Making Comparisons
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Descriptive Statistics (Part 2): Interpreting Study Results
Statistical Notes II Descriptive statistics (Part 2): Interpreting study results A Cook and A Sheikh he previous paper in this series looked at ‘baseline’. Investigations of treatment effects can be descriptive statistics, showing how to use and made in similar fashion by comparisons of disease T interpret fundamental measures such as the probability in treated and untreated patients. mean and standard deviation. Here we continue with descriptive statistics, looking at measures more The relative risk (RR), also sometimes known as specific to medical research. We start by defining the risk ratio, compares the risk of exposed and risk and odds, the two basic measures of disease unexposed subjects, while the odds ratio (OR) probability. Then we show how the effect of a disease compares odds. A relative risk or odds ratio greater risk factor, or a treatment, can be measured using the than one indicates an exposure to be harmful, while relative risk or the odds ratio. Finally we discuss the a value less than one indicates a protective effect. ‘number needed to treat’, a measure derived from the RR = 1.2 means exposed people are 20% more likely relative risk, which has gained popularity because of to be diseased, RR = 1.4 means 40% more likely. its clinical usefulness. Examples from the literature OR = 1.2 means that the odds of disease is 20% higher are used to illustrate important concepts. in exposed people. RISK AND ODDS Among workers at factory two (‘exposed’ workers) The probability of an individual becoming diseased the risk is 13 / 116 = 0.11, compared to an ‘unexposed’ is the risk. -

Challenging Issues in Clinical Trial Design: Part 4 of a 4-Part Series on Statistics for Clinical Trials
Challenging Issues in Clinical Trial Design: Part 4 of a 4-part Series on Statistics for Clinical Trials Brief title: Challenges in Trial Design Stuart J. Pocock, PHD,* Tim C. Clayton, MSC,* Gregg W. Stone, MD† From the: *London School of Hygiene and Tropical Medicine, London, United Kingdom; †Columbia University Medical Center, New York-Presbyterian Hospital and the Cardiovascular Research Foundation, New York, New York <COR> Reprint requests and correspondence: Prof. Stuart J. Pocock, Department of Medical Statistics, London School of Hygiene and Tropical Medicine, Keppel Street, London, WC1E 7HT, United Kingdom Telephone: +44 20 7927 2413 Fax: +44 20 7637 2853 E-mail: [email protected] Disclosures: The authors declare no conflicts of interest for this paper. 1 Abstract As a sequel to last week’s article on the fundamentals of clinical trial design, this article tackles related controversial issues: noninferiority trials; the value of factorial designs; the importance and challenges of strategy trials; Data Monitoring Committees (including when to stop a trial early); and the role of adaptive designs. All topics are illustrated by relevant examples from cardiology trials. <KW>Key words: Noninferiority trials; Factorial designs; Strategy trials, Data Monitoring Committees; Statistical stopping guidelines; Adaptive designs; Randomized Controlled Trials As Topic; Abbreviations ACS = acute coronary syndrome CABG = coronary artery bypass graft CI = confidence interval CV = cardiovascular DMC = Data Monitoring Committee FDA = Food and Drug Administration MACE = major adverse cardiovascular event OMT = optimal medical therapy PCI = percutaneous coronary intervention 2 Introduction Randomized controlled trials are the cornerstone of clinical guidelines informing best therapeutic practices, however their design and interpretation may be complex and nuanced. -

Meta-Analysis of Proportions
NCSS Statistical Software NCSS.com Chapter 456 Meta-Analysis of Proportions Introduction This module performs a meta-analysis of a set of two-group, binary-event studies. These studies have a treatment group (arm) and a control group. The results of each study may be summarized as counts in a 2-by-2 table. The program provides a complete set of numeric reports and plots to allow the investigation and presentation of the studies. The plots include the forest plot, radial plot, and L’Abbe plot. Both fixed-, and random-, effects models are available for analysis. Meta-Analysis refers to methods for the systematic review of a set of individual studies with the aim to combine their results. Meta-analysis has become popular for a number of reasons: 1. The adoption of evidence-based medicine which requires that all reliable information is considered. 2. The desire to avoid narrative reviews which are often misleading. 3. The desire to interpret the large number of studies that may have been conducted about a specific treatment. 4. The desire to increase the statistical power of the results be combining many small-size studies. The goals of meta-analysis may be summarized as follows. A meta-analysis seeks to systematically review all pertinent evidence, provide quantitative summaries, integrate results across studies, and provide an overall interpretation of these studies. We have found many books and articles on meta-analysis. In this chapter, we briefly summarize the information in Sutton et al (2000) and Thompson (1998). Refer to those sources for more details about how to conduct a meta- analysis. -

