Tips for Managing Treatment-Related Rash and Dry Skin
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Women in Business Awards Luncheon at the Hotel Irvine, Where Aston Martin Americas President Laura Schwab Delivered the Keynote Address
10.5.20 SR_WIB.qxp_Layout 1 10/2/20 12:14 PM Page 29 WOMEN IN BUSINESS NOMINEES START ON PAGE B-60 INSIDE 2019 WINNERS GO BIG IN IRVINE, LAND NEW PARTNERS, INVESTMENTS PAGE 30 PRESENTED BY DIAMOND SPONSOR PLATINUM SPONSORS GOLD SPONSOR SILVER SPONSORS 10.5.20 SR_WIB.qxp_Layout 1 10/2/20 1:36 PM Page 30 30 ORANGE COUNTY BUSINESS JOURNAL www.ocbj.com OCTOBER 5, 2020 Winning Execs Don’t Rest on Their Laurels $1B Cancer Center Underway; Military Wins; Spanish Drug Investment Orange County’s business community last year celebrated the Business Journal’s 25th annual Women in Business Awards luncheon at the Hotel Irvine, where Aston Martin Americas President Laura Schwab delivered the keynote address. The winners, selected from 200 nominees, have not been resting on their laurels, even in the era of the coronavirus. Here are updates on what the five winners have been doing. —Peter J. Brennan Avatar Partners City of Hope Shortly after Marlo Brooke won the Busi- (AR) quality assurance solution for the U.S. As president of the City of Hope Orange employees down from Duarte. Area univer- ness Journal’s award for co-founding Hunt- Navy for aircraft wiring maintenance for the County, Annette Walker is orchestrating a sities could partner with City of Hope. ington Beach-based Avatar Partners Inc., Naval Air Systems Command’s Boeing V- $1 billion project to build one of the biggest, While the larger campus near the Orange she was accepted into the Forbes Technol- 22 Osprey aircraft. and scientifically advanced, cancer research County Great Park is being built, Walker in ogy Council, an invitation-only community Then the Air Force is using Avatar’s solu- centers in the world. -

Concurrent Beau Lines, Onychomadesis, and Retronychia Following Scurvy
CASE REPORT Concurrent Beau Lines, Onychomadesis, and Retronychia Following Scurvy Dayoung Ko, BS; Shari R. Lipner, MD, PhD the proximal nail plate from the distal nail plate leading to shedding of the nail. It occurs due to a complete growth PRACTICE POINTS arrest in the nail matrix and is thought to be on a con- • Beau lines, onychomadesis, and retronychia are nail tinuum with Beau lines. The etiologies of these 2 condi- conditions with distinct clinical findings. tions overlap and include trauma, inflammatory diseases, • Beau lines and onychomadesis may be seen 1-5 concurrently following trauma, inflammatory dis- systemic illnesses, hereditary conditions, and infections. eases, systemic illnesses, hereditary conditions, In almost all cases of both conditions, normal nail plate and infections. production ensues upon identification and removal of the 3,4,6 • Retronychia shares a common pathophysiology inciting agent or recuperation from the causal illness. with Beau lines and onychomadesis, and all reflect Beau lines will move distally as the nail grows out and slowing or cessation of nail plate production. can be clipped. In onychomadesis, the affected nails will be shed with time. Resolution of these nail defects can be estimated from average nail growth rates (1 mm/mo for fingernails and 2–3 mm/mo for toenails).7 Beau lines, onychomadesis, and retronychia are nail conditions with Retronychia is defined as a proximal ingrowing of their own characteristic clinical findings. It has been hypothesized the nail plate into the ventral surface of the proximal nail that these 3 disorders may share a common pathophysiologic fold.4,6 It is thought to occur via vertical progression of mechanism of slowing and/or halting nail plate production at the the nail plate into the proximal nail fold, repetitive nail nail matrix. -

