Mass Spectrometry—The Early Years
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Cathode Rays 89 Cathode Rays
Cathode Rays 89 Cathode Rays Theodore Arabatzis C The detection of cathode rays was a by-product of the investigation of the discharge of electricity through rarefied gases. The latter phenomenon had been studied since the early eighteenth century. By the middle of the nineteenth century it was known that the passage of electricity through a partly evacuated tube produced a glow in the gas, whose color depended on its chemical composition and its pressure. Below a certain pressure the glow assumed a stratified pattern of bright and dark bands. During the second half of the nineteenth century the discharge of electricity through gases became a topic of intense exploratory experimentation, primarily in Germany [21]. In 1855 the German instrument maker Heinrich Geißler (1815– 1879) manufactured improved vacuum tubes, which made possible the isolation and investigation of cathode rays [23]. In 1857 Geissler’s tubes were employed by Julius Pl¨ucker (1801–1868) to study the influence of a magnet on the electrical dis- charge. He observed various complex and striking phenomena associated with the discharge. Among those phenomena were a “light which appears about the negative electrode” and a fluorescence in the glass of the tube ([9], pp. 122, 130). The understanding of those phenomena was advanced by Pl¨ucker’s student and collaborator, Johann Wilhelm Hittorf (1824–1914), who observed that “if any ob- ject is interposed in the space filled with glow-light [emanating from the negative electrode], it throws a sharp shadow on the fluorescent side” ([5], p. 117). This effect implied that the “rays” emanating from the cathode followed a straight path. -

Almost Forgotten Anniversaries in 2019 Introduction
Almost Forgotten Anniversaries in 2019 Katharina Lodders Department of Earth and Planetary Sciences and Mc Donnell Center for the Space Sciences, Campus Box 1169, Washington University, Saint Louis MO 63130, USA Keywords: history, chemical elements, abundances Abstract: As we celebrate the International Year of the Periodic Table, the 50th anniversary of Apollo 11 and the meteorite falls of Allende and Murchison in 1969, other noteworthy science events with round birthdays seem to be overlooked and almost forgotten Several scientific organizations celebrate the birthdays of their foundation; and key events and discoveries related to meteoritics, astronomy, geo- and cosmochemistry, and nuclear sciences can be commemorated this year, including the anniversaries of the discoveries of eleven chemical elements, and the advancements of our knowledge of the elemental and isotopic abundances. Introduction. Introduction The 150th anniversary of the discovery of the periodic system of the elements by Dmitri Ivanovich Mendeleev (8 Feb. 1834 – 2 Feb. 1907) and independently by Julius Lothar Meyer (19 August 1830 – 11 April 1895) is the reason for celebrating the International Year of the Periodic Table in 2019. Not only that, but several scientific organizations celebrate the birthdays of their foundation: The Astronomical Society of the Pacific (1889), The American Astronomical Society (1899), the American Geophysical Union (1919), the Mineralogical Society of America (1919), and the International Astronomical Union IAU (1919). The anniversaries in 2019 give us reasons to reflect on the major impacts of space exploration. In 1969, the first men landed on the moon and Apollo 11 safely returned with lunar rocks for study. The same year was blessed by the fall of the important carbonaceous chondrites Allende and Murchison. -

