This PDF Was Created from the British Library's Microfilm Copy of The
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Picrotoxin-Like Channel Blockers of GABAA Receptors
COMMENTARY Picrotoxin-like channel blockers of GABAA receptors Richard W. Olsen* Department of Molecular and Medical Pharmacology, Geffen School of Medicine, University of California, Los Angeles, CA 90095-1735 icrotoxin (PTX) is the prototypic vous system. Instead of an acetylcholine antagonist of GABAA receptors (ACh) target, the cage convulsants are (GABARs), the primary media- noncompetitive GABAR antagonists act- tors of inhibitory neurotransmis- ing at the PTX site: they inhibit GABAR Psion (rapid and tonic) in the nervous currents and synapses in mammalian neu- system. Picrotoxinin (Fig. 1A), the active rons and inhibit [3H]dihydropicrotoxinin ingredient in this plant convulsant, struc- binding to GABAR sites in brain mem- turally does not resemble GABA, a sim- branes (7, 9). A potent example, t-butyl ple, small amino acid, but it is a polycylic bicyclophosphorothionate, is a major re- compound with no nitrogen atom. The search tool used to assay GABARs by compound somehow prevents ion flow radio-ligand binding (10). through the chloride channel activated by This drug target appears to be the site GABA in the GABAR, a member of the of action of the experimental convulsant cys-loop, ligand-gated ion channel super- pentylenetetrazol (1, 4) and numerous family. Unlike the competitive GABAR polychlorinated hydrocarbon insecticides, antagonist bicuculline, PTX is clearly a including dieldrin, lindane, and fipronil, noncompetitive antagonist (NCA), acting compounds that have been applied in not at the GABA recognition site but per- huge amounts to the environment with haps within the ion channel. Thus PTX major agricultural economic impact (2). ͞ appears to be an excellent example of al- Some of the other potent toxicants insec- losteric modulation, which is extremely ticides were also radiolabeled and used to important in protein function in general characterize receptor action, allowing and especially for GABAR (1). -
Qrno. 1 2 3 4 5 6 7 1 CP 2903 77 100 0 Cfcl3
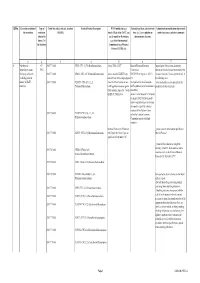
QRNo. General description of Type of Tariff line code(s) affected, based on Detailed Product Description WTO Justification (e.g. National legal basis and entry into Administration, modification of previously the restriction restriction HS(2012) Article XX(g) of the GATT, etc.) force (i.e. Law, regulation or notified measures, and other comments (Symbol in and Grounds for Restriction, administrative decision) Annex 2 of e.g., Other International the Decision) Commitments (e.g. Montreal Protocol, CITES, etc) 12 3 4 5 6 7 1 Prohibition to CP 2903 77 100 0 CFCl3 (CFC-11) Trichlorofluoromethane Article XX(h) GATT Board of Eurasian Economic Import/export of these ozone destroying import/export ozone CP-X Commission substances from/to the customs territory of the destroying substances 2903 77 200 0 CF2Cl2 (CFC-12) Dichlorodifluoromethane Article 46 of the EAEU Treaty DECISION on August 16, 2012 N Eurasian Economic Union is permitted only in (excluding goods in dated 29 may 2014 and paragraphs 134 the following cases: transit) (all EAEU 2903 77 300 0 C2F3Cl3 (CFC-113) 1,1,2- 4 and 37 of the Protocol on non- On legal acts in the field of non- _to be used solely as a raw material for the countries) Trichlorotrifluoroethane tariff regulation measures against tariff regulation (as last amended at 2 production of other chemicals; third countries Annex No. 7 to the June 2016) EAEU of 29 May 2014 Annex 1 to the Decision N 134 dated 16 August 2012 Unit list of goods subject to prohibitions or restrictions on import or export by countries- members of the -

