Aristada Initio (Aripiprazole Lauroxil)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Aristada™ (Aripiprazole Lauroxil)
Aristada™ (aripiprazole lauroxil) – New Drug Approval • On October 5, 2015, Alkermes’ announced the FDA approval of Aristada (aripiprazole lauroxil) extended-release injection, an atypical antipsychotic, for the treatment of schizophrenia. • Schizophrenia is a chronic, severe and disabling brain disorder affecting an estimated 2.4 million Americans. Typically, symptoms include hearing voices, believing other people are reading their minds or controlling their thoughts, and being suspicious or withdrawn. • Aristada’s approval was based on data from a double-blind, placebo-controlled 12-week trial involving 622 patients with schizophrenia. In addition, the efficacy of Aristada was established, in part, on the basis of efficacy data from trials with oral aripiprazole. — Aristada significantly improved symptoms of schizophrenia compared to placebo at day 85. • Similar to other atypical antipsychotics, Aristada carries a boxed warning for increased mortality in elderly patients with dementia-related psychosis. • Other warnings and precautions for Aristada include cerebrovascular adverse reactions, including stroke; neuroleptic malignant syndrome; tardive dyskinesia; metabolic changes; orthostatic hypotension; leukopenia, neutropenia, and agranulocytosis; seizures; potential for cognitive and motor impairment; body temperature regulation; and dysphagia. • The most common adverse reaction (≥ 5% and at least twice that for placebo) with Aristada use was akathisia. • Aristada is administered by intramuscular injection in the deltoid (441 mg dose only) or gluteal (441 mg, 662 mg, or 882 mg) muscle by a healthcare professional. — Aristada can be initiated at a monthly dose (441 mg, 662 mg or 882 mg) or every 6 week dose (882 mg). — For patients naïve to aripiprazole, tolerability should be established with oral aripiprazole prior to initiating treatment with Aristada. -

Product Monograph
PRODUCT MONOGRAPH PrFLUANXOL® Flupentixol Tablets (as flupentixol dihydrochloride) 0.5 mg, 3 mg, and 5 mg PrFLUANXOL® DEPOT Flupentixol Decanoate Intramuscular Injection 2% and 10% flupentixol decanoate Antipsychotic Agent Lundbeck Canada Inc. Date of Revision: 2600 Alfred-Nobel December 12th, 2017 Suite 400 St-Laurent, QC H4S 0A9 Submission Control No : 209135 Page 1 of 35 Table of Contents PART I: HEALTH PROFESSIONAL INFORMATION .........................................................3 SUMMARY PRODUCT INFORMATION ........................................................................3 INDICATIONS AND CLINICAL USE ..............................................................................3 CONTRAINDICATIONS ...................................................................................................4 WARNINGS AND PRECAUTIONS ..................................................................................4 ADVERSE REACTIONS ..................................................................................................10 DRUG INTERACTIONS ..................................................................................................13 DOSAGE AND ADMINISTRATION ..............................................................................15 OVERDOSAGE ................................................................................................................18 ACTION AND CLINICAL PHARMACOLOGY ............................................................19 STORAGE AND STABILITY ..........................................................................................21 -

