INVESTIGATIONS on Mrna MATURATION DISTURBANCIES in the PATHOGENESIS of PSORIASIS
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Molecular and Physiological Basis for Hair Loss in Near Naked Hairless and Oak Ridge Rhino-Like Mouse Models: Tracking the Role of the Hairless Gene
University of Tennessee, Knoxville TRACE: Tennessee Research and Creative Exchange Doctoral Dissertations Graduate School 5-2006 Molecular and Physiological Basis for Hair Loss in Near Naked Hairless and Oak Ridge Rhino-like Mouse Models: Tracking the Role of the Hairless Gene Yutao Liu University of Tennessee - Knoxville Follow this and additional works at: https://trace.tennessee.edu/utk_graddiss Part of the Life Sciences Commons Recommended Citation Liu, Yutao, "Molecular and Physiological Basis for Hair Loss in Near Naked Hairless and Oak Ridge Rhino- like Mouse Models: Tracking the Role of the Hairless Gene. " PhD diss., University of Tennessee, 2006. https://trace.tennessee.edu/utk_graddiss/1824 This Dissertation is brought to you for free and open access by the Graduate School at TRACE: Tennessee Research and Creative Exchange. It has been accepted for inclusion in Doctoral Dissertations by an authorized administrator of TRACE: Tennessee Research and Creative Exchange. For more information, please contact [email protected]. To the Graduate Council: I am submitting herewith a dissertation written by Yutao Liu entitled "Molecular and Physiological Basis for Hair Loss in Near Naked Hairless and Oak Ridge Rhino-like Mouse Models: Tracking the Role of the Hairless Gene." I have examined the final electronic copy of this dissertation for form and content and recommend that it be accepted in partial fulfillment of the requirements for the degree of Doctor of Philosophy, with a major in Life Sciences. Brynn H. Voy, Major Professor We have read this dissertation and recommend its acceptance: Naima Moustaid-Moussa, Yisong Wang, Rogert Hettich Accepted for the Council: Carolyn R. -

Independent Apoptotic Pathways
An Sp1 transcription factor coordinates caspase- dependent and -independent apoptotic pathways The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation Hirose, Takashi, and H. Robert Horvitz. “An Sp1 Transcription Factor Coordinates Caspase-Dependent and -Independent Apoptotic Pathways.” Nature 500, no. 7462 (July 14, 2013): 354–358. As Published http://dx.doi.org/10.1038/nature12329 Publisher Nature Publishing Group Version Author's final manuscript Citable link http://hdl.handle.net/1721.1/85566 Terms of Use Article is made available in accordance with the publisher's policy and may be subject to US copyright law. Please refer to the publisher's site for terms of use. Published as: Nature. 2013 August 15; 500(7462): 354–358. HHMI Author Manuscript HHMI Author Manuscript HHMI Author Manuscript An Sp1 transcription factor coordinates caspase-dependent and -independent apoptotic pathways Takashi Hirose and H. Robert Horvitz Howard Hughes Medical Institute, Department of Biology, 68-425, Massachusetts Institute of Technology, 77 Massachusetts Avenue, Cambridge, MA 02139, USA Abstract During animal development, the proper regulation of apoptosis requires the precise spatial and temporal execution of cell-death programs, which can include both caspase-dependent and caspase-independent pathways1, 2. While the mechanisms of caspase-dependent and caspase- independent cell killing have been examined extensively, how these pathways are coordinated within a single cell that is fated to die is unknown. Here we show that the C. elegans Sp1 transcription factor SPTF-3 specifies the programmed cell deaths of at least two cells, the sisters of the pharyngeal M4 motor neuron and of the AQR sensory neuron, by transcriptionally activating both caspase-dependent and caspase-independent apoptotic pathways. -