Marie Curie and Her Time
Marie Curie and Her Time by Hélène Langevin-Joliot to pass our lives near each other hypnotized by our dreams, your patriotic dream, our humanitarian dream, arie Curie (1867–1934) belongs to that exclu- and our scientific dream.” sive group of women whose worldwide rec- Frederick Soddy wrote about Marie that she was Mognition and fame have endured for a century “the most beautiful discovery of Pierre Curie.” Of or more. She was indeed one of the major agents of course, it might also be said that Pierre Curie was the scientific revolution which allowed experimen- “the most beautiful discovery of Marie Skłodowska.” tal investigation to extend beyond the macroscopic It is difficult to imagine more contrasting personali- world. Her work placed the first stone in the founda- ties than those of Pierre and of Marie. In spite of that, tion of a new discipline: radiochemistry. And Curie’s or because of that, they complemented each other achievements are even more remarkable since they astonishingly well. Pierre was as dreamy as Marie was occurred in the field of science, an intellectual activ- organized. At the same time, they shared similar ideas ity traditionally forbidden to women. However, these about family and society. accomplishments alone don’t seem to fully explain the near mythic status of Marie Curie today. One hundred years ago, she was often considered to be just an assistant to her husband. Perhaps the reason her name still resonates is because of the compelling story of her life and her intriguing personality. The Most Beautiful Discovery of Pierre Curie The story of the young Maria Skłodowska leaving In this iconic photograph of participants at the Fifth her native Poland to pursue upper-level studies in Solvay Conference in 1927, Marie Curie is third from Paris sounds like something out of a novel. -

George De Hevesy in America
Journal of Nuclear Medicine, published on July 13, 2019 as doi:10.2967/jnumed.119.233254 George de Hevesy in America George de Hevesy was a Hungarian radiochemist who was awarded the Nobel Prize in Chemistry in 1943 for the discovery of the radiotracer principle (1). As the radiotracer principle is the foundation of all diagnostic and therapeutic nuclear medicine procedures, Hevesy is widely considered the father of nuclear medicine (1). Although it is well-known that he spent time at a number of European institutions, it is not widely known that he also spent six weeks at Cornell University in Ithaca, NY, in the fall of 1930 as that year’s Baker Lecturer in the Department of Chemistry (2-6). “[T]he Baker Lecturer gave two formal presentations per week, to a large and diverse audience and provided an informal seminar weekly for students and faculty members interested in the subject. The lecturer had an office in Baker Laboratory and was available to faculty and students for further discussion.” (7) There is also evidence that, “…Hevesy visited Harvard [University, Cambridge, MA] as a Baker Lecturer at Cornell in 1930…” (8). Neither of the authors of this Note/Letter was aware of Hevesy’s association with Cornell University despite our longstanding ties to Cornell until one of us (WCK) noticed the association in Hevesy’s biographical page on the official Nobel website (6). WCK obtained both his undergraduate degree and medical degree from Cornell in Ithaca and New York City, respectively, and spent his career in nuclear medicine. JRO did his nuclear medicine training at Columbia University and has subsequently been a faculty member of Weill Cornell Medical College for the last eleven years (with a brief tenure at Memorial Sloan Kettering Cancer Center (affiliated with Cornell)), and is now the program director of the Nuclear Medicine residency and Chief of the Molecular Imaging and Therapeutics Section. -

Historical Development of the Periodic Classification of the Chemical Elements
THE HISTORICAL DEVELOPMENT OF THE PERIODIC CLASSIFICATION OF THE CHEMICAL ELEMENTS by RONALD LEE FFISTER B. S., Kansas State University, 1962 A MASTER'S REPORT submitted in partial fulfillment of the requirements for the degree FASTER OF SCIENCE Department of Physical Science KANSAS STATE UNIVERSITY Manhattan, Kansas 196A Approved by: Major PrafeLoor ii |c/ TABLE OF CONTENTS t<y THE PROBLEM AND DEFINITION 0? TEH-IS USED 1 The Problem 1 Statement of the Problem 1 Importance of the Study 1 Definition of Terms Used 2 Atomic Number 2 Atomic Weight 2 Element 2 Periodic Classification 2 Periodic Lav • • 3 BRIEF RtiVJiM OF THE LITERATURE 3 Books .3 Other References. .A BACKGROUND HISTORY A Purpose A Early Attempts at Classification A Early "Elements" A Attempts by Aristotle 6 Other Attempts 7 DOBEREBIER'S TRIADS AND SUBSEQUENT INVESTIGATIONS. 8 The Triad Theory of Dobereiner 10 Investigations by Others. ... .10 Dumas 10 Pettehkofer 10 Odling 11 iii TEE TELLURIC EELIX OF DE CHANCOURTOIS H Development of the Telluric Helix 11 Acceptance of the Helix 12 NEWLANDS' LAW OF THE OCTAVES 12 Newlands' Chemical Background 12 The Law of the Octaves. .........' 13 Acceptance and Significance of Newlands' Work 15 THE CONTRIBUTIONS OF LOTHAR MEYER ' 16 Chemical Background of Meyer 16 Lothar Meyer's Arrangement of the Elements. 17 THE WORK OF MENDELEEV AND ITS CONSEQUENCES 19 Mendeleev's Scientific Background .19 Development of the Periodic Law . .19 Significance of Mendeleev's Table 21 Atomic Weight Corrections. 21 Prediction of Hew Elements . .22 Influence -