Truncal Rashes Stan L
Healthy Baby Practical advice for treating newborns and toddlers. Getting Truculent with Truncal Rashes Stan L. Block, MD, FAAP A B C All images courtesy of Stan L. Block, MD, FAAP. Figure 1. Afebrile 22-month-old white male presents to your office with this slowly spreading, somewhat generalized, and refractory truncal rash for the past 4 weeks. It initially started on the right side of his trunk (A) and later extended down his right upper thigh (B). The rash has now spread to the contralateral side on his back (C), and is most confluent and thickest over his right lateral ribs. n a daily basis, we pediatricians would not be readily able to identify this rash initially began on the right side of his encounter a multitude of rashes relatively newly described truncal rash trunk (see Figure 1A) and then extended O of varied appearance in children shown in some of the following cases. distally down to his right upper thigh (see of all ages. Most of us gently-seasoned As is typical, certain clues are critical, Figure 1B). Although the rash is now dis- clinicians have seen nearly all versions including the child’s age, the duration tributed over most of his back (see Figure of these “typical” rashes. Yet, I venture and the distribution of the rash. Several 1C), it is most confluent and most dense to guess that many practitioners, who of these rashes notably mimic more com- over his right lateral ribs. would be in good company with some of mon etiologies, as discussed in some of From Figure 1, you could speculate my quite erudite partners (whom I asked), the following cases. -

Johnson & Johnson Reports 2010 Second-Quarter Results
Johnson & Johnson Reports 2010 Second-Quarter Results: Sales of $15.3 Billion Increased 0.6% Versus 2009 Second-Quarter; EPS was $1.23 Excluding Special Items, 2010 Second-Quarter EPS was $1.21, an increase of 5.2%* NEW BRUNSWICK, N.J., July 20, 2010 /PRNewswire via COMTEX News Network/ -- Johnson & Johnson (NYSE: JNJ) today announced sales of $15.3 billion for the second quarter of 2010, an increase of 0.6% as compared to the second quarter of 2009. Operational results increased 0.1% and the positive impact of currency was 0.5%. Domestic sales declined 2.8%, while international sales increased 4.1%, reflecting operational growth of 3.0% and a positive currency impact of 1.1%. Net earnings and diluted earnings per share for the second quarter of 2010 were $3.4 billion and $1.23, respectively. Second- quarter 2010 net earnings included an after-tax gain of $67 million representing the net impact of litigation matters. Excluding this special item, net earnings for the current quarter were $3.4 billion and diluted earnings per share were $1.21, representing increases of 5.4% and 5.2%, respectively, as compared to the same period in 2009.* The Company updated its earnings guidance for full-year 2010 to $4.65 - $4.75 per share, which excludes the impact of special items. The Company's guidance now reflects the impact of the voluntary recalls announced earlier this year of certain over-the-counter medicines and the suspension of manufacturing at the McNeil Consumer Healthcare facility in Fort Washington, Pa., as well as unfavorable changes in foreign currency exchange rates. -
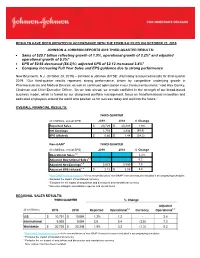
Sales of $20.7 Billion Reflecting Growth of 1.9%, Operational Growth of 3.2
RESULTS HAVE BEEN UPDATED IN ACCORDANCE WITH THE FORM 8-K FILED ON OCTOBER 23, 2019 JOHNSON & JOHNSON REPORTS 2019 THIRD-QUARTER RESULTS: • Sales of $20.7 billion reflecting growth of 1.9%, operational growth of 3.2%* and adjusted operational growth of 5.2%* • EPS of $0.66 decreased (54.2)%; adjusted EPS of $2.12 increased 3.4%* • Company increasing Full Year Sales and EPS guidance due to strong performance New Brunswick, N.J. (October 23, 2019) – Johnson & Johnson (NYSE: JNJ) today announced results for third-quarter 2019. “Our third-quarter results represent strong performance, driven by competitive underlying growth in Pharmaceuticals and Medical Devices, as well as continued optimization in our Consumer business,” said Alex Gorsky, Chairman and Chief Executive Officer. “As we look ahead, we remain confident in the strength of our broad-based business model, which is fueled by our disciplined portfolio management, focus on transformational innovation and dedicated employees around the world who position us for success today and well into the future.” OVERALL FINANCIAL RESULTS: THIRD QUARTER ($ in Millions, except EPS) 2019 2018 % Change Reported Sales $ 20,729 $ 20,348 1.9% Net Earnings 1,753 3,934 (55.4) EPS (diluted) $ 0.66 $ 1.44 (54.2) Non-GAAP* THIRD QUARTER ($ in Millions, except EPS) 2019 2018 % Change Operational Sales1,2 3.2% Adjusted Operational Sales1,3 5.2 Adjusted Net Earnings1,4 5,672 5,590 1.5 Adjusted EPS (diluted)1,4 $ 2.12 $ 2.05 3.4 1 Non-GAAP financial measure; refer to reconciliations of non-GAAP financial measures -