Pinin (PNN) Rabbit Polyclonal Antibody – TA337687 | Origene
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for TA337687 Pinin (PNN) Rabbit Polyclonal Antibody Product data: Product Type: Primary Antibodies Applications: WB Recommended Dilution: WB Reactivity: Human Host: Rabbit Isotype: IgG Clonality: Polyclonal Immunogen: The immunogen for anti-PNN antibody: synthetic peptide directed towards the N terminal of human PNN. Synthetic peptide located within the following region: MAVAVRTLQEQLEKAKESLKNVDENIRKLTGRDPNDVRPIQARLLALSGP Formulation: Liquid. Purified antibody supplied in 1x PBS buffer with 0.09% (w/v) sodium azide and 2% sucrose. Note that this product is shipped as lyophilized powder to China customers. Purification: Affinity Purified Conjugation: Unconjugated Storage: Store at -20°C as received. Stability: Stable for 12 months from date of receipt. Predicted Protein Size: 81 kDa Gene Name: pinin, desmosome associated protein Database Link: NP_002678 Entrez Gene 5411 Human Q9H307 Background: PNN is the transcriptional activator binding to the E-box 1 core sequence of the E-cadherin promoter gene; the core-binding sequence is 5'CAGGTG-3'. PNN is capable of reversing CTBP1-mediated transcription repression. Component of a splicing-dependent mul Synonyms: DRS; DRSP; memA; SDK3 Note: Immunogen Sequence Homology: Dog: 100%; Pig: 100%; Rat: 100%; Human: 100%; Mouse: 100%; Bovine: 100%; Rabbit: 100%; Zebrafish: 100%; Guinea pig: 100% This product -

Greg's Awesome Thesis
Analysis of alignment error and sitewise constraint in mammalian comparative genomics Gregory Jordan European Bioinformatics Institute University of Cambridge A dissertation submitted for the degree of Doctor of Philosophy November 30, 2011 To my parents, who kept us thinking and playing This dissertation is the result of my own work and includes nothing which is the out- come of work done in collaboration except where specifically indicated in the text and acknowledgements. This dissertation is not substantially the same as any I have submitted for a degree, diploma or other qualification at any other university, and no part has already been, or is currently being submitted for any degree, diploma or other qualification. This dissertation does not exceed the specified length limit of 60,000 words as defined by the Biology Degree Committee. November 30, 2011 Gregory Jordan ii Analysis of alignment error and sitewise constraint in mammalian comparative genomics Summary Gregory Jordan November 30, 2011 Darwin College Insight into the evolution of protein-coding genes can be gained from the use of phylogenetic codon models. Recently sequenced mammalian genomes and powerful analysis methods developed over the past decade provide the potential to globally measure the impact of natural selection on pro- tein sequences at a fine scale. The detection of positive selection in particular is of great interest, with relevance to the study of host-parasite conflicts, immune system evolution and adaptive dif- ferences between species. This thesis examines the performance of methods for detecting positive selection first with a series of simulation experiments, and then with two empirical studies in mammals and primates. -

Exon Junction Complexes Can Have Distinct Functional Flavours To
www.nature.com/scientificreports OPEN Exon Junction Complexes can have distinct functional favours to regulate specifc splicing events Received: 9 November 2017 Zhen Wang1, Lionel Ballut2, Isabelle Barbosa1 & Hervé Le Hir1 Accepted: 11 June 2018 The exon junction complex (EJC) deposited on spliced mRNAs, plays a central role in the post- Published: xx xx xxxx transcriptional gene regulation and specifc gene expression. The EJC core complex is associated with multiple peripheral factors involved in various post-splicing events. Here, using recombinant complex reconstitution and transcriptome-wide analysis, we showed that the EJC peripheral protein complexes ASAP and PSAP form distinct complexes with the EJC core and can confer to EJCs distinct alternative splicing regulatory activities. This study provides the frst evidence that diferent EJCs can have distinct functions, illuminating EJC-dependent gene regulation. Te Exon Junction Complex (EJC) plays a central role in post-transcriptional gene expression control. EJCs tag mRNA exon junctions following intron removal by spliceosomes and accompany spliced mRNAs from the nucleus to the cytoplasm where they are displaced by the translating ribosomes1,2. Te EJC is organized around a core complex made of the proteins eIF4A3, MAGOH, Y14 and MLN51, and this EJC core serves as platforms for multiple peripheral factors during diferent post-transcriptional steps3,4. Dismantled during translation, EJCs mark a very precise period in mRNA life between nuclear splicing and cytoplasmic translation. In this window, EJCs contribute to alternative splicing5–7, intra-cellular RNA localization8, translation efciency9–11 and mRNA stability control by nonsense-mediated mRNA decay (NMD)12–14. At a physiological level, developmental defects and human pathological disorders due to altered expression of EJC proteins show that the EJC dosage is critical for specifc cell fate determinations, such as specifcation of embryonic body axis in drosophila, or Neural Stem Cells division in the mouse8,15,16. -
Supplementary Table 1
Supplementary Table 1. Large-scale quantitative phosphoproteomic profiling was performed on paired vehicle- and hormone-treated mTAL-enriched suspensions (n=3). A total of 654 unique phosphopeptides corresponding to 374 unique phosphoproteins were identified. The peptide sequence, phosphorylation site(s), and the corresponding protein name, gene symbol, and RefSeq Accession number are reported for each phosphopeptide identified in any one of three experimental pairs. For those 414 phosphopeptides that could be quantified in all three experimental pairs, the mean Hormone:Vehicle abundance ratio and corresponding standard error are also reported. Peptide Sequence column: * = phosphorylated residue Site(s) column: ^ = ambiguously assigned phosphorylation site Log2(H/V) Mean and SE columns: H = hormone-treated, V = vehicle-treated, n/a = peptide not observable in all 3 experimental pairs Sig. column: * = significantly changed Log 2(H/V), p<0.05 Log (H/V) Log (H/V) # Gene Symbol Protein Name Refseq Accession Peptide Sequence Site(s) 2 2 Sig. Mean SE 1 Aak1 AP2-associated protein kinase 1 NP_001166921 VGSLT*PPSS*PK T622^, S626^ 0.24 0.95 PREDICTED: ATP-binding cassette, sub-family A 2 Abca12 (ABC1), member 12 XP_237242 GLVQVLS*FFSQVQQQR S251^ 1.24 2.13 3 Abcc10 multidrug resistance-associated protein 7 NP_001101671 LMT*ELLS*GIRVLK T464, S468 -2.68 2.48 4 Abcf1 ATP-binding cassette sub-family F member 1 NP_001103353 QLSVPAS*DEEDEVPVPVPR S109 n/a n/a 5 Ablim1 actin-binding LIM protein 1 NP_001037859 PGSSIPGS*PGHTIYAK S51 -3.55 1.81 6 Ablim1 actin-binding -

