The Isotope Effect: Prediction, Discussion, and Discovery
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Arnold Sommerfeld in Einigen Zitaten Von Ihm Und Über Ihn1
K.-P. Dostal, Arnold Sommerfeld in einigen Zitaten von ihm und über ihn Seite 1 Karl-Peter Dostal, Arnold Sommerfeld in einigen Zitaten von ihm und über ihn1 Kurze biographische Bemerkungen Arnold Sommerfeld [* 5. Dezember 1868 in Königsberg, † 26. April 1951 in München] zählt neben Max Planck, Albert Einstein und Niels Bohr zu den Begründern der modernen theoretischen Physik. Durch die Ausarbeitung der Bohrschen Atomtheorie, als Lehrbuchautor (Atombau und Spektrallinien, Vorlesungen über theoretische Physik) und durch seine „Schule“ (zu der etwa die Nobelpreisträger Peter Debye, Wolfgang Pauli, Werner Heisenberg und Hans Bethe gehören) sorgte Sommerfeld wie kein anderer für die Verbreitung der modernen Physik.2 Je nach Auswahl könnte Sommerfeld [aber] nicht nur als theoretischer Physiker, sondern auch als Mathematiker, Techniker oder Wissenschaftsjournalist porträtiert werden.3 Als Schüler der Mathematiker Ferdinand von Lindemann, Adolf Hurwitz, David Hilbert und Felix Klein hatte sich Sommerfeld zunächst vor allem der Mathematik zugewandt (seine erste Professur: 1897 - 1900 für Mathematik an der Bergakademie Clausthal). Als Professor an der TH Aachen von 1900 - 1906 gewann er zunehmendes Interesse an der Technik. 1906 erhielt er den seit Jahren verwaisten Lehrstuhl für theoretische Physik in München, an dem er mit wenigen Unterbrechungen noch bis 1940 (und dann wieder ab 19464) unterrichtete. Im Gegensatz zur etablierten Experimen- talphysik war die theoretische Physik anfangs des 20. Jh. noch eine junge Disziplin. Sie wurde nun zu -

Chlorine Stable Isotopes in Sedimentary Systems: Does Size Matter?
Chlorine stable isotopes in sedimentary systems: does size matter? Max Coleman Jet Propulsion Laboratory, Caltech, Pasadena, California, USA ABSTRACT: Stable isotope abundances vary because of size (atomic mass) differences. The chlorine stable isotope system was one of the first described theoretically, but had a slow, disappointment-strewn develop- ment, relative to other elements. Method improvement gave only small, but significant, differences (-1 %.) in compositions of geological materials. Eventually, brines and groundwater chlorides gave larger differences (-5%0). Physical processes like diffusion and adsorption, probably are the main controls of groundwater com- positions. In contrast, the processes producing brine compositions are still enigmatic and need further work for full understanding. Recent work on anthropogenic groundwater contaminants (chlorinated aliphatic hy- drocarbons and perchlorate) shows variations resulting from manufacturing processes; implying possibilities of tracing sources. However, they are subject to microbial degradation, producing much larger fractionations (115%0),but therefore offering better possibility of monitoring natural attenuation. So atomic size is important to give isotopic fractionation and the size of fractionation matters, allowing observation of otherwise unde- tectable processes. lecular and atomic weights by weighing the gases and relating them to the weight of hydrogen. All 1 INTRODUCTION atomic weights were integer multiples the weight of This paper aims to give a short and selective -