Papers and Originals
BRITISH MEDICAL JOURNAL 19 FEBRUARY 1972 463 PAPERS AND ORIGINALS Drug Interaction: Inhibitory Effect of Neuroleptics on Metabolism of Tricyclic Antidepressants in Man LARS F. GRAM, KERSTIN FREDRICSON OVERO British Medical Journal, 1972, 1, 463-465 excretion (Anders, 1971). There are relatively few reports on interaction concerning neuroleptics or tricyclic antidepressants. Summary Chlorpromazine has both stimulatory and inhibitory effects on the metabolism of hexobarbitone in Total urinary excretion of radioactivity after oral or experimental animals of a test of (Riimke and Bout, 1960-1). Furthermore, chlorpromazine intravenous administration dose '4C-imi- accelerates the metabolism pramine was measured in were tested ofmeprobamate (Kato and Vassanelli, eight patients. They 1962). Tricyclic antidepressants have been shown to inhibit the before, during, and after treatment with neuroleptics. metabolism Excretion diminished while the were of tremorine and oxotremorine in rats in vitro and patients being in vivo (Hammer and Sjoqvist, 1967). It has also been found treated with perphenazine, haloperidol, or chlorproma- that zine, not treatment. desipramine hydrochloride inhibits the metabolism of though during flupenthixol amphetamine in isolated, perfused rat liver (Dingell and Bass, Total urinary excretion of radioactivity and plasma 1969). Pretreatment levels of metabolites and were measured with phenobarbitone decreased steady-state unchanged drug plasma levels of desipramine and nortriptyline in man (Sjoqvist in five patients after a test dose of 14C-nortriptyline. Each et was before and al. 1968). These findings were later confirmed in a twin study patient tested again during perphenazine (Alexanderson et al., 1969). Forrest et treatment. In all patients perphenazine treatment caused: al. (1970) found increased (1) decrease of total urinary excretion, (2) decreased urinary excretion of chlorpromazine metabolites when patients plasma level of metabolites, and (3) increased plasma were given additional treatment with phenobarbitone. -
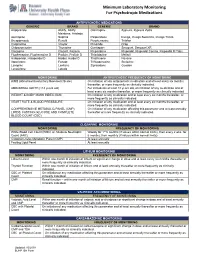
Minimum Laboratory Monitoring for Psychotropic Medications
Minimum Laboratory Monitoring For Psychotropic Medications ANTIPSYCHOTIC MEDICATIONS GENERIC BRAND GENERIC BRAND Aripiprazole Abilify, Abilify Olanzapine Zyprexa, Zyprexa Zydis Maintena, Aristada Asenapine Saphris Paliperidone Invega, Invega Sustenna, Invega Trinza Brexpiprazole Rexulti Perphenazine Trilafon Cariprazine Vraylar Pimozide Orap Chlorpromazine Thorazine Quetiapine Seroquel, Seroquel XR Clozapine Clozaril, Fazaclo Risperidone Risperdal, Risperdal Consta, Risperdal M Tabs Fluphenazine, Fluphenazine D Prolixin, Prolixin D Thioridazine Mellaril Haloperidol, Haloperidol D Haldol, Haldol D Thiothixene Navane Iloperidone Fanapt Trifluoperazine Stelazine Loxapine Loxitane Ziprasidone Geodon Lurasidone Latuda MONITORING ANTIPSYCHOTIC FREQUENCY OF MONITORING AIMS (Abnormal Involuntary Movement Scale) On initiation of any antipsychotic medication and at least every six months thereafter, or more frequently as clinically indicated. ABDOMINAL GIRTH (>18 years old) For individuals at least 18 years old, on initiation of any medication and at least every six months thereafter, or more frequently as clinically indicated. WEIGHT & BODY MASS INDEX (BMI) On initiation of any medication and at least every six months thereafter, or more frequently as clinically indicated. HEART RATE & BLOOD PRESSSURE On initiation of any medication and at least every six months thereafter, or more frequently as clinically indicated. COMPREHENSIVE METABOLIC PANEL (CMP), On initiation of any medication affecting this parameter and at least annually LIPIDS, FASTING -