A Brief History of Nuclear Astrophysics
A BRIEF HISTORY OF NUCLEAR ASTROPHYSICS PART I THE ENERGY OF THE SUN AND STARS Nikos Prantzos Institut d’Astrophysique de Paris Stellar Origin of Energy the Elements Nuclear Astrophysics Astronomy Nuclear Physics Thermodynamics: the energy of the Sun and the age of the Earth 1847 : Robert Julius von Mayer Sun heated by fall of meteors 1854 : Hermann von Helmholtz Gravitational energy of Kant’s contracting protosolar nebula of gas and dust turns into kinetic energy Timescale ~ EGrav/LSun ~ 30 My 1850s : William Thompson (Lord Kelvin) Sun heated at formation from meteorite fall, now « an incadescent liquid mass » cooling Age 10 – 100 My 1859: Charles Darwin Origin of species : Rate of erosion of the Weald valley is 1 inch/century or 22 miles wild (X 1100 feet high) in 300 My Such large Earth ages also required by geologists, like Charles Lyell A gaseous, contracting and heating Sun 푀⊙ Mean solar density : ~1.35 g/cc Sun liquid Incompressible = 4 3 푅 3 ⊙ 1870s: J. Homer Lane ; 1880s :August Ritter : Sun gaseous Compressible As it shrinks, it releases gravitational energy AND it gets hotter Earth Mayer – Kelvin - Helmholtz Helmholtz - Ritter A gaseous, contracting and heating Sun 푀⊙ Mean solar density : ~1.35 g/cc Sun liquid Incompressible = 4 3 푅 3 ⊙ 1870s: J. Homer Lane ; 1880s :August Ritter : Sun gaseous Compressible As it shrinks, it releases gravitational energy AND it gets hotter Earth Mayer – Kelvin - Helmholtz Helmholtz - Ritter A gaseous, contracting and heating Sun 푀⊙ Mean solar density : ~1.35 g/cc Sun liquid Incompressible = 4 3 푅 3 ⊙ 1870s: J. -