Marie Curie and Her Time
Marie Curie and Her Time by Hélène Langevin-Joliot to pass our lives near each other hypnotized by our dreams, your patriotic dream, our humanitarian dream, arie Curie (1867–1934) belongs to that exclu- and our scientific dream.” sive group of women whose worldwide rec- Frederick Soddy wrote about Marie that she was Mognition and fame have endured for a century “the most beautiful discovery of Pierre Curie.” Of or more. She was indeed one of the major agents of course, it might also be said that Pierre Curie was the scientific revolution which allowed experimen- “the most beautiful discovery of Marie Skłodowska.” tal investigation to extend beyond the macroscopic It is difficult to imagine more contrasting personali- world. Her work placed the first stone in the founda- ties than those of Pierre and of Marie. In spite of that, tion of a new discipline: radiochemistry. And Curie’s or because of that, they complemented each other achievements are even more remarkable since they astonishingly well. Pierre was as dreamy as Marie was occurred in the field of science, an intellectual activ- organized. At the same time, they shared similar ideas ity traditionally forbidden to women. However, these about family and society. accomplishments alone don’t seem to fully explain the near mythic status of Marie Curie today. One hundred years ago, she was often considered to be just an assistant to her husband. Perhaps the reason her name still resonates is because of the compelling story of her life and her intriguing personality. The Most Beautiful Discovery of Pierre Curie The story of the young Maria Skłodowska leaving In this iconic photograph of participants at the Fifth her native Poland to pursue upper-level studies in Solvay Conference in 1927, Marie Curie is third from Paris sounds like something out of a novel. -

Lead Isotopes As Indicators of Environ Contamination from U Mining
._ .- - Preprint LA-UR-78-2147 , Title: LEAD ISOTOPES AS INDICATORS OF ENVIRONt1 ENTAL CONTAMINATION FRCM THE URANIUM MINING AND MILLING INDUSTRY IN THE GRANTS MINERAL BELT, NEW MEXICO Author (s): David B. Curtis and A. J. Gancarz 'k |- . 9 M/ - ' # - L .. %|.RkN: %| * & ,AQ' ,,7 ,e- d*157 ff' k g*/h T .s .. ; f*M & f g W , w -e ~ - " f:} + - pYW pm K .A ,W.% :M & Lw< *b* ? --~ k Y Q Q A -ell' 2_W h _ -- $| - f.s c t Q & Ye # C4HEhreSE33rzgmF %gh gM n [,315 ? M e % @N Y $ $ j3 h e fe l 2 D S $s - - #5 ~N [ ' & 9; - - - f _ '- ~ w e- * g r e w w 2 P As Nc- ' ** * -- - - - - $%y~ - . 9 . j'*( -, ' & . i .- f 1 * - -- ** -- e=* -h .Mr g g* ~ A _. y . gh(An ,, - ?fw h. y C: *, 1% * ~ ?, ?hSlh$i $ ': ffg w s - gp$lQ?$4$5;.>qgg - , .. INS is a Wegint of a paper intended for publication in 4 * '" M jour'ai or procet%1 ergs. Because changes rnay be made before f h , ' , publicction, this preprint is seiade available with the under. f ,- ' , starviing that it will not be died or reproduced without the * +' % '""'***'*C**' " permiss;or of the author, ') O 9 7 - _. _ _ . _ _ _ . _ - _ 0808.0 [ L * ' * . Q - 42 - 72 2)<f y . 1 LEAD ISOTOPES AS INDICATORS OF ENVIRONMENTAL CONTAMINATION FROM THE URANIUM MINING AND MILI.ING INDUSTitY .IN THE GRANIS MINERAL BELT, NEW NEXICO David B. Curtis and A. J. Gancar2 Los Alamos Scientine Laboratory Los Alamos, New Mexico, USA The unique isotopie composition oflead from uranium ores can be useful in studying ihe impact of ore processing efHuents on the env:ronment. -

Power Sources Challenge
POWER SOURCES CHALLENGE FUSION PHYSICS! A CLEAN ENERGY Summary: What if we could harness the power of the Sun for energy here on Fusion reactions occur when two nuclei come together to form one Earth? What would it take to accomplish this feat? Is it possible? atom. The reaction that happens in the sun fuses two Hydrogen atoms together to produce Helium. It looks like this in a very simplified way: Many researchers including our Department of Energy scientists and H + H He + ENERGY. This energy can be calculated by the famous engineers are taking on this challenge! In fact, there is one DOE Laboratory Einstein equation, E = mc2. devoted to fusion physics and is committed to being at the forefront of the science of magnetic fusion energy. Each of the colliding hydrogen atoms is a different isotope of In order to understand a little more about fusion energy, you will learn about hydrogen, one deuterium and one the atom and how reactions at the atomic level produce energy. tritium. The difference in these isotopes is simply one neutron. Background: It all starts with plasma! If you need to learn more about plasma Deuterium has one proton and one physics, visit the Power Sources Challenge plasma activities. neutron, tritium has one proton and two neutrons. Look at the The Fusion Reaction that happens in the SUN looks like this: illustration—do you see how the mass of the products is less than the mass of the reactants? That is called a mass deficit and that difference in mass is converted into energy. -

