust very rapidly. For most of the Iron Age, which began about 1000 BC, cast and wrought iron was used; iron with a high carbon content and various unrefined impurities. Steel did not begin to be produced in large quantities until the nineteenth century. -

American Bronze Co., Chicago
/American j^ronze C^- 41 Vai| pUreii S^ree^, - cHICAGO, ILLS- Co.i Detroit. ite arid <r\ntique t^ponze JVlonumer|tal Wopk.. Salesroom: ART FOUNDRY. II CHICAGO. H. N. HIBBARD, Pres't. PAUL CORNELL, Vice-Pres't, JAS, STEWART, Treas, R J, HAIGHT, Sec'y, THE HEMRT FRAXCIS du POJ^ WIXrERTHUR MUSEUM LIBRARIES Digitized by the Internet Archive in 2010 with funding from Lyrasis IVIembers and Sloan Foundation http://www.archive.org/details/whiteantiquebronOOamer Tillr rr|ore prominent cemeteries in this country are noW arranged or) what is l<;noWn as the LfavVn I'lan, which gives the grounds a park-like appearaqce, n]ore in harmony With the impulse of our natures to make tl^ese lastresting places beautiful; in striking contrast to the gloomy burying places of olden times. pences, hedges, curbiqg aqd enclosures of all kinds are prol]ibited and tl^e money formerly expended for such fittings is invested in a central monu- ment, theicby enabling the lot oWner to purchase a better niemorial tl]an could otherwise haVe been afforded. (Corner posts are barely Visible aboVe the surface of the ground, and markers at the head of graVes are allowed ' only a feW inches higher, thus preserving the beautiful landscape effect. JViaiiy of tl]e n"ionum|ents novV being erected, and several that are illustrated in this pamphlet, bear feW, if aiw, fcmiily records, thus illustrating the growing desire to provide a fan]ily resting place and an enduring n-jonu- rqent. Without deferring it until there Fjos been a death in the family, as has been the custom in tlie past. -

Piece Mold, Lost Wax & Composite Casting Techniques of The
Piece Mold, Lost Wax & Composite Casting Techniques of the Chinese Bronze Age Behzad Bavarian and Lisa Reiner Dept. of MSEM College of Engineering and Computer Science September 2006 Table of Contents Abstract Approximate timeline 1 Introduction 2 Bronze Transition from Clay 4 Elemental Analysis of Bronze Alloys 4 Melting Temperature 7 Casting Methods 8 Casting Molds 14 Casting Flaws 21 Lost Wax Method 25 Sanxingdui 28 Environmental Effects on Surface Appearance 32 Conclusion 35 References 36 China can claim a history rich in over 5,000 years of artistic, philosophical and political advancement. As well, it is birthplace to one of the world's oldest and most complex civilizations. By 1100 BC, a high level of artistic and technical skill in bronze casting had been achieved by the Chinese. Bronze artifacts initially were copies of clay objects, but soon evolved into shapes invoking bronze material characteristics. Essentially, the bronze alloys represented in the copper-tin-lead ternary diagram are not easily hot or cold worked and are difficult to shape by hammering, the most common techniques used by the ancient Europeans and Middle Easterners. This did not deter the Chinese, however, for they had demonstrated technical proficiency with hard, thin walled ceramics by the end of the Neolithic period and were able to use these skills to develop a most unusual casting method called the piece mold process. Advances in ceramic technology played an influential role in the progress of Chinese bronze casting where the piece mold process was more of a technological extension than a distinct innovation. Certainly, the long and specialized experience in handling clay was required to form the delicate inscriptions, to properly fit the molds together and to prevent them from cracking during the pour. -

