A Proposal for the Clinical Use of Flecainide
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Table 2. 2012 AGS Beers Criteria for Potentially
Table 2. 2012 AGS Beers Criteria for Potentially Inappropriate Medication Use in Older Adults Strength of Organ System/ Recommendat Quality of Recomm Therapeutic Category/Drug(s) Rationale ion Evidence endation References Anticholinergics (excludes TCAs) First-generation antihistamines Highly anticholinergic; Avoid Hydroxyzin Strong Agostini 2001 (as single agent or as part of clearance reduced with e and Boustani 2007 combination products) advanced age, and promethazi Guaiana 2010 Brompheniramine tolerance develops ne: high; Han 2001 Carbinoxamine when used as hypnotic; All others: Rudolph 2008 Chlorpheniramine increased risk of moderate Clemastine confusion, dry mouth, Cyproheptadine constipation, and other Dexbrompheniramine anticholinergic Dexchlorpheniramine effects/toxicity. Diphenhydramine (oral) Doxylamine Use of diphenhydramine in Hydroxyzine special situations such Promethazine as acute treatment of Triprolidine severe allergic reaction may be appropriate. Antiparkinson agents Not recommended for Avoid Moderate Strong Rudolph 2008 Benztropine (oral) prevention of Trihexyphenidyl extrapyramidal symptoms with antipsychotics; more effective agents available for treatment of Parkinson disease. Antispasmodics Highly anticholinergic, Avoid Moderate Strong Lechevallier- Belladonna alkaloids uncertain except in Michel 2005 Clidinium-chlordiazepoxide effectiveness. short-term Rudolph 2008 Dicyclomine palliative Hyoscyamine care to Propantheline decrease Scopolamine oral secretions. Antithrombotics Dipyridamole, oral short-acting* May -
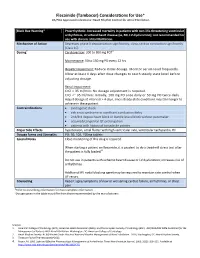
Flecainide Considerations For
Flecainide (Tambocor) Considerations for Use* US/FDA Approved Indications: Heart Rhythm Control for Atrial Fibrillation Black Box Warning* Proarrhythmic. Increased mortality in patients with non-life-threatening ventricular arrhythmias, structural heart disease (ie, MI, LV dysfunction); not recommended for use with chronic atrial fibrillation. Mechanism of Action Depresses phase 0 depolarization significantly, slows cardiac conduction significantly (Class 1C). Dosing† Cardioversion: 200 to 300 mg PO‡1 Maintenance: 50 to 150 mg PO every 12 hrs Hepatic Impairment: Reduce initial dosage. Monitor serum level frequently. Allow at least 4 days after dose changes to reach steady state level before adjusting dosage. Renal Impairment: CrCl > 35 ml/min: No dosage adjustment is required. CrCl <= 35 ml/min: Initially, 100 mg PO once daily or 50 mg PO twice daily. Adjust dosage at intervals > 4 days, since steady-state conditions may take longer to achieve in these patient Contraindications cardiogenic shock sick sinus syndrome or significant conduction delay 2nd/3rd degree heart block or bundle brand block without pacemaker acquired/congenital QT prolongation patients with history of torsade de pointes Major Side Effects hypotension, atrial flutter with high ventricular rate, ventricular tachycardia, HF Dosage forms and Strengths PO: 50, 100, 150mg tablets Special Notes Close monitoring of this drug is required. When starting a patient on flecainide, it is prudent to do a treadmill stress test after the patient is fully loaded.4 Do not use in patients with ischemic heart disease or LV dysfunction; increases risk of arrhythmias. Additional AV nodal blocking agent may be required to maintain rate control when AF recurs. -

Guideline for Preoperative Medication Management
Guideline: Preoperative Medication Management Guideline for Preoperative Medication Management Purpose of Guideline: To provide guidance to physicians, advanced practice providers (APPs), pharmacists, and nurses regarding medication management in the preoperative setting. Background: Appropriate perioperative medication management is essential to ensure positive surgical outcomes and prevent medication misadventures.1 Results from a prospective analysis of 1,025 patients admitted to a general surgical unit concluded that patients on at least one medication for a chronic disease are 2.7 times more likely to experience surgical complications compared with those not taking any medications. As the aging population requires more medication use and the availability of various nonprescription medications continues to increase, so does the risk of polypharmacy and the need for perioperative medication guidance.2 There are no well-designed trials to support evidence-based recommendations for perioperative medication management; however, general principles and best practice approaches are available. General considerations for perioperative medication management include a thorough medication history, understanding of the medication pharmacokinetics and potential for withdrawal symptoms, understanding the risks associated with the surgical procedure and the risks of medication discontinuation based on the intended indication. Clinical judgement must be exercised, especially if medication pharmacokinetics are not predictable or there are significant risks associated with inappropriate medication withdrawal (eg, tolerance) or continuation (eg, postsurgical infection).2 Clinical Assessment: Prior to instructing the patient on preoperative medication management, completion of a thorough medication history is recommended – including all information on prescription medications, over-the-counter medications, “as needed” medications, vitamins, supplements, and herbal medications. Allergies should also be verified and documented. -