Neurotransmitters-Drugs Andbrain Function.Pdf
Neurotransmitters, Drugs and Brain Function. Edited by Roy Webster Copyright & 2001 John Wiley & Sons Ltd ISBN: Hardback 0-471-97819-1 Paperback 0-471-98586-4 Electronic 0-470-84657-7 Neurotransmitters, Drugs and Brain Function Neurotransmitters, Drugs and Brain Function. Edited by Roy Webster Copyright & 2001 John Wiley & Sons Ltd ISBN: Hardback 0-471-97819-1 Paperback 0-471-98586-4 Electronic 0-470-84657-7 Neurotransmitters, Drugs and Brain Function Edited by R. A. Webster Department of Pharmacology, University College London, UK JOHN WILEY & SONS, LTD Chichester Á New York Á Weinheim Á Brisbane Á Singapore Á Toronto Neurotransmitters, Drugs and Brain Function. Edited by Roy Webster Copyright & 2001 John Wiley & Sons Ltd ISBN: Hardback 0-471-97819-1 Paperback 0-471-98586-4 Electronic 0-470-84657-7 Copyright # 2001 by John Wiley & Sons Ltd. Bans Lane, Chichester, West Sussex PO19 1UD, UK National 01243 779777 International ++44) 1243 779777 e-mail +for orders and customer service enquiries): [email protected] Visit our Home Page on: http://www.wiley.co.uk or http://www.wiley.com All Rights Reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted, in any form or by any means, electronic, mechanical, photocopying, recording, scanning or otherwise, except under the terms of the Copyright, Designs and Patents Act 1988 or under the terms of a licence issued by the Copyright Licensing Agency Ltd, 90 Tottenham Court Road, London W1P0LP,UK, without the permission in writing of the publisher. Other Wiley Editorial Oces John Wiley & Sons, Inc., 605 Third Avenue, New York, NY 10158-0012, USA WILEY-VCH Verlag GmbH, Pappelallee 3, D-69469 Weinheim, Germany John Wiley & Sons Australia, Ltd. -

Fluorides in the Environment
Color profile: Disabled Composite 150 lpi at 45 degrees Fluorides in the Environment A4662 - Weinstein - Vouchers - VP10 #K.prn 1 Z:\Customer\CABI\A4642 - Weinstein\A4662 - Weinstein - Vouchers - VP10 #K.vp Monday, November 10, 2003 3:35:50 PM Color profile: Disabled Composite 150 lpi at 45 degrees A4662 - Weinstein - Vouchers - VP10 #K.prn 2 Z:\Customer\CABI\A4642 - Weinstein\A4662 - Weinstein - Vouchers - VP10 #K.vp Monday, November 10, 2003 3:35:50 PM Color profile: Disabled Composite 150 lpi at 45 degrees Fluorides in the Environment Effects on Plants and Animals Professor L.H. Weinstein Boyce Thompson Institute for Plant Research Tower Road Ithaca NY 14853 USA and Professor A. Davison School of Biology Ridley Building University of Newcastle Newcastle upon Tyne NE1 7RU UK CABI Publishing A4662 - Weinstein - Vouchers - VP10 #K.prn 3 Z:\Customer\CABI\A4642 - Weinstein\A4662 - Weinstein - Vouchers - VP10 #K.vp Monday, November 10, 2003 3:35:51 PM Color profile: Disabled Composite 150 lpi at 45 degrees CABI Publishing is a division of CAB International CABI Publishing CABI Publishing CAB International 875 Massachusetts Avenue Wallingford 7th Floor Oxon OX10 8DE Cambridge, MA 02139 UK USA Tel: +44 (0)1491 832111 Tel: +1 617 395 4056 Fax: +44 (0)1491 833508 Fax: +1 617 354 6875 E-mail: [email protected] E-mail: [email protected] Web site: www.cabi-publishing.org ©L.H. Weinstein and A. Davison 2004. All rights reserved. No part of this publication may be reproduced in any form or by any means, electronically, mechanically, by photocopying, recording or otherwise, without the prior permission of the copyright owners. -

