Joint Industry Guide (Jig)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
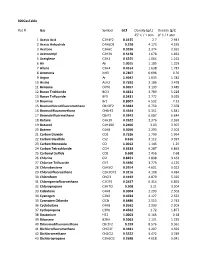
Gas Conversion Factor for 300 Series
300GasTable Rec # Gas Symbol GCF Density (g/L) Density (g/L) 25° C / 1 atm 0° C / 1 atm 1 Acetic Acid C2H4F2 0.4155 2.7 2.947 2 Acetic Anhydride C4H6O3 0.258 4.173 4.555 3 Acetone C3H6O 0.3556 2.374 2.591 4 Acetonitryl C2H3N 0.5178 1.678 1.832 5 Acetylene C2H2 0.6255 1.064 1.162 6 Air Air 1.0015 1.185 1.293 7 Allene C3H4 0.4514 1.638 1.787 8 Ammonia NH3 0.7807 0.696 0.76 9 Argon Ar 1.4047 1.633 1.782 10 Arsine AsH3 0.7592 3.186 3.478 11 Benzene C6H6 0.3057 3.193 3.485 12 Boron Trichloride BCl3 0.4421 4.789 5.228 13 Boron Triflouride BF3 0.5431 2.772 3.025 14 Bromine Br2 0.8007 6.532 7.13 15 Bromochlorodifluoromethane CBrClF2 0.3684 6.759 7.378 16 Bromodifluoromethane CHBrF2 0.4644 5.351 5.841 17 Bromotrifluormethane CBrF3 0.3943 6.087 6.644 18 Butane C4H10 0.2622 2.376 2.593 19 Butanol C4H10O 0.2406 3.03 3.307 20 Butene C4H8 0.3056 2.293 2.503 21 Carbon Dioxide CO2 0.7526 1.799 1.964 22 Carbon Disulfide CS2 0.616 3.112 3.397 23 Carbon Monoxide CO 1.0012 1.145 1.25 24 Carbon Tetrachloride CCl4 0.3333 6.287 6.863 25 Carbonyl Sulfide COS 0.668 2.456 2.68 26 Chlorine Cl2 0.8451 2.898 3.163 27 Chlorine Trifluoride ClF3 0.4496 3.779 4.125 28 Chlorobenzene C6H5Cl 0.2614 4.601 5.022 29 Chlorodifluoroethane C2H3ClF2 0.3216 4.108 4.484 30 Chloroform CHCl3 0.4192 4.879 5.326 31 Chloropentafluoroethane C2ClF5 0.2437 6.314 6.892 32 Chloropropane C3H7Cl 0.308 3.21 3.504 33 Cisbutene C4H8 0.3004 2.293 2.503 34 Cyanogen C2N2 0.4924 2.127 2.322 35 Cyanogen Chloride ClCN 0.6486 2.513 2.743 36 Cyclobutane C4H8 0.3562 2.293 2.503 37 Cyclopropane C3H6 0.4562 -

(12) United States Patent (10) Patent No.: US 8,911,640 B2 Nappa Et Al
USOO891. 1640B2 (12) United States Patent (10) Patent No.: US 8,911,640 B2 Nappa et al. (45) Date of Patent: Dec. 16, 2014 (54) COMPOSITIONS COMPRISING 5,736,063 A 4/1998 Richard et al. FLUOROOLEFNS AND USES THEREOF 5,744,052 A 4/1998 Bivens 5,788,886 A 8, 1998 Minor et al. 5,897.299 A * 4/1999 Fukunaga ..................... 417.316 (71) Applicant: E I du Pont de Nemours and 5,969,198 A 10/1999 Thenappan et al. Company, Wilmington, DE (US) 6,053,008 A 4/2000 Arman et al. 6,065.305 A 5/2000 Arman et al. (72) Inventors: Mario Joseph Nappa, Newark, DE 6,076,372 A 6/2000 Acharya et al. 6,111,150 A 8/2000 Sakyu et al. (US); Barbara Haviland Minor, Elkton, 6,176,102 B1 1/2001 Novak et al. MD (US); Allen Capron Sievert, 6,258,292 B1 7/2001 Turner Elkton, MD (US) 6,300,378 B1 10/2001 Tapscott 6.426,019 B1 7/2002 Acharya et al. (73) Assignee: E I du Pont de Nemours and 6,610,250 B1 8, 2003 Tuma Company, Wilmington, DE (US) 6,858,571 B2 2/2005 Pham et al. 6,969,701 B2 11/2005 Singh et al. 7,708,903 B2 5, 2010 Sievert et al. *) Notice: Subject to anyy disclaimer, the term of this 8,012,368 B2 9/2011 Nappa et al. patent is extended or adjusted under 35 8,070,976 B2 12/2011 Nappa et al. U.S.C. 154(b) by 0 days. -