Modern Antipsychotic Drugs: a Critical Overview
Review Synthèse Modern antipsychotic drugs: a critical overview David M. Gardner, Ross J. Baldessarini, Paul Waraich Abstract mine was based primarily on the finding that dopamine ago- nists produced or worsened psychosis and that antagonists CONVENTIONAL ANTIPSYCHOTIC DRUGS, used for a half century to treat were clinically effective against psychotic and manic symp- a range of major psychiatric disorders, are being replaced in clini- 5 toms. Blocking dopamine D2 receptors may be a critical or cal practice by modern “atypical” antipsychotics, including ari- even sufficient neuropharmacologic action of most clinically piprazole, clozapine, olanzapine, quetiapine, risperidone and effective antipsychotic drugs, especially against hallucina- ziprasidone among others. As a class, the newer drugs have been promoted as being broadly clinically superior, but the evidence for tions and delusions, but it is not necessarily the only mecha- this is problematic. In this brief critical overview, we consider the nism for antipsychotic activity. Moreover, this activity, and pharmacology, therapeutic effectiveness, tolerability, adverse ef- subsequent pharmacocentric and circular speculations about fects and costs of individual modern agents versus older antipsy- altered dopaminergic function, have not led to a better un- chotic drugs. Because of typically minor differences between derstanding of the pathophysiology or causes of the several agents in clinical effectiveness and tolerability, and because of still idiopathic psychotic disorders, nor have they provided a growing concerns about potential adverse long-term health conse- non-empirical, theoretical basis for the design or discovery quences of some modern agents, it is reasonable to consider both of improved treatments for psychotic disorders. older and newer drugs for clinical use, and it is important to inform The neuropharmacodynamics of specific modern anti- patients of relative benefits, risks and costs of specific choices. -
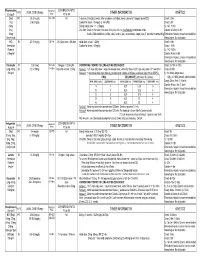
Medication Conversion Chart
Fluphenazine FREQUENCY CONVERSION RATIO ROUTE USUAL DOSE (Range) (Range) OTHER INFORMATION KINETICS Prolixin® PO to IM Oral PO 2.5-20 mg/dy QD - QID NA ↑ dose by 2.5mg/dy Q week. After symptoms controlled, slowly ↓ dose to 1-5mg/dy (dosed QD) Onset: ≤ 1hr 1mg (2-60 mg/dy) Caution for doses > 20mg/dy (↑ risk EPS) Cmax: 0.5hr 2.5mg Elderly: Initial dose = 1 - 2.5mg/dy t½: 14.7-15.3hr 5mg Oral Soln: Dilute in 2oz water, tomato or fruit juice, milk, or uncaffeinated carbonated drinks Duration of Action: 6-8hr 10mg Avoid caffeinated drinks (coffee, cola), tannics (tea), or pectinates (apple juice) 2° possible incompatibilityElimination: Hepatic to inactive metabolites 5mg/ml soln Hemodialysis: Not dialyzable HCl IM 2.5-10 mg/dy Q6-8 hr 1/3-1/2 po dose = IM dose Initial dose (usual): 1.25mg Onset: ≤ 1hr Immediate Caution for doses > 10mg/dy Cmax: 1.5-2hr Release t½: 14.7-15.3hr 2.5mg/ml Duration Action: 6-8hr Elimination: Hepatic to inactive metabolites Hemodialysis: Not dialyzable Decanoate IM 12.5-50mg Q2-3 wks 10mg po = 12.5mg IM CONVERTING FROM PO TO LONG-ACTING DECANOATE: Onset: 24-72hr (4-72hr) Long-Acting SC (12.5-100mg) (1-4 wks) Round to nearest 12.5mg Method 1: 1.25 X po daily dose = equiv decanoate dose; admin Q2-3wks. Cont ½ po daily dose X 1st few mths Cmax: 48-96hr 25mg/ml Method 2: ↑ decanoate dose over 4wks & ↓ po dose over 4-8wks as follows (accelerate taper for sx of EPS): t½: 6.8-9.6dy (single dose) ORAL DECANOATE (Administer Q 2 weeks) 15dy (14-100dy chronic administration) ORAL DOSE (mg/dy) ↓ DOSE OVER (wks) INITIAL DOSE (mg) TARGET DOSE (mg) DOSE OVER (wks) Steady State: 2mth (1.5-3mth) 5 4 6.25 6.25 0 Duration Action: 2wk (1-6wk) Elimination: Hepatic to inactive metabolites 10 4 6.25 12.5 4 Hemodialysis: Not dialyzable 20 8 6.25 12.5 4 30 8 6.25 25 4 40 8 6.25 25 4 Method 3: Admin equivalent decanoate dose Q2-3wks. -

Drug Use Evaluation: Antipsychotic Utilization in Schizophrenia Patients
© Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 | Fax 503-947-1119 Drug Use Evaluation: Antipsychotic Utilization in Schizophrenia Patients Research Questions: 1. How many schizophrenia patients are prescribed recommended first-line second-generation treatments for schizophrenia? 2. How many schizophrenia patients switch to an injectable antipsychotic after stabilization on an oral antipsychotic? 3. How many schizophrenia patients are prescribed 2 or more concomitant antipsychotics? 4. Are claims for long-acting injectable antipsychotics primarily billed as pharmacy or physician administered claims? 5. Does adherence to antipsychotic therapy differ between patients with claims for different routes of administration (oral vs. long-acting injectable)? Conclusions: In total, 4663 schizophrenia patients met inclusion criteria, and approximately 14% of patients (n=685) were identified as treatment naïve without claims for antipsychotics in the year before their first antipsychotic prescription. Approximately 45% of patients identified as treatment naïve had a history of remote antipsychotic use, but it is unclear if antipsychotics were historically prescribed for schizophrenia. Oral second-generation antipsychotics which are recommended as first-line treatment in the MHCAG schizophrenia algorithm were prescribed as initial treatment in 37% of treatment naive patients and 28% of all schizophrenia patients. Recommended agents include risperidone, paliperidone, and aripiprazole. Utilization of parenteral antipsychotics was limited in patients with schizophrenia. Overall only 8% of patients switched from an oral to an injectable therapy within 6 months of their first claim. Approximately, 60% of all schizophrenia patients (n=2512) had claims for a single antipsychotic for at least 12 continuous weeks and may be eligible to transition to a long-acting injectable antipsychotic. -