GABA Receptors
D Reviews • BIOTREND Reviews • BIOTREND Reviews • BIOTREND Reviews • BIOTREND Reviews Review No.7 / 1-2011 GABA receptors Wolfgang Froestl , CNS & Chemistry Expert, AC Immune SA, PSE Building B - EPFL, CH-1015 Lausanne, Phone: +41 21 693 91 43, FAX: +41 21 693 91 20, E-mail: [email protected] GABA Activation of the GABA A receptor leads to an influx of chloride GABA ( -aminobutyric acid; Figure 1) is the most important and ions and to a hyperpolarization of the membrane. 16 subunits with γ most abundant inhibitory neurotransmitter in the mammalian molecular weights between 50 and 65 kD have been identified brain 1,2 , where it was first discovered in 1950 3-5 . It is a small achiral so far, 6 subunits, 3 subunits, 3 subunits, and the , , α β γ δ ε θ molecule with molecular weight of 103 g/mol and high water solu - and subunits 8,9 . π bility. At 25°C one gram of water can dissolve 1.3 grams of GABA. 2 Such a hydrophilic molecule (log P = -2.13, PSA = 63.3 Å ) cannot In the meantime all GABA A receptor binding sites have been eluci - cross the blood brain barrier. It is produced in the brain by decarb- dated in great detail. The GABA site is located at the interface oxylation of L-glutamic acid by the enzyme glutamic acid decarb- between and subunits. Benzodiazepines interact with subunit α β oxylase (GAD, EC 4.1.1.15). It is a neutral amino acid with pK = combinations ( ) ( ) , which is the most abundant combi - 1 α1 2 β2 2 γ2 4.23 and pK = 10.43. -

Picrotoxinin and Diazepam Bind to Two Distinct Proteins: Further Evidence That Pentobarbital May Act at the Picrotoxinin Site1
0270~6474/81/0109-1036$02.00/O The Journal of Neuroscience Copyright 0 Society for Neuroscience Vol. 1, No. 9, pp. 1036-1042 Printed in U.S.A. September 1981 PICROTOXININ AND DIAZEPAM BIND TO TWO DISTINCT PROTEINS: FURTHER EVIDENCE THAT PENTOBARBITAL MAY ACT AT THE PICROTOXININ SITE1 WILLIAM C. DAVIS AND MAHARAJ K. TICKU2 Division of Molecular Pharmacology, Department of Pharmacology, The University of Texas Health Science Center at San Antonio, San Antonio. Texas 78284 Abstract a-[3H]Dihydropicrotoxinin (DHP) and [3H]diazepam binding proteins were solubilized from rat brain membranes with 1% Lubrol-Px. Gel filtration of the Lubrol-solubilized fraction revealed that [3H]DHP and [3H]diazepam bind to two distinct peaks with apparent molecular weights of 185,000 and 61,000, respectively. The signal-to-noise ratio of [3H]DHP binding to 185,000-dalton fractions was improved significantly. [3H]DHP bound to the 185,000-dalton fraction with two binding constants. Muscimol and pentobarbital, while enhancing [3H]diazepam binding to membrane and crude Lubrol-solubilized fractions, failed to enhance [3H]diazepam binding to the 61,000-dalton fraction. Pentobarbital inhibited the binding of [3H]DHP to the 185,000-dalton fraction with an IC5” value of 60 + 12 PM. The binding of [3H]DHP also was inhibited by several depressant and convulsant drugs which affect y-aminobutyric acid (GABA)-mediated transmission. These results provide strong evidence that picrotoxinin and diazepam bind to two distinct proteins and that pentobarbital may act at the picrotoxinin-sensitive site of the benzodiazepine . GABA receptor. ionophore complex. The y-aminobutyric acid (GABA) receptor system ap- Olsen, 1978, 1979; Olsen et al., 1979). -

Cell Surface Mobility of GABAB Receptors Saad Bin