Environmental Health Criteria 166 METHYL BROMIDE
Environmental Health Criteria 166 METHYL BROMIDE Please note that the layout and pagination of this web version are not identical with the printed version. Methyl Bromide (EHC 166, 1995) INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY ENVIRONMENTAL HEALTH CRITERIA 166 METHYL BROMIDE This report contains the collective views of an international group of experts and does not necessarily represent the decisions or the stated policy of the United Nations Environment Programme, the International Labour Organisation, or the World Health Organization. First draft prepared by Dr. R.F. Hertel and Dr. T. Kielhorn. Fraunhofer Institute of Toxicology and Aerosol Research, Hanover, Germany Published under the joint sponsorship of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization World Health Orgnization Geneva, 1995 The International Programme on Chemical Safety (IPCS) is a joint venture of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization. The main objective of the IPCS is to carry out and disseminate evaluations of the effects of chemicals on human health and the quality of the environment. Supporting activities include the development of epidemiological, experimental laboratory, and risk-assessment methods that could produce internationally comparable results, and the development of manpower in the field of toxicology. Other activities carried out by the IPCS include the development of know-how for coping with chemical accidents, -

Status of Numerical Modeling of Halocarbon Impacts on Stratospheric Ozone
THE STATUS OF NUMERICAL MODELING OF HALOCARBON IMPACTS ON STRATOSPHERIC OZONE Donald J. Wuebbles Department of Atmospheric Sciences University of Illinois, Urbana, IL 61801 Tel: 217-244-1568; Fax: 217-244-4393 [email protected] INTRODUCTION Numerical models of the chemistry and physics of the global atmosphere have played a key role in the scientific understanding of past, current and potential future effects of human-related emissions of halocarbons, including Halons, other brominated compounds and various replacement compounds on stratospheric ozone and climate change. As a result, these models have led directly to the Montreal Protocol and other national and international policy decisions regarding halocarbon controls due to their effects on stratospheric ozone. The purpose of this paper is to provide a perspective on atmospheric models, the role they have played in studies of the effects of Halons and other halocarbons on ozone, and to discuss what role models will likely play in future studies of stratospheric ozone THE COMPLEXITY OF STRATOSPHERIC OZONE It is important to recognize that the stratospheric ozone layer is a naturally occurring phenomenon that has great benefits to life on Earth. In fact, the formation of the ozone layer is generally believed to have played an important role in the development of life here on Earth. The accumulation of oxygen molecules in the atmosphere allowed for the production of ozone. Gradually the increasing levels of ozone led to the formation of the stratosphere, a region of the upper atmosphere where temperature increases with altitude largely as a result of the absorption of solar radiation by ozone. -

Al-46% ATTORNEY
Aug. 20, 1963 H. A. WIIST 3,101,304 DISTILLATION PROCESS FOR FLUOROCARBONS Filed April 28, 1960 INVENTOR ... al-46%HERBERT A. WIST ATTORNEY 3, 2,304 United States Patent Office Paterated Aug. 20, 1963 2 17 to 53 inclusive by extractive distillation. Compounds 3,105,304 causing the separation of the perfluorocarbons from the DSTELLATION PRO-CESS FOR FLUORO CARBONS close boiling or azeotrope forming fluorocarbons are aro Herbert Albert Wist, Wilmington, Dei. assignor to matic hydrocarbons, halogenated aromatic hydrocarbons E. I. du Pont de Nemours and Company, Wilmington, and halogenated aliphatic hydrocarbons in which the halo Del, a corporation of Delaware gen has an atomic number of 17 to 53, and which are Filed Apr. 28, 1960, Ser. No. 25,362 liquid at distillation conditions. The process of the pres 17 Clains. (C. 202-39.5) ent invention is based on the discovery that the volatility The process of the present invention relates to the sepa of fluorocarbons boiling close to the volatile perfuoro ration of fluorocarbons, and, more particularly, to the ex 10 carbons, which generally have from 1 to 5 carbon atoms, tractive distillation of fluorocarbons. or form. azeotropes with the fluorocarbons, is so changed Fluorocarbons, i.e., compounds of carbon and fluorine by the addition of the extractive distillation agents of the which may also contain other elements, such as hydro present invention, that the separation of these groups of . gen and chlorine, and particularly, perfluorocarbons, i.e., compounds becomes a matter of simple distillation. The compounds consisting solely of carbon and fluorine, are 15 close boiling or azeotrope forming fluorocarbons generally highly useful as refrigerants and solvents. -