Second Generation Antiarrhythmic Agents: Have We Reached Antiarrhythmic Nirvana?
JACC Vol. 9. NO.2 459 February 1987:459-63 EDITORIAL REVIEWS Second Generation Antiarrhythmic Agents: Have We Reached Antiarrhythmic Nirvana? LEONARD N. HOROWITZ, MD, FACC, JOEL MORGANROTH, MD, FACC Philadelphia, Pennsylvania During the first half of the 20th century, antiarrhythmic some cases novel, but because their preapproval evaluations therapy was principally directed toward supraventricular ar followed a more rigorous and circumspect path. More in rhythmia; only relatively recently has therapy of ventricular formation is available to us so we can decide when and how arrhythmias been emphasized. In fact, significant pharma these agents should be employed. cologic treatment of ventricular arrhythmias was minimal Like all antiarrhythmic drugs of the first generation, the until routine use of quinidine and procainamide began in new agents have the potential to provoke or worsen ven the 1950s (1,2). The first generation oral antiarrhythmic tricular arrhythmias. These proarrhythmic effects vary in drugs, quinidine, procainamide and disopyramide, previ incidence and may present a major problem in certain patient ously constituted the principal antiarrhythmic agents for long groups such as those with sustained ventricular tachyar term treatment of ventricular arrhythmias in the United States. rhythrnias, reduced left ventricular function and conduction Compared with modem regulatory standards, the data on disturbances. In common with other antiarrhythmic drugs, which the use of these drugs was based were modest at best the second generation drugs have not been shown to prevent and rudimentary at worst. However, because good clinical sudden death in patients with ventricular ectopic activity. judgment compensates for a multitude of deficiencies, we These factors, along with efficacy and potential toxicity, have been able to provide effective antiarrhythmic therapy must be considered in selecting antiarrhythmic regimens for to many patients with this rather limited pharmacopoeia. -

Ventricular Tachycardia Drugs Versus Devices John Camm St
Cardiology Update 2015 Davos, Switzerland: 8-12th February 2015 Ventricular Arrhythmias Ventricular Tachycardia Drugs versus Devices John Camm St. George’s University of London, UK Imperial College, London, UK Declaration of Interests Chairman: NICE Guidelines on AF, 2006; ESC Guidelines on Atrial Fibrillation, 2010 and Update, 2012; ACC/AHA/ESC Guidelines on VAs and SCD; 2006; NICE Guidelines on ACS and NSTEMI, 2012; NICE Guidelines on heart failure, 2008; NICE Guidelines on Atrial Fibrillation, 2006; ESC VA and SCD Guidelines, 2015 Steering Committees: multiple trials including novel anticoagulants DSMBs: multiple trials including BEAUTIFUL, SHIFT, SIGNIFY, AVERROES, CASTLE- AF, STAR-AF II, INOVATE, and others Events Committees: one trial of novel oral anticoagulants and multiple trials of miscellaneous agents with CV adverse effects Editorial Role: Editor-in-Chief, EP-Europace and Clinical Cardiology; Editor, European Textbook of Cardiology, European Heart Journal, Electrophysiology of the Heart, and Evidence Based Cardiology Consultant/Advisor/Speaker: Astellas, Astra Zeneca, ChanRX, Gilead, Merck, Menarini, Otsuka, Sanofi, Servier, Xention, Bayer, Boehringer Ingelheim, Bristol- Myers Squibb, Daiichi Sankyo, Pfizer, Boston Scientific, Biotronik, Medtronic, St. Jude Medical, Actelion, GlaxoSmithKline, InfoBionic, Incarda, Johnson and Johnson, Mitsubishi, Novartis, Takeda Therapy for Ventricular Tachycardia Medical therapy Antiarrhythmic drugs Autonomic management Ventricular tachycardia Monomorphic Polymorphic Ventricular fibrillation Ventricular storms Ablation therapy Device therapy Surgical Defibrillation Catheter Antitachycardia pacing History of Antiarrhythmic Drugs 1914 - Quinidine 1950 - Lidocaine 1951 - Procainamide 1946 – Digitalis 1956 – Ajmaline 1962 - Verapamil 1962 – Disopyramide 1964 - Propranolol 1967 – Amiodarone 1965 – Bretylium 1972 – Mexiletine 1973 – Aprindine, Tocainide 1969 - Diltiazem 1975- Flecainide 1976 – Propafenone Encainide Ethmozine 2000 - Sotalol D-sotalol 1995 - Ibutilide (US) Recainam 2000 – Dofetilide US) IndecainideX Etc. -