Department of Public Health 105 Cmr 720.000
105 CMR: DEPARTMENT OF PUBLIC HEALTH 105 CMR 720.000: LIST OF INTERCHANGEABLE DRUG PRODUCTS Section 720.001: Purpose 720.002: Citation 720.010: Scope and Application 720.020: Definitions Standards 720:040: Commission Review of Relevant Drug Products 720.050: List of Interchangeable Drug Products 720.060: Drug Products Excluded 720.070: Amendments to the Massachusetts List of Interchangeable Drugs Procedures for Amending List of Interchangeable Drug Products 720.080: Procedures for Amending the Massachusetts List of Interchangeable Drugs 720.081: Petition to Amend List of Interchangeable Drug Products 720.082: Commission Review of Petition 720.083: Notice of Public Comment Period 720.084: Commission Recommendation of Amendments to Department 720.090: Department Adoption of Amendments 720.100: Severability 720.200: Appendix A: List of Interchangeable Drugs 720.001: Purpose The purpose of 105 CMR 720.000 is to establish a drug formulary, or list of interchangeable drug products, for use by physicians, other practitioners, and pharmacists licensed to practice within the commonwealth, so that consumers of prescription drug products may realize cost savings by buying less expensive, safe drug products. 720.002: Citation 105 CMR 720.000 shall be known as the 105 CMR 720.000: Massachusetts List of Interchangeable Drug Products. 720.010: Scope and Application 105 CMR 720.000 establishes the list of interchangeable drug products from which a pharmacist must interchange a reasonably available less expensive drug product than that written, when a prescription written by a practitioner indicates "interchange". 105 CMR 720.000 also establishes criteria and procedures for inclusion of drug products on this list. -

Exhibitor & Sponsor Program
Exhibitor & Sponsor Program www.AtlanticDermConference.org @AtlanticDermADC The Philadelphia Dermatological Society and the Atlantic Derm Conference are pleased to acknowledge and express our sincere appreciation for our corporate sponsors, exhibitors, and commercial supporters. Disclosure of Commercial Support | This activity is supported by an educational donation provided by Amgen, an educational grant provided by Galderma Laboratories, LP, and an educational grant provided by Valeant Dermatology. SPONSORS DIAMOND PLATINUM GOLD SILVER Pharmaceuticals North America LLC www.AtlanticDermConference.org | Page 1 " !"!%! "%#%! !! !#! !% "!! #!!#%$ %"!"! ! !!!! " !%"!! '!!!&! ! "! ' !!! !!"!% "%# ! ( ! # ' % "& ## #!!#" #"$" Exhibitors Exhibitors 3Gen, Inc. | 31521 Rancho Viejo Road, #104, San Juan Capistrano, CA 92675 | 949.481.6384 | www.dermlite.com Table #15 ADDRESSING 3Gen manufactures the DermLite brand of skin imaging devices. THE TOUGHEST AbbVie | 1 N. Waukegan Road, North Chicago, IL 60064 | 847.937.7390 | abbvie.com | www.psoriasis.com Table #1 & 2 AbbVie is a global, research-based biopharmaceutical company which combines the focus of a leading-edge biotech with DERMATOLOGY the expertise and structure of a long-established pharmaceutical leader. AbbVie is committed to using unique approaches to innovation to develop and market advanced therapies that address some of the world’s most complex, serious diseases. CHALLENGES Actelion Pharmaceuticals US, Inc. | 5000 Shoreline Ct., South San Francisco, -