Schizophrenia Care Guide
August 2015 CCHCS/DHCS Care Guide: Schizophrenia SUMMARY DECISION SUPPORT PATIENT EDUCATION/SELF MANAGEMENT GOALS ALERTS Minimize frequency and severity of psychotic episodes Suicidal ideation or gestures Encourage medication adherence Abnormal movements Manage medication side effects Delusions Monitor as clinically appropriate Neuroleptic Malignant Syndrome Danger to self or others DIAGNOSTIC CRITERIA/EVALUATION (PER DSM V) 1. Rule out delirium or other medical illnesses mimicking schizophrenia (see page 5), medications or drugs of abuse causing psychosis (see page 6), other mental illness causes of psychosis, e.g., Bipolar Mania or Depression, Major Depression, PTSD, borderline personality disorder (see page 4). Ideas in patients (even odd ideas) that we disagree with can be learned and are therefore not necessarily signs of schizophrenia. Schizophrenia is a world-wide phenomenon that can occur in cultures with widely differing ideas. 2. Diagnosis is made based on the following: (Criteria A and B must be met) A. Two of the following symptoms/signs must be present over much of at least one month (unless treated), with a significant impact on social or occupational functioning, over at least a 6-month period of time: Delusions, Hallucinations, Disorganized Speech, Negative symptoms (social withdrawal, poverty of thought, etc.), severely disorganized or catatonic behavior. B. At least one of the symptoms/signs should be Delusions, Hallucinations, or Disorganized Speech. TREATMENT OPTIONS MEDICATIONS Informed consent for psychotropic -

Trifluoperazine 1Mg/5Ml Syrup Can Trifluoperazine 1Mg/5Ml Syrup Have Effects on Muscle Control
• Rarely patients may develop Neuroleptic Malignant Syndrome. This causes a high temperature, rigid muscles, drowsiness, occasional loss of consciousness, and requires emergency admission to hospital for treatment. PATIENT INFORMATION LEAFLET • If you have chest pain (angina) and your pain is getting worse. • Very occasionally, medicines such as Trifluoperazine 1mg/5ml Syrup can Trifluoperazine 1mg/5ml Syrup have effects on muscle control. If this happens, symptoms can include slurred speech, odd movements of the face, particularly of the tongue, eyes, head or neck (such as twisting of the neck which causes an Read all of this leaflet carefully before you start taking this medicine. unnatural positioning of the head, rigid muscles, tremors or restlessness Keep this leaflet. You may need to read it again. and difficulty in sitting still). Some patients (especially on high doses of If you have any further questions, ask your doctor or pharmacist. this medicine) experience problems with muscle control which may This medicine has been prescribed for you. Do not pass it on to others. It continue for years. Such patients may experience constant chewing or may harm them, even if their symptoms are the same as yours. tongue movements or other gentle movements of the neck, head or trunk. If any of the side effects become serious, or if you notice any side effects Uncontrollable movements of the arms and legs have also been reported not listed in this leaflet, please tell your doctor or pharmacist. in these patients. In this leaflet: • Occasionally, some patients have complained of feeling slowed down, 1. What Trifluoperazine 1mg/5ml Syrup is and what it is used for whilst • Rarely, jaundice (yellowing of skin and whites of eyes), eye problems, 2. -