Cell surface mobility of GABAB receptors Saad Bin Hannan September 2011 A thesis submitted in fulfilment of the requirements for the degree of Doctor of Philosophy of the University College London Department of Neuroscience, Physiology, and Pharmacology University College London Gower Street London WC1E 6BT UK Declaration ii ‘I, Saad Hannan confirm that the work presented in this thesis is my own. Where information has been derived from other sources, I confirm that this has been indicated in the thesis.' ____________________ Saad Hannan September 2011 To Ammu, Abbu, Polu Abstract ivi Abstract Type-B γ-aminobutyric acid receptors (GABABRs) are important for mediating slow inhibition in the central nervous system and the kinetics of their internalisation and lateral mobility will be a major determinant of their signalling efficacy. Functional GABABRs require R1 and R2 subunit co-assembly, but how heterodimerisation affects the trafficking kinetics of GABABRs is unknown. Here, an α- bungarotoxin binding site (BBS) was inserted into the N-terminus of R2 to monitor receptor mobility in live cells. GABABRs are internalised via clathrin- and dynamin- dependent pathways and recruited to endosomes. By mutating the BBS, a new technique was developed to differentially track R1a and R2 simultaneously, revealing the subunits internalise as heteromers and that R2 dominantly-affects constitutive internalisation of GABABRs. Notably, the internalisation profile of R1aR2 heteromers, but not R1a homomers devoid of their ER retention motif (R1ASA), is similar to R2 homomers in heterologous systems. The internalisation of R1aASA was slowed to that of R2 by mutating a di-leucine motif in the R1 C-terminus, indicating a new role for heterodimerisation, whereby R2 subunits slow the internalization of surface GABABRs. -

Toxicological and Pharmacological Profile of Amanita Muscaria (L.) Lam
Pharmacia 67(4): 317–323 DOI 10.3897/pharmacia.67.e56112 Review Article Toxicological and pharmacological profile of Amanita muscaria (L.) Lam. – a new rising opportunity for biomedicine Maria Voynova1, Aleksandar Shkondrov2, Magdalena Kondeva-Burdina1, Ilina Krasteva2 1 Laboratory of Drug metabolism and drug toxicity, Department “Pharmacology, Pharmacotherapy and Toxicology”, Faculty of Pharmacy, Medical University of Sofia, Bulgaria 2 Department of Pharmacognosy, Faculty of Pharmacy, Medical University of Sofia, Bulgaria Corresponding author: Magdalena Kondeva-Burdina ([email protected]) Received 2 July 2020 ♦ Accepted 19 August 2020 ♦ Published 26 November 2020 Citation: Voynova M, Shkondrov A, Kondeva-Burdina M, Krasteva I (2020) Toxicological and pharmacological profile of Amanita muscaria (L.) Lam. – a new rising opportunity for biomedicine. Pharmacia 67(4): 317–323. https://doi.org/10.3897/pharmacia.67. e56112 Abstract Amanita muscaria, commonly known as fly agaric, is a basidiomycete. Its main psychoactive constituents are ibotenic acid and mus- cimol, both involved in ‘pantherina-muscaria’ poisoning syndrome. The rising pharmacological and toxicological interest based on lots of contradictive opinions concerning the use of Amanita muscaria extracts’ neuroprotective role against some neurodegenerative diseases such as Parkinson’s and Alzheimer’s, its potent role in the treatment of cerebral ischaemia and other socially significant health conditions gave the basis for this review. Facts about Amanita muscaria’s morphology, chemical content, toxicological and pharmacological characteristics and usage from ancient times to present-day’s opportunities in modern medicine are presented. Keywords Amanita muscaria, muscimol, ibotenic acid Introduction rica, the genus had an ancestral origin in the Siberian-Be- ringian region in the Tertiary period (Geml et al. -