2019 SMDP Biotech Training Agenda & Scholar Bios
2019 SMDP Biotech 1-5 June PHILADELPHIA www.icpdprograms.org 2019 Scientist Mentoring & Diversity Program for biotechnology (SMDP Biotech) www.icpdprograms.org Training Session June 1-6, 2019 in Philadelphia, PA about the program who attends: the one-year career mentoring program pairs ethnically diverse post-baccalaureate students, graduate students and post-doctoral researchers with industry mentors who work at biotechnology and consumer healthcare companies. With their mentors, SMDP Biotech Scholars attend a 5-day training session to learn about career opportunities in industry and receive career coaching. SMDP Scholars and Mentors also attend The BIO International Convention. how to dress, what to bring: business attire with comfortable shoes. Scholars, bring 100 business cards and 10 copies of your resume. Mentors will need business cards too. Informal dinner where to go: SMDP training Scholars and Mentors you’re Host Hotel session day 1 already on the guest list, we Mentors you will arrange the bus leaves the leave J&J Consumer at 5:15pm your own accommodation host hotel at 6:30am Ladder 15, 1528 Sansom Street, Scholars a shared room Scholars you will be Philadelphia, PA 19102 reservation has already been introduced to your made for you at this location: Mentors at the Reading Terminal MarKet Philadelphia 201, 201 N. 17th training session The bus leaves from the SMDP J&J Consumer, Street, Philadelphia, PA 19103 training session at 11:30am 199 Grandview Road, Corner of North 12th Street “Celebration of Mentoring Skillman, NJ 08558 and Arch Street (South Bldg. SK1912) & Diversity” reception ($10 will be provided) (Saturday, June 1st at 6pm) SMDP training The BIO Intl Convention Scholars and Mentors you’re session day 2 registration information will be already on the guest list the bus leaves the provided during the SMDP The Pyramid Club, host hotel at 7am training session. -

Propylene Glycol
PROPYLENE GLYCOL Your patch test result indicates that you have a contact allergy to propylene glycol. This contact allergy may cause your skin to react when it is exposed to this substance although it may take several days for the symptoms to appear. Typical symptoms include redness, swelling, itching, and fluid-filled blisters. Where is propylene glycol found? Propylene glycol is used as a softening agent, preservative, humectants, and solvent in cosmetics, fragrances, topical medications, soaps and cleansers, hair care products, and deodorants. Propylene glycol is also found in oral treatments as well as many foods. It is also added during the manufacture of many industrial fluids, such as solvents, thinners, antifreeze, other de-icing fluids, desiccants, brake fluids, and polyester resins. How can you avoid contact with propylene glycol? Avoid products that list any of the following names in the ingredients: • Propylene glycol • 1,2-Dihydroxypropane • CASRN: 57-55-6 • Methylethyl glycol • 1,2-Propanediol • 2-Hydroxypropanol • Isopropylene glycol What are some products that may contain propylene glycol? Antiperspirants and Deodorants: • Old Spice High Endurance • Meguiars Vinyl/Rubber Cleaner/Condition • Adidas 24 Hour Deodorant Control Antiperspirant & Deodorant • Pennzoil Roadside Fix A Flat Tire Sealant & • Adidas 24 Hour Fragrance Clear Stick • Old Spice High Endurance Deodorant Flat Preventative Deodorant • Old Spice Red Zone Clear Gel • Rain-X De-Icer (Aerosol) • Adidas Action 3 Tech F • Old Spice Red Zone Deodorant Stick • Slime -