Enthalpies of Vaporization of Organic and Organometallic Compounds, 1880–2002
Enthalpies of Vaporization of Organic and Organometallic Compounds, 1880–2002 James S. Chickosa… Department of Chemistry, University of Missouri-St. Louis, St. Louis, Missouri 63121 William E. Acree, Jr.b… Department of Chemistry, University of North Texas, Denton, Texas 76203 ͑Received 17 June 2002; accepted 17 October 2002; published 21 April 2003͒ A compendium of vaporization enthalpies published within the period 1910–2002 is reported. A brief review of temperature adjustments of vaporization enthalpies from temperature of measurement to the standard reference temperature, 298.15 K, is included as are recently suggested reference materials. Vaporization enthalpies are included for organic, organo-metallic, and a few inorganic compounds. This compendium is the third in a series focusing on phase change enthalpies. Previous compendia focused on fusion and sublimation enthalpies. Sufficient data are presently available for many compounds that thermodynamic cycles can be constructed to evaluate the reliability of the measure- ments. A protocol for doing so is described. © 2003 American Institute of Physics. ͓DOI: 10.1063/1.1529214͔ Key words: compendium; enthalpies of condensation; evaporation; organic compounds; vaporization enthalpy. Contents inorganic compounds, 1880–2002. ............. 820 1. Introduction................................ 519 8. References to Tables 6 and 7.................. 853 2. Reference Materials for Vaporization Enthalpy Measurements.............................. 520 List of Figures 3. Heat Capacity Adjustments. ................. 520 1. A thermodynamic cycle for adjusting vaporization ϭ 4. Group Additivity Values for C (298.15 K) enthalpies to T 298.15 K.................... 521 pl 2. A hypothetical molecule illustrating the different Estimations................................ 521 hydrocarbon groups in estimating C ........... 523 5. A Thermochemical Cycle: Sublimation, p Vaporization, and Fusion Enthalpies........... -

United States Patent (19) 11) Patent Number: 4,806,276 Maier (45) Date of Patent: Feb
United States Patent (19) 11) Patent Number: 4,806,276 Maier (45) Date of Patent: Feb. 21, 1989 (54 ADDITIVE FORTRANSFORMER OILS EPRI Report Project 2028-12, Mar. 1986, pp. S-2-S-4 and 4-1-4-4. (76) Inventor: Bruce R. Maier, 3000 Woodbine, Moore, Fire Protection Handbook, 16th ed., Section 19, Columbia, Mo. 65203 Ch. 2, "Halogenated Agents and Systems'. 21 Appl. No.: 130,014 Primary Examiner-Robert A. Wax 22) Filed: Dec. 8, 1987 Attorney, Agent, or Firm-Kokjer, Kircher, Bradley, 51) Int. Cl. ............................................... HOB 3/24 Wharton, Bowman & Johnson 52 U.S. C. .................................... 252/570; 252/162; 57 ABSTRACT 252/171; 252/172; 252/400.1; 585/6.6 An additive for transformer oils comprising a non-ionic 58) Field of Search ............ 252/162, 171, 172,400.1, fluorosurfactant and a halogenated hydrocarbon where 252/570; 585/6.6 the co-mixture is a liquid at least 70 F. and methods for (56) References Cited preparing and using same. The halogenated hydrocar U.S. PATENT DOCUMENTS bon is preferably chosen from the group comprising C1-C3 alkanes which can be fully halogenated or can 3,772,195 11/1973 Francen ............................. 252/8.05 3,954,882 5/1976 Kalopissis et al. ... 424/70 retain some hydrogen atoms on the structure. The most 4,169,076 9/1979 Kawakami et al. ... ... 252/558 preferred halogenated hydrocarbons being dibromotet 4,207,421 6/1980 Scardera et al. .................... 568/625 rafluoroethane, dibromodifluoromethane or bromo 4,238,373 12/1980 Hardy et al. ........................ 252/546 chloromethane. The most useful surfactants for use in 4,242,516 12/1980 Mueller .............................. -