Dissertation
Regulation of gene silencing: From microRNA biogenesis to post-translational modifications of TNRC6 complexes DISSERTATION zur Erlangung des DOKTORGRADES DER NATURWISSENSCHAFTEN (Dr. rer. nat.) der Fakultät Biologie und Vorklinische Medizin der Universität Regensburg vorgelegt von Johannes Danner aus Eggenfelden im Jahr 2017 Das Promotionsgesuch wurde eingereicht am: 12.09.2017 Die Arbeit wurde angeleitet von: Prof. Dr. Gunter Meister Johannes Danner Summary ‘From microRNA biogenesis to post-translational modifications of TNRC6 complexes’ summarizes the two main projects, beginning with the influence of specific RNA binding proteins on miRNA biogenesis processes. The fate of the mature miRNA is determined by the incorporation into Argonaute proteins followed by a complex formation with TNRC6 proteins as core molecules of gene silencing complexes. miRNAs are transcribed as stem-loop structured primary transcripts (pri-miRNA) by Pol II. The further nuclear processing is carried out by the microprocessor complex containing the RNase III enzyme Drosha, which cleaves the pri-miRNA to precursor-miRNA (pre-miRNA). After Exportin-5 mediated transport of the pre-miRNA to the cytoplasm, the RNase III enzyme Dicer cleaves off the terminal loop resulting in a 21-24 nt long double-stranded RNA. One of the strands is incorporated in the RNA-induced silencing complex (RISC), where it directly interacts with a member of the Argonaute protein family. The miRNA guides the mature RISC complex to partially complementary target sites on mRNAs leading to gene silencing. During this process TNRC6 proteins interact with Argonaute and recruit additional factors to mediate translational repression and target mRNA destabilization through deadenylation and decapping leading to mRNA decay. -