Limitations of the Odds Ratio in Gauging the Performance of a Diagnostic, Prognostic, Or Screening Marker
American Journal of Epidemiology Vol. 159, No. 9 Copyright © 2004 by the Johns Hopkins Bloomberg School of Public Health Printed in U.S.A. All rights reserved DOI: 10.1093/aje/kwh101 Limitations of the Odds Ratio in Gauging the Performance of a Diagnostic, Prognostic, or Screening Marker Margaret Sullivan Pepe1,2, Holly Janes2, Gary Longton1, Wendy Leisenring1,2,3, and Polly Newcomb1 1 Division of Public Health Sciences, Fred Hutchinson Cancer Research Center, Seattle, WA. 2 Department of Biostatistics, University of Washington, Seattle, WA. 3 Division of Clinical Research, Fred Hutchinson Cancer Research Center, Seattle, WA. Downloaded from Received for publication June 24, 2003; accepted for publication October 28, 2003. A marker strongly associated with outcome (or disease) is often assumed to be effective for classifying persons http://aje.oxfordjournals.org/ according to their current or future outcome. However, for this assumption to be true, the associated odds ratio must be of a magnitude rarely seen in epidemiologic studies. In this paper, an illustration of the relation between odds ratios and receiver operating characteristic curves shows, for example, that a marker with an odds ratio of as high as 3 is in fact a very poor classification tool. If a marker identifies 10% of controls as positive (false positives) and has an odds ratio of 3, then it will correctly identify only 25% of cases as positive (true positives). The authors illustrate that a single measure of association such as an odds ratio does not meaningfully describe a marker’s ability to classify subjects. Appropriate statistical methods for assessing and reporting the classification power of a marker are described. -

Guidance for Industry E2E Pharmacovigilance Planning
Guidance for Industry E2E Pharmacovigilance Planning U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center for Biologics Evaluation and Research (CBER) April 2005 ICH Guidance for Industry E2E Pharmacovigilance Planning Additional copies are available from: Office of Training and Communication Division of Drug Information, HFD-240 Center for Drug Evaluation and Research Food and Drug Administration 5600 Fishers Lane Rockville, MD 20857 (Tel) 301-827-4573 http://www.fda.gov/cder/guidance/index.htm Office of Communication, Training and Manufacturers Assistance, HFM-40 Center for Biologics Evaluation and Research Food and Drug Administration 1401 Rockville Pike, Rockville, MD 20852-1448 http://www.fda.gov/cber/guidelines.htm. (Tel) Voice Information System at 800-835-4709 or 301-827-1800 U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center for Biologics Evaluation and Research (CBER) April 2005 ICH Contains Nonbinding Recommendations TABLE OF CONTENTS I. INTRODUCTION (1, 1.1) ................................................................................................... 1 A. Background (1.2) ..................................................................................................................2 B. Scope of the Guidance (1.3)...................................................................................................2 II. SAFETY SPECIFICATION (2) ..................................................................................... -