Tracing the Recorded History of Thin-Film Sputter Deposition: from the 1800S to 2017
Review Article: Tracing the recorded history of thin-film sputter deposition: From the 1800s to 2017 Cite as: J. Vac. Sci. Technol. A 35, 05C204 (2017); https://doi.org/10.1116/1.4998940 Submitted: 24 March 2017 . Accepted: 10 May 2017 . Published Online: 08 September 2017 J. E. Greene COLLECTIONS This paper was selected as Featured ARTICLES YOU MAY BE INTERESTED IN Review Article: Plasma–surface interactions at the atomic scale for patterning metals Journal of Vacuum Science & Technology A 35, 05C203 (2017); https:// doi.org/10.1116/1.4993602 Microstructural evolution during film growth Journal of Vacuum Science & Technology A 21, S117 (2003); https://doi.org/10.1116/1.1601610 Overview of atomic layer etching in the semiconductor industry Journal of Vacuum Science & Technology A 33, 020802 (2015); https:// doi.org/10.1116/1.4913379 J. Vac. Sci. Technol. A 35, 05C204 (2017); https://doi.org/10.1116/1.4998940 35, 05C204 © 2017 Author(s). REVIEW ARTICLE Review Article: Tracing the recorded history of thin-film sputter deposition: From the 1800s to 2017 J. E. Greenea) D. B. Willett Professor of Materials Science and Physics, University of Illinois, Urbana, Illinois, 61801; Tage Erlander Professor of Physics, Linkoping€ University, Linkoping,€ Sweden, 58183, Sweden; and University Professor of Materials Science, National Taiwan University Science and Technology, Taipei City, 106, Taiwan (Received 24 March 2017; accepted 10 May 2017; published 8 September 2017) Thin films, ubiquitous in today’s world, have a documented history of more than 5000 years. However, thin-film growth by sputter deposition, which required the development of vacuum pumps and electrical power in the 1600s and the 1700s, is a much more recent phenomenon. -

Europe's Biggest General Science Conference Concludes Successfully
SCIENCEScience PagesPAGES Special Report - ESOF 2016 Europe’s biggest generalSpecial Report science conference concludes successfully ESOF 2016, Europe's biggest general science conference concludes successfully in Manchester,in Manchester,UK UK Theme: Science as Revolution Theme: Science as Revolution - Veena Patwardhan rom 23rd to 27th July, 2016, FManchester flaunted its City of Science status as the host city of the seventh edition of EuroScience Open Forum (ESOF 2016). A bi- ennial event held in a different European city every two years, this time it was Manchester's turn to host this globally reputed science conference. Around 4500 delegates – scien- tists, innovators, academics, young researchers, journalists, policy makers, industry representatives and others – converged on the world's first industrial city to dis- cover and have discussions about the latest advancements in scien- rd th Manchester Central, venue of ESOF 2016 tific and technological researchFrom 23 to 27 July, 2016, Manchester flaunted its City of Science status as the host city of the across Europe and beyond. The seventhmain theme edition this of EuroScienceyear Laureates Open Forumand distinguished (ESOF 2016). Ascientists biennial inevent the held packed in a different was 'Science as Revolution', indicatingEuropean that city the every focus two of years,Exchange this time Hall it wasof Manchester Manchester's Central, turn to hostthe venuethis globally of the reputed the conference would be on how sciencescience andconference. technology conference. could transform life on the planet, revolutionise econo- The proceedings began with a string quartet render- mies, and help in overcoming challenges faced by global ing a piece of specially composed music. -

The Nobel Laureate George De Hevesy (1885-1966) - Universal Genius and Father of Nuclear Medicine Niese S* Am Silberblick 9, 01723 Wilsdruff, Germany
Open Access SAJ Biotechnology LETTER ISSN: 2375-6713 The Nobel Laureate George de Hevesy (1885-1966) - Universal Genius and Father of Nuclear Medicine Niese S* Am Silberblick 9, 01723 Wilsdruff, Germany *Corresponding author: Niese S, Am Silberblick 9, 01723 Wilsdruff, Germany, Tel: +49 35209 22849, E-mail: [email protected] Citation: Niese S, The Nobel Laureate George de Hevesy (1885-1966) - Universal Genius and Father of Nuclear Medicine. SAJ Biotechnol 5: 102 Article history: Received: 20 March 2018, Accepted: 29 March 2018, Published: 03 April 2018 Abstract The scientific work of the universal genius the Nobel Laureate George de Hevesy who has discovered and developed news in physics, chemistry, geology, biology and medicine is described. Special attention is given to his work in life science which he had done in the second half of his scientific career and was the base of the development of nuclear medicine. Keywords: George de Hevesy; Radionuclides; Nuclear Medicine Introduction George de Hevesy has founded Radioanalytical Chemistry and Nuclear Medicine, discovered the element hafnium and first separated stable isotopes. He was an inventor in many disciplines and his interest was not only focused on the development and refinement of methods, but also on the structure of matter and its changes: atoms, molecules, cells, organs, plants, animals, men and cosmic objects. He was working under complicated political situation in Europe in the 20th century. During his stay in Germany, Austria, Hungary, Switzerland, Denmark, and Sweden he wrote a lot papers in German. In 1962 he edited a large part of his articles in a collection where German papers are translated in English [1]. -