Isoguvacine Hydrochloride | Medchemexpress
Inhibitors Product Data Sheet Isoguvacine hydrochloride • Agonists Cat. No.: HY-100810 CAS No.: 68547-97-7 Molecular Formula: C₆H₁₀ClNO₂ • Molecular Weight: 163.6 Screening Libraries Target: GABA Receptor Pathway: Membrane Transporter/Ion Channel; Neuronal Signaling Storage: Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month SOLVENT & SOLUBILITY In Vitro H2O : ≥ 100 mg/mL (611.25 mM) DMSO : 25 mg/mL (152.81 mM; Need ultrasonic) * "≥" means soluble, but saturation unknown. Mass Solvent 1 mg 5 mg 10 mg Concentration Preparing 1 mM 6.1125 mL 30.5623 mL 61.1247 mL Stock Solutions 5 mM 1.2225 mL 6.1125 mL 12.2249 mL 10 mM 0.6112 mL 3.0562 mL 6.1125 mL Please refer to the solubility information to select the appropriate solvent. In Vivo 1. Add each solvent one by one: 10% DMSO >> 40% PEG300 >> 5% Tween-80 >> 45% saline Solubility: ≥ 2.08 mg/mL (12.71 mM); Clear solution 2. Add each solvent one by one: 10% DMSO >> 90% (20% SBE-β-CD in saline) Solubility: ≥ 2.08 mg/mL (12.71 mM); Clear solution 3. Add each solvent one by one: 10% DMSO >> 90% corn oil Solubility: ≥ 2.08 mg/mL (12.71 mM); Clear solution BIOLOGICAL ACTIVITY Description Isoguvacine hydrochloride is a GABA receptor agonist. IC₅₀ & Target GABA[1] In Vitro Isoguvacine binds to a mouse forebrain synaptic membrane preparation. The specific binding is displaceable by GABA, Page 1 of 2 www.MedChemExpress.com muscimol and bicuculline but not by picrotoxin or diaminobutyric acid. -

Amanita Muscaria: Ecology, Chemistry, Myths
Entry Amanita muscaria: Ecology, Chemistry, Myths Quentin Carboué * and Michel Lopez URD Agro-Biotechnologies Industrielles (ABI), CEBB, AgroParisTech, 51110 Pomacle, France; [email protected] * Correspondence: [email protected] Definition: Amanita muscaria is the most emblematic mushroom in the popular representation. It is an ectomycorrhizal fungus endemic to the cold ecosystems of the northern hemisphere. The basidiocarp contains isoxazoles compounds that have specific actions on the central nervous system, including hallucinations. For this reason, it is considered an important entheogenic mushroom in different cultures whose remnants are still visible in some modern-day European traditions. In Siberian civilizations, it has been consumed for religious and recreational purposes for millennia, as it was the only inebriant in this region. Keywords: Amanita muscaria; ibotenic acid; muscimol; muscarine; ethnomycology 1. Introduction Thanks to its peculiar red cap with white spots, Amanita muscaria (L.) Lam. is the most iconic mushroom in modern-day popular culture. In many languages, its vernacular names are fly agaric and fly amanita. Indeed, steeped in a bowl of milk, it was used to Citation: Carboué, Q.; Lopez, M. catch flies in houses for centuries in Europe due to its ability to attract and intoxicate flies. Amanita muscaria: Ecology, Chemistry, Although considered poisonous when ingested fresh, this mushroom has been consumed Myths. Encyclopedia 2021, 1, 905–914. as edible in many different places, such as Italy and Mexico [1]. Many traditional recipes https://doi.org/10.3390/ involving boiling the mushroom—the water containing most of the water-soluble toxic encyclopedia1030069 compounds is then discarded—are available. In Japan, the mushroom is dried, soaked in brine for 12 weeks, and rinsed in successive washings before being eaten [2]. -