Survival Analysis: Part I — Analysis Korean Journal of Anesthesiology of Time-To-Event
Statistical Round pISSN 2005-6419 • eISSN 2005-7563 KJA Survival analysis: Part I — analysis Korean Journal of Anesthesiology of time-to-event Junyong In1 and Dong Kyu Lee2 Department of Anesthesiology and Pain Medicine, 1Dongguk University Ilsan Hospital, Goyang, 2Guro Hospital, Korea University School of Medicine, Seoul, Korea Length of time is a variable often encountered during data analysis. Survival analysis provides simple, intuitive results concerning time-to-event for events of interest, which are not confined to death. This review introduces methods of ana- lyzing time-to-event. The Kaplan-Meier survival analysis, log-rank test, and Cox proportional hazards regression model- ing method are described with examples of hypothetical data. Keywords: Censored data; Cox regression; Hazard ratio; Kaplan-Meier method; Log-rank test; Medical statistics; Power analysis; Proportional hazards; Sample size; Survival analysis. Introduction mation of the time [2]. The Korean Journal of Anesthesiology has thus far published In a clinical trial or clinical study, an investigational treat- several papers using survival analysis for clinical outcomes: a ment is administered to subjects, and the resulting outcome data comparison of 90-day survival rate for pressure ulcer develop- are collected and analyzed after a certain period of time. Most ment or non-development after surgery under general anesthesia statistical analysis methods do not include the length of time [3], a comparison of postoperative 5-year recurrence rate in as a variable, and analysis is made only on the trial outcomes patients with breast cancer depending on the type of anesthetic upon completion of the study period, as specified in the study agent used [4], and a comparison of airway intubation success protocol. -
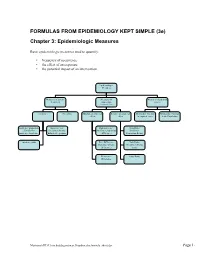
FORMULAS from EPIDEMIOLOGY KEPT SIMPLE (3E) Chapter 3: Epidemiologic Measures
FORMULAS FROM EPIDEMIOLOGY KEPT SIMPLE (3e) Chapter 3: Epidemiologic Measures Basic epidemiologic measures used to quantify: • frequency of occurrence • the effect of an exposure • the potential impact of an intervention. Epidemiologic Measures Measures of disease Measures of Measures of potential frequency association impact (“Measures of Effect”) Incidence Prevalence Absolute measures of Relative measures of Attributable Fraction Attributable Fraction effect effect in exposed cases in the Population Incidence proportion Incidence rate Risk difference Risk Ratio (Cumulative (incidence density, (Incidence proportion (Incidence Incidence, Incidence hazard rate, person- difference) Proportion Ratio) Risk) time rate) Incidence odds Rate Difference Rate Ratio (Incidence density (Incidence density difference) ratio) Prevalence Odds Ratio Difference Macintosh HD:Users:buddygerstman:Dropbox:eks:formula_sheet.doc Page 1 of 7 3.1 Measures of Disease Frequency No. of onsets Incidence Proportion = No. at risk at beginning of follow-up • Also called risk, average risk, and cumulative incidence. • Can be measured in cohorts (closed populations) only. • Requires follow-up of individuals. No. of onsets Incidence Rate = ∑person-time • Also called incidence density and average hazard. • When disease is rare (incidence proportion < 5%), incidence rate ≈ incidence proportion. • In cohorts (closed populations), it is best to sum individual person-time longitudinally. It can also be estimated as Σperson-time ≈ (average population size) × (duration of follow-up). Actuarial adjustments may be needed when the disease outcome is not rare. • In an open populations, Σperson-time ≈ (average population size) × (duration of follow-up). Examples of incidence rates in open populations include: births Crude birth rate (per m) = × m mid-year population size deaths Crude mortality rate (per m) = × m mid-year population size deaths < 1 year of age Infant mortality rate (per m) = × m live births No. -