Refrigerants
CHAPTER 19 REFRIGERANTS Phaseout of Refrigerants .............................................................................................................. 19.1 Refrigerant Properties .................................................................................................................. 19.4 Refrigerant Performance ............................................................................................................. 19.6 Safety ............................................................................................................................................ 19.6 Leak Detection ............................................................................................................................. 19.7 Effect on Construction Materials ................................................................................................ 19.11 EFRIGERANTS are the working fluids in refrigeration, air- Transport properties of thermal conductivity and viscosity affect Rconditioning, and heat pumping systems. They absorb heat the performance of heat exchangers and piping. High thermal con- from one area, such as an air-conditioned space, and reject it into ductivity and low viscosity are desirable. another, such as outdoors, usually through evaporation and conden- No single fluid satisfies all the attributes desired of a refrigerant; sation, respectively. These phase changes occur both in absorption as a result, a variety of refrigerants is used. This chapter describes and mechanical vapor compression systems, but -
C1-C4 Halogenated HC Info 16FEB2018.Xlsx
C1-C4 Halogenated Hydrocarbons/Halocarbons Not Otherwise Listed (C1-C4 NOL) Feb 28, 2018 Administrative Council Mtg - Printed 2/16/20184:11 PM Reported Uses (SOURCES:HAZMAP, NJDHSS, Wikipedia, TSCA CDR, mfr literature) TURA TSCA CDR Chemical CAS/Chemica chemical name chemical fire 2015 Chemical Name Pesti- blowing feedstock / supressant / propell- Inventory? TRI? List l Number (synonyms) formula Solvent refriger-ant etchant Other Tier II cide agent intermed-iate flame ant status* retardant insulation for refrigerators, freezers, commercial refrigeration equipment, refrigerated containers and LNG ships; spray foam insulation; insulated metal panels; slabstock and molded flexible foam; refrigerant for chillers; and solvents for metal cleaning and electronics, and circuit flush. https://www.honeywell-blowingagents.com/?document=solstice-lba-technical- HFO-1233zd (E) brochure&download=1 Hydro-fluoro-olefin C H ClF YYY yhttps://www.honeywell-refrigerants.com/americas/product/solstice-zd/ (HFO) 3 2 3 CDR 2016 Honeywell: 100% as propellants and blowing agents for plastics product mfg; R-1233zd CDR Honeywell Solstice® ZD refrigerant, Solstice® 1233zdE, Solstice Blowing Agent; Arkema (current Forane® 1233zd blowing agent for spray PU foam, appliance insulation, etc. and solvent (may Honeywell not be available in US) product) C1-C4 Cat 102687-65-0 1-Chloro-3,3,3-trifluoropropene Y 106‐95‐6 Allyl bromide 2-Propenyl bromide C3H5Br Y y Manufacture of synthetic perfumes, other allyl compounds; Insecticidal fumigant; 1-Propene, 3-bromo- Chemical intermediate in organic synthesis, for resins (copolymer with sulfur dioxide) and fragrances; As a fumigant (if quite volatile) or as a contact poison; C1-C4 Cat Used in the manufacture of plastics and dyestuff. -

Flammable Refrigerants the Evolving Impact on Codes
Flammable Refrigerants The Evolving Impact on Codes With appreciation to Danfoss North America and Tidwell Consulting Today’s Goal Explore the changing refrirgerant environment and its potential affect on codes. Courtesy: Johnson Controls (www.johnson controls.com) 2016 ICC Annual Conference Education Programs Kansas City, MO 1 Learning Objectives • Identify the environmental and code issues associated with the refrigerant gas changes. • Describe simple refrigeration physics, terminology and chemistry. • Identify current public safety code requirements and issues. • Identify proposed changes in refrigeration gases. Challenge . Traditional refrigerants are being banned or phased down . Potent greenhouse gases . Both air conditioning and refrigeration . Alternatives are available, but many are flammable . Codes currently do not allow their use 2016 ICC Annual Conference Education Programs Kansas City, MO 2 Part I: Refrigeration Concerns Why Change? GLOBAL WARMING POTENTIAL Chlorofluorocarbons (CFCs) and Hydrochlorofluorocarbons (HCFC) INFLUENCE WARMING GLOBAL 1960 1970 1980 1990 2000 2010 2020 2030 2040 2050 2016 ICC Annual Conference Education Programs Kansas City, MO 3 Montreal Protocol Phase-out Begins INFLUENCE January 1, 1989 WARMING GLOBAL WARMING POTENTIAL Chlorofluorocarbons (CFCs) and GLOBAL Hydrochlorofluorocarbons (HCFC) 1960 1970 1980 1990 2000 2010 2020 2030 2040 2050 Hydrofluorcarbons GLOBAL WARMING POTENTIAL Hydrofluorocarbons (HFCs) Phase-out Begins INFLUENCE January 1, 1989 WARMING GLOBAL 1960 1970 1980 1990 2000 2010 2020 -