Drug Interaction of Amiodarone, Flecainide, Metoprolol and Diltiazem Leading to Heart Block Vijay Kahajuria, Sanjeev Gupta, Roshi, Neelam Rani
JK SCIENCE CASE REPORT Drug Interaction of Amiodarone, Flecainide, Metoprolol and Diltiazem leading to Heart Block Vijay Kahajuria, Sanjeev Gupta, Roshi, Neelam Rani Abstract Amiodarone, flecainide, metoprolol and diltiazem individually are known to cause heart blocks due to their cardiac depressant property but the current case report is worth reporting because it resulted because of drug interaction of multiple drugs due to possible medication error. Key Words Amiodarone, Flecainide, Metoprolol, Diltiazem, Heart Blocks Introduction Adverse Drug Reactions (ADRs) in cardiology are thyroid dysfunction. His hemoglobin levels were 13g/dl, common. There are numerous reports of drug induced total leucocyte count 6000/UL, neutrophils 56.5%, bradycardia and heart block (1). Co-morbid conditions lymphocytes 30.5%, eosinophils 5.2%, monocytes often coexist with cardiac ailments and results in 7.6%,basophils 0.2%, platelet count 1.5 lacs, serum urea- polypharmacy which enhance the chances of drug 33mg/dl, serum creatinine 0.90mg/dl, prothrombin time interactions culminating to adverse events. In the current 10.9 sec ,INR 1.04,hepatitis serology was non reactive, study report patient was prescribed multiple drugs for blood sugar random 182 mg/dl, TSH 1.4uIU/ml, serum cardiac arrhythmia and he presented with heart block sodium 142 mmol/L,serum potassium 5 mmol/L. and bradycardia. Though amiodarone, flecainide, He was prescribed tab.atenolol 20mg and metoprolol and diltiazem individually are known to cause tab.procainamide 20mg which did not bring any relief. heart blocks due to their cardiac depressant property but So radiofrequency ablation was done which was the current case report is worth reporting because it uneventful. -

Flecainide in Ventricular Arrhythmias: from Old Myths to New Perspectives
Journal of Clinical Medicine Review Flecainide in Ventricular Arrhythmias: From Old Myths to New Perspectives Carlo Lavalle 1,*, Sara Trivigno 1 , Giampaolo Vetta 1 , Michele Magnocavallo 1 , Marco Valerio Mariani 1 , Luca Santini 2, Giovanni Battista Forleo 3 , Massimo Grimaldi 4, Roberto Badagliacca 1, Luigi Lanata 5 and Renato Pietro Ricci 6 1 Department of Cardiovascular, Respiratory, Nephrological, Anesthesiological and Geriatric Sciences, Sapienza University of Rome, Policlinico Umberto I, 00161 Rome, Italy; [email protected] (S.T.); [email protected] (G.V.); [email protected] (M.M.); [email protected] (M.V.M.); [email protected] (R.B.) 2 Department of Cardiology, Ospedale GB Grassi, 00121 Ostia, Italy; [email protected] 3 Department of Cardiology, Azienda Ospedaliera-Universitaria “Luigi Sacco”, 20057 Milan, Italy; [email protected] 4 Department of Cardiology, Ospedale Generale Regionale F. Miulli, Acquaviva delle Fonti, 70021 Bari, Italy; fi[email protected] 5 Medical Affairs Department, Dompé Farmaceutici SpA, 20057 Milan, Italy; [email protected] 6 Centro Cardio-Aritmologico, 00152 Rome, Italy; [email protected] * Correspondence: [email protected]; Tel.: +39-335376901 Abstract: Flecainide is an IC antiarrhythmic drug (AAD) that received in 1984 Food and Drug Administration approval for the treatment of sustained ventricular tachycardia (VT) and subsequently Citation: Lavalle, C.; Trivigno, S.; for rhythm control of atrial fibrillation (AF). Currently, flecainide is mainly employed for sinus Vetta, G.; Magnocavallo, M.; Mariani, rhythm maintenance in AF and the treatment of idiopathic ventricular arrhythmias (IVA) in absence M.V.; Santini, L.; Forleo, G.B.; Grimaldi, M.; Badagliacca, R.; Lanata, of ischaemic and structural heart disease on the basis of CAST data. -