Relative Risks, Odds Ratios and Cluster Randomised Trials . (1)
1 Relative Risks, Odds Ratios and Cluster Randomised Trials Michael J Campbell, Suzanne Mason, Jon Nicholl Abstract It is well known in cluster randomised trials with a binary outcome and a logistic link that the population average model and cluster specific model estimate different population parameters for the odds ratio. However, it is less well appreciated that for a log link, the population parameters are the same and a log link leads to a relative risk. This suggests that for a prospective cluster randomised trial the relative risk is easier to interpret. Commonly the odds ratio and the relative risk have similar values and are interpreted similarly. However, when the incidence of events is high they can differ quite markedly, and it is unclear which is the better parameter . We explore these issues in a cluster randomised trial, the Paramedic Practitioner Older People’s Support Trial3 . Key words: Cluster randomized trials, odds ratio, relative risk, population average models, random effects models 1 Introduction Given two groups, with probabilities of binary outcomes of π0 and π1 , the Odds Ratio (OR) of outcome in group 1 relative to group 0 is related to Relative Risk (RR) by: . If there are covariates in the model , one has to estimate π0 at some covariate combination. One can see from this relationship that if the relative risk is small, or the probability of outcome π0 is small then the odds ratio and the relative risk will be close. One can also show, using the delta method, that approximately (1). ________________________________________________________________________________ Michael J Campbell, Medical Statistics Group, ScHARR, Regent Court, 30 Regent St, Sheffield S1 4DA [email protected] 2 Note that this result is derived for non-clustered data. -

CHAPTER 3 Comparing Disease Frequencies
© Smartboy10/DigitalVision Vectors/Getty Images CHAPTER 3 Comparing Disease Frequencies LEARNING OBJECTIVES By the end of this chapter the reader will be able to: ■ Organize disease frequency data into a two-by-two table. ■ Describe and calculate absolute and relative measures of comparison, including rate/risk difference, population rate/risk difference, attributable proportion among the exposed and the total population and rate/risk ratio. ■ Verbally interpret each absolute and relative measure of comparison. ■ Describe the purpose of standardization and calculate directly standardized rates. ▸ Introduction Measures of disease frequency are the building blocks epidemiologists use to assess the effects of a disease on a population. Comparing measures of disease frequency organizes these building blocks in a meaningful way that allows one to describe the relationship between a characteristic and a disease and to assess the public health effect of the exposure. Disease frequencies can be compared between different populations or between subgroups within a population. For example, one might be interested in comparing disease frequencies between residents of France and the United States or between subgroups within the U.S. population according to demographic characteristics, such as race, gender, or socio- economic status; personal habits, such as alcoholic beverage consump- tion or cigarette smoking; and environmental factors, such as the level of air pollution. For example, one might compare incidence rates of coronary heart disease between residents of France and those of the United States or 57 58 Chapter 3 Comparing Disease Frequencies among U.S. men and women, Blacks and Whites, alcohol drinkers and nondrinkers, and areas with high and low pollution levels. -

Understanding Relative Risk, Odds Ratio, and Related Terms: As Simple As It Can Get Chittaranjan Andrade, MD
Understanding Relative Risk, Odds Ratio, and Related Terms: As Simple as It Can Get Chittaranjan Andrade, MD Each month in his online Introduction column, Dr Andrade Many research papers present findings as odds ratios (ORs) and considers theoretical and relative risks (RRs) as measures of effect size for categorical outcomes. practical ideas in clinical Whereas these and related terms have been well explained in many psychopharmacology articles,1–5 this article presents a version, with examples, that is meant with a view to update the knowledge and skills to be both simple and practical. Readers may note that the explanations of medical practitioners and examples provided apply mostly to randomized controlled trials who treat patients with (RCTs), cohort studies, and case-control studies. Nevertheless, similar psychiatric conditions. principles operate when these concepts are applied in epidemiologic Department of Psychopharmacology, National Institute research. Whereas the terms may be applied slightly differently in of Mental Health and Neurosciences, Bangalore, India different explanatory texts, the general principles are the same. ([email protected]). ABSTRACT Clinical Situation Risk, and related measures of effect size (for Consider a hypothetical RCT in which 76 depressed patients were categorical outcomes) such as relative risks and randomly assigned to receive either venlafaxine (n = 40) or placebo odds ratios, are frequently presented in research (n = 36) for 8 weeks. During the trial, new-onset sexual dysfunction articles. Not all readers know how these statistics was identified in 8 patients treated with venlafaxine and in 3 patients are derived and interpreted, nor are all readers treated with placebo. These results are presented in Table 1. -