Gabaergic Signaling Linked to Autophagy Enhances Host Protection Against Intracellular Bacterial Infections
ARTICLE DOI: 10.1038/s41467-018-06487-5 OPEN GABAergic signaling linked to autophagy enhances host protection against intracellular bacterial infections Jin Kyung Kim1,2,3, Yi Sak Kim1,2,3, Hye-Mi Lee1,3, Hyo Sun Jin4, Chiranjivi Neupane 2,5, Sup Kim1,2,3, Sang-Hee Lee6, Jung-Joon Min7, Miwa Sasai8, Jae-Ho Jeong 9,10, Seong-Kyu Choe11, Jin-Man Kim12, Masahiro Yamamoto8, Hyon E. Choy 9,10, Jin Bong Park 2,5 & Eun-Kyeong Jo1,2,3 1234567890():,; Gamma-aminobutyric acid (GABA) is the principal inhibitory neurotransmitter in the brain; however, the roles of GABA in antimicrobial host defenses are largely unknown. Here we demonstrate that GABAergic activation enhances antimicrobial responses against intracel- lular bacterial infection. Intracellular bacterial infection decreases GABA levels in vitro in macrophages and in vivo in sera. Treatment of macrophages with GABA or GABAergic drugs promotes autophagy activation, enhances phagosomal maturation and antimicrobial responses against mycobacterial infection. In macrophages, the GABAergic defense is mediated via macrophage type A GABA receptor (GABAAR), intracellular calcium release, and the GABA type A receptor-associated protein-like 1 (GABARAPL1; an Atg8 homolog). Finally, GABAergic inhibition increases bacterial loads in mice and zebrafish in vivo, sug- gesting that the GABAergic defense plays an essential function in metazoan host defenses. Our study identified a previously unappreciated role for GABAergic signaling in linking antibacterial autophagy to enhance host innate defense against intracellular bacterial infection. 1 Department of Microbiology, Chungnam National University School of Medicine, Daejeon 35015, Korea. 2 Department of Medical Science, Chungnam National University School of Medicine, Daejeon 35015, Korea. -

Preparation of N-‐(Boc)-‐Allylglycine Methyl Ester
Preparation of N-(Boc)-Allylglycine Methyl Ester Using a Zinc-mediated, Palladium-catalyzed Cross-coupling Reaction N. D. Prasad Atmuri and William D. Lubell* Département de Chimie, Université de Montréal, P.O. Box 6128, Station Centre-ville, Montréal, QC H3C 3J7, Canada Checked by Jean-Nicolas Desrosiers, Nizar Haddad, and Chris H. Senanayake Iodine (1.3 equiv), Ph3P (1.3 equiv), OH Imidazole (1.3 equiv) I A. DCM, 0 °C - rt BocHN CO2Me 82% BocHN CO2Me 1 2 1. Zn (6 equiv), DMF Br Br (0.6 equiv), 60 °C I TMSCl (0.2 equiv), rt - 35 °C B. BocHN CO Me BocHN CO2Me 2 2. Br (1 M in THF) 2 3 Pd2(dba)3 (0.02 equiv) P(o-tol)3 (0.1 equiv) –78 °C - rt, 12 h 65% Procedure A. tert-Butyl (R)-1-(methoxycarbonyl)-2-iodoethylcarbamate (2). An oven- dried 1000-mL, three-necked, round-bottomed flask containing an egg- shaped Teflon®-coated magnetic stir bar (7 cm long) is equipped with a rubber septum with a thermometer, a 125 mL addition funnel and an argon inlet adaptor. The apparatus is purged with argon (Note 1). Keeping a Org. Synth. 2015, 92, 103-116 103 Published on the Web 4/6/2015 DOI: 10.15227/orgsyn.092.0103 © 2015 Organic Syntheses, Inc. positive flow of argon, the septum is removed temporarily and the flask is charged with triphenylphosphine (Note 2) (32.66 g, 124.5 mmol, 1.3 equiv) and 400 mL of dichloromethane (Note 3). The solution is stirred at room temperature and imidazole (8.47 g, 124.5 mmol, 1.3 equiv) (Note 2) is added in one portion.