Network Pharmacology Interpretation of Fuzheng–Jiedu Decoction Against Colorectal Cancer
Hindawi Evidence-Based Complementary and Alternative Medicine Volume 2021, Article ID 4652492, 16 pages https://doi.org/10.1155/2021/4652492 Research Article Network Pharmacology Interpretation of Fuzheng–Jiedu Decoction against Colorectal Cancer Hongshuo Shi ,1 Sisheng Tian,2 and Hu Tian 3 1College of Traditional Chinese Medicine, Shandong University of Traditional Chinese Medicine, Jinan, Shandong, China 2School of Management, Shandong University of Traditional Chinese Medicine, Jinan, Shandong, China 3College of Traditional Chinese Medicine, Shandong University of Traditional Chinese Medicine, Jinan, Shandong, China Correspondence should be addressed to Hu Tian; [email protected] Received 7 April 2020; Revised 3 January 2021; Accepted 21 January 2021; Published 20 February 2021 Academic Editor: George B. Lenon Copyright © 2021 Hongshuo Shi et al. ,is is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Introduction. Traditional Chinese medicine (TCM) believes that the pathogenic factors of colorectal cancer (CRC) are “deficiency, dampness, stasis, and toxin,” and Fuzheng–Jiedu Decoction (FJD) can resist these factors. In this study, we want to find out the potential targets and pathways of FJD in the treatment of CRC and also explain from a scientific point of view that FJD multidrug combination can resist “deficiency, dampness, stasis, and toxin.” Methods. We get the composition of FJD from the TCMSP database and get its potential target. We also get the potential target of colorectal cancer according to the OMIM Database, TTD Database, GeneCards Database, CTD Database, DrugBank Database, and DisGeNET Database. -

The Exon Junction Complex Controls Transposable Element Activity by Ensuring Faithful Splicing of the Piwi Transcript
Downloaded from genesdev.cshlp.org on September 25, 2021 - Published by Cold Spring Harbor Laboratory Press The exon junction complex controls transposable element activity by ensuring faithful splicing of the piwi transcript Colin D. Malone,1,2,5 Claire Mestdagh,3,5 Junaid Akhtar,3 Nastasja Kreim,3 Pia Deinhard,3 Ravi Sachidanandam,4 Jessica Treisman,1 and Jean-Yves Roignant3 1Kimmel Center for Biology and Medicine at the Skirball Institute of Biomolecular Medicine, Department of Cell Biology, New York Universtiy School of Medicine, New York, New York 10016, USA; 2Howard Hughes Medical Institute, 3Institute of Molecular Biology (IMB), 55128 Mainz, Germany; 4Department of Oncological Sciences, Icahn School of Medicine at Mount Sinai, New York, New York 10029, USA The exon junction complex (EJC) is a highly conserved ribonucleoprotein complex that binds RNAs during splicing and remains associated with them following export to the cytoplasm. While the role of this complex in mRNA localization, translation, and degradation has been well characterized, its mechanism of action in splicing a subset of Drosophila and human transcripts remains to be elucidated. Here, we describe a novel function for the EJC and its splicing subunit, RnpS1, in preventing transposon accumulation in both Drosophila germline and surrounding somatic follicle cells. This function is mediated specifically through the control of piwi transcript splicing, where, in the absence of RnpS1, the fourth intron of piwi is retained. This intron contains a weak polypyrimidine tract that is sufficient to confer dependence on RnpS1. Finally, we demonstrate that RnpS1-dependent removal of this intron requires splicing of the flanking introns, suggesting a model in which the EJC facilitates the splicing of weak introns following its initial deposition at adjacent exon junctions. -

Transcriptome Analysis of Different Multidrug-Resistant Gastric Carcinoma Cells
in vivo 19: 583-590 (2005) Transcriptome Analysis of Different Multidrug-resistant Gastric Carcinoma Cells STEFFEN HEIM1 and HERMANN LAGE2 1Europroteome AG, Berlin-Hennigsdorf; 2Institute of Pathology, Charité Campus Mitte, Schumannstr. 20/21, D-10117 Berlin, Germany Abstract. Multidrug resistance (MDR) of human cancers is advanced cancer. These therapeutic protocols produce an the major cause of failure of chemotherapy. To better overall response rate of about 20% at best using a single- understand the molecular events associated with the agent, and up to 50% using combination chemotherapy. development of different types of MDR, two different multidrug- Thus, a large number of malignancies are incurable. resistant gastric carcinoma cell lines, the MDR1/P-glycoprotein- Generally, gastrointestinal cancers, including gastric expressing cell line EPG85-257RDB and the MDR1/P- carcinoma, are naturally resistant to many anticancer drugs. glycoprotein-negative cell variant EPG85-257RNOV, as well as Additionally, these tumors are able to develop acquired the corresponding drug-sensitive parental cell line EPG85-257P, drug resistance phenotypes, which include the multidrug were used for analyses of the mRNA expression profiles by resistance (MDR) phenomenon. cDNA array hybridization. Of more than 12,000 genes spotted The MDR phenotype is characterized by simultaneous on the arrays, 156 genes were detected as being significantly resistance of tumor cells to various antineoplastic agents regulated in the cell line EPG85-257RDB in comparison to the which are structurally and functionally unrelated. Besides non-resistant cell variant, and 61 genes were found to be the classical MDR phenotype, mediated by the enhanced differentially expressed in the cell line EPG85-257RNOV. -