The Emergence of Radioecology
Cambridge University Press 978-1-107-09602-8 — Radioecology R. J. Pentreath Excerpt More Information 1 The Emergence of Radioecology 1.1 Glowing in the Dark On the frieze of the south-west facing Grenelle side of the Eiffel Tower in Paris, sandwiched between the names of Broca and Coriolis, is the name Becquerel. Paul Broca was a gifted physician and anthropologist and Gaspard-Gustave de Coriolis a famous engineer and scientist. Antoine César Becquerel, however, was a physicist who, with his son Alexandre- Edmund, had made major contributions to the study of electricity. Alexandre-Edmund was himself to make major contributions to science, not only in relation to the study of electricity but also to the early development of photography; he was very interested in the curious phenomena of luminescence and phosphorescence as well. But it was Alexandre-Edmund’s son, Antoine Henri Becquerel, who was to stumble upon a completely different phenom- enon that was to change forever our understanding of the world around us, and that phenomenon was radioactivity. Just as fortunate, perhaps, was the fact that Henri did so only because of the very recent discovery of what we now know as ionising radiation by Wilhelm Conrad Röntgen, and that discovery had been very dramatic indeed. Röntgen had been experimenting with the discharge of electricity in vacuum tubes that had metal plates sealed into their ends so that they could be connected to a battery or an induction coil. Such tubes, first produced by Johann Geissler in 1857, had been used by Julius Plücker in 1858 to study the nature of electricity, the flow of which caused a glowing light to emerge from the negative plate (the cathode) and then disappear into the positive plate (the anode). -
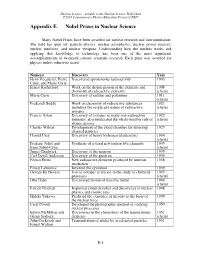
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

ARIE SKLODOWSKA CURIE Opened up the Science of Radioactivity
ARIE SKLODOWSKA CURIE opened up the science of radioactivity. She is best known as the discoverer of the radioactive elements polonium and radium and as the first person to win two Nobel prizes. For scientists and the public, her radium was a key to a basic change in our understanding of matter and energy. Her work not only influenced the development of fundamental science but also ushered in a new era in medical research and treatment. This file contains most of the text of the Web exhibit “Marie Curie and the Science of Radioactivity” at http://www.aip.org/history/curie/contents.htm. You must visit the Web exhibit to explore hyperlinks within the exhibit and to other exhibits. Material in this document is copyright © American Institute of Physics and Naomi Pasachoff and is based on the book Marie Curie and the Science of Radioactivity by Naomi Pasachoff, Oxford University Press, copyright © 1996 by Naomi Pasachoff. Site created 2000, revised May 2005 http://www.aip.org/history/curie/contents.htm Page 1 of 79 Table of Contents Polish Girlhood (1867-1891) 3 Nation and Family 3 The Floating University 6 The Governess 6 The Periodic Table of Elements 10 Dmitri Ivanovich Mendeleev (1834-1907) 10 Elements and Their Properties 10 Classifying the Elements 12 A Student in Paris (1891-1897) 13 Years of Study 13 Love and Marriage 15 Working Wife and Mother 18 Work and Family 20 Pierre Curie (1859-1906) 21 Radioactivity: The Unstable Nucleus and its Uses 23 Uses of Radioactivity 25 Radium and Radioactivity 26 On a New, Strongly Radio-active Substance