CRITICAL CONSTANTS References
CRITICAL CONSTANTS The parameters of the liquid–gas critical point are important 15. VonNiederhausern, D. M., Wilson, G. M., and Giles, N. F., J. Chem. constants in determining the behavior of fluids. This table lists the Eng. Data , 45, 157, 2000. critical temperature, pressure, and molar volume, as well as the 16. Nikitin, E. D., Popov, A. P., Bogatishcheva, N. S., and Yatluk, Y. G., J. normal boiling point, for approximately 850 inorganic and organic Chem. Eng. Data, 47, 1012, 2002. 17. Wilson, G. M., VonNiederhausern, D. M., and Giles, N. F., J. Chem. substances. The properties and their units are: Eng. Data, 47, 761, 2002. 18. Wang, B. H., Adcock, J. L., Mathur, S. B., and Van Hook, W. A., J. T : Normal boiling point in kelvins at a pressure of 101.325 b Chem. Thermodynamics, 23, 699, 1991. kPa (1 atmosphere); an “s” following the value indicates 19. Chae, H. B., Schmidt, J. W., and Moldover, M. R., J. Phys. Chem ., 94, a sublimation point (temperature at which the solid is in 8840, 1990. equilibrium with the gas at a pressure of 101.325 kPa) 20. Dillon, I. G., Nelson, P. A., and Swanson, B. S., J. Chem. Phys ., 44, 4229, 1966. Tc: Critical temperature in kelvins 21. Physical Constants of Hydrocarbon and Non-Hydrocarbon Pc: Critical pressure in megapascals V : Critical molar volume in cm 3/mol Compounds , ASTM Data Series DS 4B, ASTM, Philadelphia, 1988. c 22. Nowak, P., Tielkes, T., Kleinraum, R., and Wagner, W., J. Chem. Thermodynamics, 29, 885, 1997. The number of digits given for Tb, Tc, and Pc indicates the esti- mated accuracy of these quantities; however, values of T greater 23. -

UNITED STATES PATENT OFFICE 2,658,086 BROMINATION of POLYHALOMETHANES Robert E
Patented Nov. 3, 1953 2,658,086 UNITED STATES PATENT OFFICE 2,658,086 BROMINATION OF POLYHALOMETHANES Robert E. Ruh and Ralph A. Davis, Midland, Mich, assignors to The Dow Chemical Com pany, Midland, Mich., a corporation of Dela Ware No Drawing. Application July 2, 1952, Serial No. 296,927 9 Claims. (C. 260-653) 2 This invention relates to the bromination of carried out employing an equimolecular propor polyhalomethanes, particularly difluoromethane tion of halogen, i.e., total bromine and chlorine, and trifluoromethane. theoretically required to effect the desired de In the vapor phase halogenation of fluorometh gree of bromination. To completely monobro anes, it has been observed that chlorine reacts minate a polyhalomethane, it is theoretically much more rapidly than bromine. For instance, di necessary to employ at least an equimolecular fluoromethane Will react with chlorine at a prac proportion of halogen consisting of a mixture of tical rate at temperatures of 100 to 400° C. On bromine and chlorine and containing at least 0.5 the other hand, when bromine is to be reacted mole of bromine per mole of polyhalomethane. with difluoromethane, temperatures of 400° to O Although reaction will proceed with less than 600° C. are required for satisfactory results. equimolecular proportions of halogen to poly It has now been found, quite surprisingly, that halomethane, it is usually desirable to employ When a mixture of chlorine and bromine is con greater than an equimolecular proportion of tacted at elevated temperatures with a polyhalo halogen. The halogen mixture should prefer methane, particularly a fluoromethane, the pre 5 ably contain an equimolecular or greater than ponderant reaction is bromination rather than equimolecular proportion of bromine to chlorine, chlorination.