Drug Eruptions- When to Worry
3/17/2017 Drug reactions: Drug Eruptions‐ When to Worry 3 things you need to know 1. Type of drug reaction 2. Statistics: – Which drugs are most likely to cause that type of Lindy P. Fox, MD reaction? Associate Professor 3. Timing: Director, Hospital Consultation Service – How long after the drug started did the reaction Department of Dermatology University of California, San Francisco begin? [email protected] I have no conflicts of interest to disclose 1 Drug Eruptions: Common Causes of Cutaneous Drug Degrees of Severity Eruptions • Antibiotics Simple Complex • NSAIDs Morbilliform drug eruption Drug hypersensitivity reaction Stevens-Johnson syndrome •Sulfa (SJS) Toxic epidermal necrolysis (TEN) • Allopurinol Minimal systemic symptoms Systemic involvement • Anticonvulsants Potentially life threatening 1 3/17/2017 Morbilliform (Simple) Drug Eruption Morbilliform (Simple) Drug Eruption Per the drug chart, the most likely culprit is: Per the drug chart, the most likely culprit is: Day Day Day ‐> ‐8 ‐7 ‐6 ‐5 ‐4 ‐3 ‐2 ‐1 0 1 Day ‐> ‐8 ‐7 ‐6 ‐5 ‐4 ‐3 ‐2 ‐1 0 1 A vancomycin x x x x A vancomycin x x x x B metronidazole x x B metronidazole x x C ceftriaxone x x x C ceftriaxone x x x D norepinephrine x x x D norepinephrine x x x E omeprazole x x x x E omeprazole x x x x F SQ heparin x x x x F SQ heparin x x x x trimethoprim/ trimethoprim/ G xxxxxxx G xxxxxxx sulfamethoxazole sulfamethoxazole Admit day Rash onset Admit day Rash onset Morbilliform (Simple) Drug Eruption Drug Induced Hypersensitivity Syndrome • Begins 5‐10 days after drug started -

Johnson & Johnson Reports 2008 Second-Quarter Results
Johnson & Johnson Reports 2008 Second-Quarter Results: Sales of $16.5 Billion Increased 8.7% Versus a Year Ago; EPS was $1.17 Excluding 2008 Special Items, Second-Quarter EPS of $1.18 Increased 12.4%* NEW BRUNSWICK, N.J., July 15, 2008 /PRNewswire-FirstCall via COMTEX News Network/ -- Johnson & Johnson today announced record sales of $16.5 billion for the second quarter of 2008, an increase of 8.7% as compared to the second quarter of 2007. Operational growth was 3.1% and the positive impact of currency was 5.6%. Domestic sales were up 2.1%, while international sales increased 16.2%, reflecting operational growth of 4.3% and a positive currency impact of 11.9%. Net earnings and diluted earnings per share for the second quarter of 2008 were $3.3 billion and $1.17, respectively. The second quarter of 2008 included an after-tax in-process research and development charge of $40 million associated with the acquisition of Amic, a developer of in vitro diagnostic technologies for use in Point-of-Care and near-patient settings. Excluding this charge, net earnings for the quarter of $3.4 billion and diluted earnings per share of $1.18 represent increases of 9.3% and 12.4 %, respectively, as compared to the same period in 2007.* The Company increased its earnings guidance for full- year 2008 to $4.45 - $4.50 per share, which does not include the impact of any in-process research and development charges or other special items. "Our solid earnings this quarter build on our strong track record of performance," said William C. -

Annual Report
ANNUAL REPORT 2019 MARCH 2020 To Our Shareholders Alex Gorsky Chairman and Chief Executive Officer By just about every measure, Johnson & These are some of the many financial and Johnson’s 133rd year was extraordinary. strategic achievements that were made possible by the commitment of our more than • We delivered strong operational revenue and 132,000 Johnson & Johnson colleagues, who adjusted operational earnings growth* that passionately lead the way in improving the health exceeded the financial performance goals we and well-being of people around the world. set for the Company at the start of 2019. • We again made record investments in research and development (R&D)—more than $11 billion across our Pharmaceutical, Medical Devices Propelled by our people, products, and and Consumer businesses—as we maintained a purpose, we look forward to the future relentless pursuit of innovation to develop vital with great confidence and optimism scientific breakthroughs. as we remain committed to leading • We proudly launched new transformational across the spectrum of healthcare. medicines for untreated and treatment-resistant diseases, while gaining approvals for new uses of many of our medicines already in the market. Through proactive leadership across our enterprise, we navigated a constant surge • We deployed approximately $7 billion, of unique and complex challenges, spanning primarily in transactions that fortify our dynamic global issues, shifting political commitment to digital surgery for a more climates, industry and competitive headwinds, personalized and elevated standard of and an ongoing litigious environment. healthcare, and that enhance our position in consumer skin health. As we have experienced for 133 years, we • And our teams around the world continued can be sure that 2020 will present a new set of working to address pressing public health opportunities and challenges.