Genetic Variation As a Tool for Identifying Novel Transducers of Itch
Genetic variation as a tool for identifying novel transducers of itch By Takeshi Morita A dissertation submitted in partial satisfaction of the requirements for the degree of Doctor of Philosophy in Molecular and Cell Biology in the Graduate Division of the University of California, Berkeley Committee in charge: Professor Diana M. Bautista, Co-Chair Professor Rachel B. Brem, Co-Chair Professor John Ngai Professor Kristin Scott Professor Michael W. Nachman Summer 2016 Abstract Genetic variation as a tool for identifying novel transducers of itch by Takeshi Morita Doctor of Philosophy in Molecular and Cell Biology University of California, Berkeley Professor Diana M. Bautista, Co-Chair Professor Rachel B. Brem, Co-Chair The mammalian somatosensory system mediates itch, the irritating sensation that elicits a desire to scratch. Millions of people worldwide suffer from chronic itch that fails to respond to current drugs and therapies. Even though recent studies have begun to elucidate the basic characteristics of the itch circuitry, we have little understanding about the molecules and signaling mechanisms that underlie detection and transduction of itch sensation, especially during chronic itch conditions. We have taken a genomic approach by harnessing natural variation in itch-evoked scratching behaviors in mice to identify novel molecular players that are involved in itch signal transduction at the level of primary sensory neurons. From our analysis, we identified numerous candidate itch genes, and further identified a serotonin receptor, HTR7 as a key transducer that is required for both development and maintenance of chronic itch. We further investigated the genetic basis of variation in itch, and identified a set of genes and regulatory pathways that may be involved in controlling itch behaviors. -
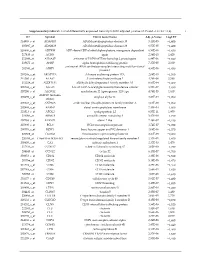
Supplementary Table S1. List of Differentially Expressed
Supplementary table S1. List of differentially expressed transcripts (FDR adjusted p‐value < 0.05 and −1.4 ≤ FC ≥1.4). 1 ID Symbol Entrez Gene Name Adj. p‐Value Log2 FC 214895_s_at ADAM10 ADAM metallopeptidase domain 10 3,11E‐05 −1,400 205997_at ADAM28 ADAM metallopeptidase domain 28 6,57E‐05 −1,400 220606_s_at ADPRM ADP‐ribose/CDP‐alcohol diphosphatase, manganese dependent 6,50E‐06 −1,430 217410_at AGRN agrin 2,34E‐10 1,420 212980_at AHSA2P activator of HSP90 ATPase homolog 2, pseudogene 6,44E‐06 −1,920 219672_at AHSP alpha hemoglobin stabilizing protein 7,27E‐05 2,330 aminoacyl tRNA synthetase complex interacting multifunctional 202541_at AIMP1 4,91E‐06 −1,830 protein 1 210269_s_at AKAP17A A‐kinase anchoring protein 17A 2,64E‐10 −1,560 211560_s_at ALAS2 5ʹ‐aminolevulinate synthase 2 4,28E‐06 3,560 212224_at ALDH1A1 aldehyde dehydrogenase 1 family member A1 8,93E‐04 −1,400 205583_s_at ALG13 ALG13 UDP‐N‐acetylglucosaminyltransferase subunit 9,50E‐07 −1,430 207206_s_at ALOX12 arachidonate 12‐lipoxygenase, 12S type 4,76E‐05 1,630 AMY1C (includes 208498_s_at amylase alpha 1C 3,83E‐05 −1,700 others) 201043_s_at ANP32A acidic nuclear phosphoprotein 32 family member A 5,61E‐09 −1,760 202888_s_at ANPEP alanyl aminopeptidase, membrane 7,40E‐04 −1,600 221013_s_at APOL2 apolipoprotein L2 6,57E‐11 1,600 219094_at ARMC8 armadillo repeat containing 8 3,47E‐08 −1,710 207798_s_at ATXN2L ataxin 2 like 2,16E‐07 −1,410 215990_s_at BCL6 BCL6 transcription repressor 1,74E‐07 −1,700 200776_s_at BZW1 basic leucine zipper and W2 domains 1 1,09E‐06 −1,570 222309_at