Sculptors' Jewellery Offers an Experience of Sculpture at Quite the Opposite End of the Scale
SCULPTORS’ JEWELLERY PANGOLIN LONDON FOREWORD The gift of a piece of jewellery seems to have taken a special role in human ritual since Man’s earliest existence. In the most ancient of tombs, archaeologists invariably excavate metal or stone objects which seem to have been designed to be worn on the body. Despite the tiny scale of these precious objects, their ubiquity in all cultures would indicate that jewellery has always held great significance.Gold, silver, bronze, precious stone, ceramic and natural objects have been fashioned for millennia to decorate, embellish and adorn the human body. Jewellery has been worn as a signifier of prowess, status and wealth as well as a symbol of belonging or allegiance. Perhaps its most enduring function is as a token of love and it is mostly in this vein that a sculptor’s jewellery is made: a symbol of affection for a spouse, loved one or close friend. Over a period of several years, through trying my own hand at making rings, I have become aware of and fascinated by the jewellery of sculptors. This in turn has opened my eyes to the huge diversity of what are in effect, wearable, miniature sculptures. The materials used are generally precious in nature and the intimacy of being worn on the body marries well with the miniaturisation of form. For this exhibition Pangolin London has been fortunate in being able to collate a very special selection of works, ranging from the historical to the contemporary. To complement this, we have also actively commissioned a series of exciting new pieces from a broad spectrum of artists working today. -

The Stainless Steel Family
The Stainless Steel Family A short description of the various grades of stainless steel and how they fit into distinct metallurgical families. It has been written primarily from a European perspective and may not fully reflect the practice in other regions. Stainless steel is the term used to describe an extremely versatile family of engineering materials, which are selected primarily for their corrosion and heat resistant properties. All stainless steels contain principally iron and a minimum of 10.5% chromium. At this level, chromium reacts with oxygen and moisture in the environment to form a protective, adherent and coherent, oxide film that envelops the entire surface of the material. This oxide film (known as the passive or boundary layer) is very thin (2-3 namometres). [1nanometre = 10-9 m]. The passive layer on stainless steels exhibits a truly remarkable property: when damaged (e.g. abraded), it self-repairs as chromium in the steel reacts rapidly with oxygen and moisture in the environment to reform the oxide layer. Increasing the chromium content beyond the minimum of 10.5% confers still greater corrosion resistance. Corrosion resistance may be further improved, and a wide range of properties provided, by the addition of 8% or more nickel. The addition of molybdenum further increases corrosion resistance (in particular, resistance to pitting corrosion), while nitrogen increases mechanical strength and enhances resistance to pitting. Categories of Stainless Steels The stainless steel family tree has several branches, which may be differentiated in a variety of ways e.g. in terms of their areas of application, by the alloying elements used in their production, or, perhaps the most accurate way, by the metallurgical phases present in their microscopic structures: . -
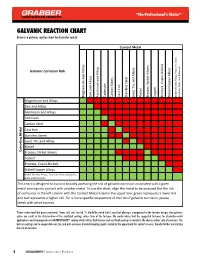
GALVANIC REACTION CHART Below Is a Galvanic Reaction Chart for Dissimilar Metals
GALVANIC REACTION CHART Below is a galvanic reaction chart for dissimilar metals. Contact Metal Silver, Silver, Galvanic Corrosion Risk Alloys Alloys Platinum Alloys Alloys, Titanium, and Alloys Steels Gold, and Iron Cast Stainless Steel Lead, Tin, and Magnesium and Zinc and Cadmium Carbon Nickel Brasses, Nickel-Silvers Copper Bronzes, Cupro-Nickels Nickel Copper Alloys Aluminum Graphite, Nickel-Chrome Magnesium and Alloys Zinc and Alloys Aluminum and Alloys Cadmium Carbon Steel Cast Iron Metal Stainless Steels Lead, Tin, and Alloys Nickel Corroding Brasses, Nickel-Silvers Copper Bronzes, Cupro-Nickels Nickel Copper Alloys Nickel-Chrome Alloys, Titanium, Silver, Graphite, Gold, and Platinum This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming into contact with another metal. To use the chart, align the metal to be assessed (for the risk of corrosion) in the left column with the Contact Metal listed in the upper row; green represents a lower risk and red represents a higher risk. For a more specific assessment of the risk of galvanic corrosion, please check with other sources. Please understand that green represents "lower risk" not "no risk." It should be noted that if sacrificial plating is incorporated in the fastener design, then galvanic action can result in the deterioration of the sacrificial coating, rather than of the fastener. We would advise that the suggested fasteners for dissimilar-metal applications would incorporate our GRABBERGARD® coating which utilizes both barrier and sacrificial coatings to minimize the chance and/or rate of corrosion. The barrier coating used to encapsulate our zinc and anti-corrosion chemical bonding agents minimize the opportunity for contact to occur, thereby further minimizing the risk of corrosion.