FLECAINIDE ACETATE Tablets, USP Rx Only DESCRIPTION CLINICAL PHARMACOLOGY
FLECAINIDE ACETATE Tablets, USP Rx only DESCRIPTION Flecainide is an antiarrhythmic drug available in tablets of 50, 100, or 150 mg for oral administration. Flecainide acetate is benzamide, N-(2-piperidinylmethyl)-2,5-bis (2,2,2-trifluoroethoxy)- monoacetate. The structural formula is given below. Flecainide acetate is a white crystalline substance with a pKa of 9.3. It has an aqueous solubility of 48.4 mg/mL at 37°C. Flecainide Acetate Tablets, USP also contain: croscarmellose sodium, hydrogenated vegetable oil, magnesium stearate, microcrystalline cellulose, and pregelatinized starch. CLINICAL PHARMACOLOGY Flecainide has local anesthetic activity and belongs to the membrane stabilizing (Class 1) group of antiarrhythmic agents; it has electrophysiologic effects characteristic of the IC class of antiarrhythmics. Electrophysiology In man, flecainide produces a dose-related decrease in intracardiac conduction in all parts of the heart with the greatest effect on the His-Purkinje system (H-V conduction). Effects upon atrioventricular (AV) nodal conduction time and intra-atrial conduction times, although present, are less pronounced than those on ventricular conduction velocity. Significant effects on refractory periods were observed only in the ventricle. Sinus node recovery times (corrected) following pacing and spontaneous cycle lengths are somewhat increased. This latter effect may become significant in patients with sinus node dysfunction. (See WARNINGS.) Flecainide causes a dose-related and plasma-level related decrease in single and multiple PVCs and can suppress recurrence of ventricular tachycardia. In limited studies of patients with a history of ventricular tachycardia, flecainide has been successful 30 to 40% of the time in fully suppressing the inducibility of arrhythmias by programmed electrical stimulation. -

Draft for Public Comment
Draft for Public Comment Practice guideline update: Treatment of painful diabetic polyneuropathy Report of the Guideline Subcommittee of the American Academy of Neurology Authors (Alphabetical order—final order to be determined later in the guideline development process) Carmel Armon, MD, MSc, MHS1,Vera Bril, MD2, Brian C. Callaghan, MD, MS3, Lindsay Colbert, MA4, William S. David, MD, PhD5, Mary Dolan O’Brien, MLIS6, Kenneth Fink, MD, MPH7, Gary Franklin, MD, MPH8, Gary Gronseth, MD9, John Halperin, MD10, Lawrence B. Harkless, DPM11, Leslie Levine, PhD, VMD, JD12, Nicole Licking, DO13, Bruce A. Perkins, MD, MPH14, Michael Pignone, MD15, Raymond Price, MD16, Alexander Rae-Grant, MD17, Don Smith, MD18, Scott R. Wessels, MPS, ELS6 1. Department of Neurology, Tel Aviv University Sackler School of Medicine, Israel 2. Division of Neurology, Department of Medicine, Toronto General Hospital, ON, Canada 3. Department of Neurology, University of Michigan, Ann Arbor 4. The Foundation for Peripheral Neuropathy, Buffalo Grove, IL 5. Department of Neurology, Massachusetts General Hospital, Boston 6. American Academy of Neurology, Minneapolis, MN 7. Kamehameha Schools, Honolulu, HI 8. Department of Neurology, University of Washington, Seattle, WA 9. Department of Neurology, University of Kansas Medical Center, Kansas City 10. Department of Neurosciences, Overlook Medical Center, Summit, NJ 11. Western University Health Sciences College of Podiatric Medicine, Pomona, CA 1 Draft for Public Comment 12. Neuropathy Action Foundation, Santa Ana, CA 13. New West Physicians, Golden, CO 14. Leadership Sinai Centre for Diabetes, Mount Sinai Hospital, Toronto, ON 15. Department of Medicine, The University of Texas at Austin 16. Department of Neurology, University of Pennsylvania, Philadelphia 17. -