Outcome Reporting Bias in COVID-19 Mrna Vaccine Clinical Trials
medicina Perspective Outcome Reporting Bias in COVID-19 mRNA Vaccine Clinical Trials Ronald B. Brown School of Public Health and Health Systems, University of Waterloo, Waterloo, ON N2L3G1, Canada; [email protected] Abstract: Relative risk reduction and absolute risk reduction measures in the evaluation of clinical trial data are poorly understood by health professionals and the public. The absence of reported absolute risk reduction in COVID-19 vaccine clinical trials can lead to outcome reporting bias that affects the interpretation of vaccine efficacy. The present article uses clinical epidemiologic tools to critically appraise reports of efficacy in Pfzier/BioNTech and Moderna COVID-19 mRNA vaccine clinical trials. Based on data reported by the manufacturer for Pfzier/BioNTech vaccine BNT162b2, this critical appraisal shows: relative risk reduction, 95.1%; 95% CI, 90.0% to 97.6%; p = 0.016; absolute risk reduction, 0.7%; 95% CI, 0.59% to 0.83%; p < 0.000. For the Moderna vaccine mRNA-1273, the appraisal shows: relative risk reduction, 94.1%; 95% CI, 89.1% to 96.8%; p = 0.004; absolute risk reduction, 1.1%; 95% CI, 0.97% to 1.32%; p < 0.000. Unreported absolute risk reduction measures of 0.7% and 1.1% for the Pfzier/BioNTech and Moderna vaccines, respectively, are very much lower than the reported relative risk reduction measures. Reporting absolute risk reduction measures is essential to prevent outcome reporting bias in evaluation of COVID-19 vaccine efficacy. Keywords: mRNA vaccine; COVID-19 vaccine; vaccine efficacy; relative risk reduction; absolute risk reduction; number needed to vaccinate; outcome reporting bias; clinical epidemiology; critical appraisal; evidence-based medicine Citation: Brown, R.B. -

Delineating Virulence of Vibrio Campbellii
www.nature.com/scientificreports OPEN Delineating virulence of Vibrio campbellii: a predominant luminescent bacterial pathogen in Indian shrimp hatcheries Sujeet Kumar1*, Chandra Bhushan Kumar1,2, Vidya Rajendran1, Nishawlini Abishaw1, P. S. Shyne Anand1, S. Kannapan1, Viswas K. Nagaleekar3, K. K. Vijayan1 & S. V. Alavandi1 Luminescent vibriosis is a major bacterial disease in shrimp hatcheries and causes up to 100% mortality in larval stages of penaeid shrimps. We investigated the virulence factors and genetic identity of 29 luminescent Vibrio isolates from Indian shrimp hatcheries and farms, which were earlier presumed as Vibrio harveyi. Haemolysin gene-based species-specifc multiplex PCR and phylogenetic analysis of rpoD and toxR identifed all the isolates as V. campbellii. The gene-specifc PCR revealed the presence of virulence markers involved in quorum sensing (luxM, luxS, cqsA), motility (faA, lafA), toxin (hly, chiA, serine protease, metalloprotease), and virulence regulators (toxR, luxR) in all the isolates. The deduced amino acid sequence analysis of virulence regulator ToxR suggested four variants, namely A123Q150 (AQ; 18.9%), P123Q150 (PQ; 54.1%), A123P150 (AP; 21.6%), and P123P150 (PP; 5.4% isolates) based on amino acid at 123rd (proline or alanine) and 150th (glutamine or proline) positions. A signifcantly higher level of the quorum-sensing signal, autoinducer-2 (AI-2, p = 2.2e−12), and signifcantly reduced protease activity (p = 1.6e−07) were recorded in AP variant, whereas an inverse trend was noticed in the Q150 variants AQ and PQ. The pathogenicity study in Penaeus (Litopenaeus) vannamei juveniles revealed that all the isolates of AQ were highly pathogenic with Cox proportional hazard ratio 15.1 to 32.4 compared to P150 variants; PP (5.4 to 6.3) or AP (7.3 to 14).