Opipramol and Trifluoperazine in the Treatment of Anxiety and Tension
A COMPARATIVE STUDY OF TWO ANTIHISTAMINES 395 In table VI are given the results of a question directed at discovering how effective the preferred treatment was compared to any other antihistamine used. Fifteen patients only were able to give this information but the results are interesting. Discussion TABLE VI The design of this study was very simple as COMPARISON WITH PREVIOUSLY USED ANTIHISTAMINE such studies must be if they are to be completed under the conditions of a busy general practice. In a condition such as hay fever where anti- Worse As good Better Total histamines are known to be etfective and where fairly rapid symptomatic relief is needed by the 2 4 9 15* patient, we did not feel justified in including a placebo. The results of the study seem fairly *In the majority of patients the previously used clear-cut that if patients are offered the choice antihistamine was the chemically related chlor- of a plain form of this antihistamine or a long- pheniramine maleate B.P. acting form more will choose the latter. Ofthose that do so, however, only about a third find a single tablet at night completely effective, the remainder have to take one further tablet during the day. Both forms of antihistamine are effective and where a retrospective comparison can be made this effectiveness is considered greater or equal to that of previously administered anti-histamines. Summary A simple comparative study under general-practice conditions of two formulations of pheniramine is described. Sixty-one per cent of patients preferred the long-acting form of this antihistamine. -

Current P SYCHIATRY
Current p SYCHIATRY N ew Investigators Tips to manage and prevent discontinuation syndromes Informed tapering can protect patients when you stop a medication Sriram Ramaswamy, MD Shruti Malik, MBBS, MHSA Vijay Dewan, MD Instructor, department of psychiatry Foreign medical graduate Assistant professor Creighton University Department of psychiatry Omaha, NE University of Nebraska Medical Center Omaha, NE bruptly stopping common psychotropics New insights on psychotropic A —particularly antidepressants, benzodi- drug safety and side effects azepines, or atypical antipsychotics—can trigger a discontinuation syndrome, with: This paper was among those entered in the 2005 • rebound or relapse of original symptoms Promising New Investigators competition sponsored • uncomfortable new physical and psycho- by the Neuroleptic Malignant Syndrome Information Service (NMSIS). The theme of this year’s competition logical symptoms was “New insights on psychotropic drug safety and • physiologic withdrawal at times. side effects.” To increase health professionals’ awareness of URRENT SYCHIATRY 1 C P is honored to publish this peer- the risk of these adverse effects, this article reviewed, evidence-based article on a clinically describes discontinuation syndromes associated important topic for practicing psychiatrists. with various psychotropics and offers strategies to NMSIS is dedicated to reducing morbidity and anticipate, recognize, and manage them. mortality of NMS by improving medical and psychiatric care of patients with heat-related disorders; providing -

Neuroprotection by Chlorpromazine and Promethazine in Severe Transient and Permanent Ischemic Stroke
Mol Neurobiol (2017) 54:8140–8150 DOI 10.1007/s12035-016-0280-x Neuroprotection by Chlorpromazine and Promethazine in Severe Transient and Permanent Ischemic Stroke Xiaokun Geng1,2 & Fengwu Li1 & James Yip2 & Changya Peng2 & Omar Elmadhoun2 & Jiamei Shen1 & Xunming Ji1,3 & Yuchuan Ding1,2 Received: 29 June 2016 /Accepted: 31 October 2016 /Published online: 28 November 2016 # Springer Science+Business Media New York 2016 Abstract Previous studies have demonstrated depressive or enhance C + P-induced neuroprotection. C + P therapy im- hibernation-like roles of phenothiazine neuroleptics [com- proved brain metabolism as determined by increased ATP bined chlorpromazine and promethazine (C + P)] in brain levels and NADH activity, as well as decreased ROS produc- activity. This ischemic stroke study aimed to establish neuro- tion. These therapeutic effects were associated with alterations protection by reducing oxidative stress and improving brain in PKC-δ and Akt protein expression. C + P treatments con- metabolism with post-ischemic C + P administration. ferred neuroprotection in severe stroke models by suppressing Sprague-Dawley rats were subjected to transient (2 or 4 h) the damaging cascade of metabolic events, most likely inde- middle cerebral artery occlusion (MCAO) followed by 6 or pendent of drug-induced hypothermia. These findings further 24 h reperfusion, or permanent (28 h) MCAO without reper- prove the clinical potential for C + P treatment and may direct fusion. At 2 h after ischemia onset, rats received either an us closer towards the development of an efficacious neuropro- intraperitoneal (IP) injection of saline or two doses of C + P. tective therapy. Body temperatures, brain infarct volumes, and neurological deficits were examined.