Oral Antiarrhythmic Drugs in Converting Recent Onset Atrial Fibrillation
Review article Oral antiarrhythmic drugs in converting recent onset atrial fibrillation • Vera H.M. Deneer, Marieke B.I. Borgh, J. Herre Kingma, Loraine Lie-A-Huen and Jacobus R.B.J. Brouwers Introduction Pharm World Sci 2004; 26: 66–78. © 2004 Kluwer Academic Publishers. Printed in the Netherlands. Atrial fibrillation is the most common arrhythmia. The incidence of atrial fibrillation depends on the age of V.H.M. Deneer (correspondence, e-mail: the study population. The incidence varies between 2 [email protected]), M.B.I. Borgh, L. Lie-A-Huen: Department of Clinical Pharmacy or 3 new cases per 1,000 population per year between J.H. Kingma: Department of Cardiology, St Antonius Hospital, the ages of 55 and 64 years to 35 new cases per 1,000 Koekoekslaan 1, 3435 CM Nieuwegein, The Netherlands population per year between the ages of 85 and 94 J.R.B.J. Brouwers: Groningen University Institute for Drug 1 Exploration (GUIDE), Section of Pharmacotherapy, University years . Treatment of an episode of paroxysmal atrial fi- of Groningen, Antonius Deusinglaan 1, 9713 AV Groningen, brillation consists of restoring sinus rhythm by DC- The Netherlands electrical cardioversion or by the intravenous adminis- Key words tration of an antiarrhythmic drug, but frequently the Atrial fibrillation arrhythmia spontaneously terminates1–3. After one or Amiodarone more episodes of atrial fibrillation chronic prophylac- Antiarrhythmic drugs Digoxin tic treatment with an antiarrhythmic drug is often Episodic treatment started for maintenance of sinus rhythm4–8. Another Flecainide treatment strategy consists of allowing the arrhythmia Propafenone Quinidine to exist in combination with pharmacological ven- Sotalol tricular rate control. -

Voltage-Gated Sodium Channels and Blockers: an Overview and Where Will They Go?*
Current Medical Science 39(6):863-873,2019 DOICurrent https://doi.org/10.1007/s11596-019-2117-0 Medical Science 39(6):2019 863 Voltage-gated Sodium Channels and Blockers: An Overview and Where Will They Go?* Zhi-mei LI1, Li-xia CHEN2#, Hua LI1# 1Hubei Key Laboratory of Natural Medicinal Chemistry and Resource Evaluation, School of Pharmacy, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430030, China 2Wuya College of Innovation, Key Laboratory of Structure-Based Drug Design & Discovery, Ministry of Education, Shenyang Pharmaceutical University, Shenyang 110016, China Huazhong University of Science and Technology 2019 Summary: Voltage-gated sodium (Nav) channels are critical players in the generation and propagation of action potentials by triggering membrane depolarization. Mutations in Nav channels are associated with a variety of channelopathies, which makes them relevant targets for pharmaceutical intervention. So far, the cryoelectron microscopic structure of the human Nav1.2, Nav1.4, and Nav1.7 has been reported, which sheds light on the molecular basis of functional mechanism of Nav channels and provides a path toward structure-based drug discovery. In this review, we focus on the recent advances in the structure, molecular mechanism and modulation of Nav channels, and state updated sodium channel blockers for the treatment of pathophysiology disorders and briefly discuss where the blockers may be developed in the future. Key words: voltage-gated sodium channels; blockers; Nav channel structures; channelopathies Life did not come into existence until living In this review, we focus on voltage-gated organisms were enclosed by one or more membranes sodium (Nav) channels, which selectively conduct which cut them off from the chaotic world at a sodium ions movement in response to variations of molecular level. -

2020 Aetna Standard Plan
Plan for your best health Aetna Standard Plan Aetna.com Aetna is the brand name used for products and services provided by one or more of the Aetna group of subsidiary companies, including Aetna Life Insurance Company and its affiliates (Aetna). Aetna Pharmacy Management refers to an internal business unit of Aetna Health Management, LLC. Aetna Pharmacy Management administers, but does not offer, insure or otherwise underwrite the prescription drug benefits portion of your health plan and has no financial responsibility therefor. 2020 Pharmacy Drug Guide - Aetna Standard Plan Table of Contents INFORMATIONAL SECTION..................................................................................................................6 *ADHD/ANTI-NARCOLEPSY/ANTI-OBESITY/ANOREXIANTS* - DRUGS FOR THE NERVOUS SYSTEM.................................................................................................................................16 *ALLERGENIC EXTRACTS/BIOLOGICALS MISC* - BIOLOGICAL AGENTS...............................18 *ALTERNATIVE MEDICINES* - VITAMINS AND MINERALS....................................................... 19 *AMEBICIDES* - DRUGS FOR INFECTIONS.....................................................................................19 *AMINOGLYCOSIDES* - DRUGS FOR INFECTIONS.......................................................................19 *ANALGESICS - ANTI-INFLAMMATORY* - DRUGS FOR PAIN AND FEVER............................19 *ANALGESICS - NONNARCOTIC* - DRUGS FOR PAIN AND FEVER.........................................