Cyclotron Produced Lead-203 P
Postgrad Med J: first published as 10.1136/pgmj.51.601.751 on 1 November 1975. Downloaded from Postgraduate Medical Journal (November 1975) 51, 751-754. Cyclotron produced lead-203 P. L. HORLOCK M. L. THAKUR L.R.I.C. M.Sc., Ph.D. I. A. WATSON M.Sc. MRC Cyclotron Unit, Hammersmith Hospital, Du Cane Road, London W12 OHS Introduction TABLE 1. Isotopes of lead Radioactive isotopes of lead occur in the natural Isotope Half-life Principal y energy radioactive series and have been used as decay 194Pb m * tracers since the early days of radiochemistry. These 195Pb 17m * are 210Pb (ti--204 y, radium D), 212Pb (t--10. 6 h, 96Pb 37 m * thorium B) and 214Pb (t--26.8 m, radium B) all of 197Pb 42 m * which are from occurring decay 197pbm 42 m * separated naturally 198pb 24 h * products ofradium or thorium. There are in addition 19spbm 25 m * Protected by copyright. to these a number of other radioactive isotopes of 199Pb 90 m * lead which can be produced artificially with the aid 19spbm 122 m * of a nuclear reactor or a accelerator 20OPb 21-5 h ** 0-109 to 0-605 (complex) charge particle (daughter radiations) (Table 1). 2Pb 9-4 h * There are at least three criteria in choosing the 0oipbm 61 s * radioactive isotope for in vitro tracer studies. First, 20pb 3 x 105y *** TI X-rays, (daughter it should emit radiation which is easily detected. radiations) have a half-life to 202pbm 3-62 h * Secondly, it should long enough aPb 52-1 h ** 0-279 (81%) 0-401 (5%) 0-680 permit studies without excessive radioactive decay 203pbm 6-1 s * (0-9%) (no daughter occurring but not so long that waste disposal radiations) creates a problem. -

Proton-Induced Cross-Sections of Nuclear Reactions on Lead up to 37 Mev
Proton-induced cross-sections of nuclear reactions on lead up to 37 MeV F. Ditroi´ a,∗, F. Tark´ anyi´ a, S. Takacs´ a, A. Hermanneb aInstitute for Nuclear Research, Hungarian Academy of Sciences (ATOMKI), Debrecen, Hungary bCyclotron Laboratory, Vrije Universiteit Brussel (VUB), Brussels, Belgium Abstract Excitation function of proton induced nuclear reactions on lead for production of 206;205;204;203;202;201gBi, 203cum;202m;201cumPb and 202cum;201cum;200cum;199cumTl radionuclides were measured up to 36 MeV by using activation method, stacked foil irradiation technique and ?-ray spectrometry. The new experimental data were compared with the few earlier experimental results and with the predictions of the EMPIRE 3.1, ALICE-IPPE (MENDL2p) and TALYS (TENDL-2012) theoretical reaction codes. Keywords: lead target, cross-section by proton activation, theoretical model codes, thin layer activation (TLA) 1. Introduction this, only a few experimental data sets exist, collected mostly by the experimental groups. In the low energy Production cross-sections of proton induced nuclear region practically only one experimental cross-section reactions on lead are important for many applications is available from the Hannover-Cologne group. In our and for the development of nuclear reaction theory. research and application work in connection with the Lead is an important technological material as pure el- activation cross-sections on lead we were involved in ement as well as alloying agent and it is also widely different projects and applications: used in different nuclear technologies, as well as tar- get material for production of 201Tl medical diagnostic • preparation of nuclear database for production of gamma-emitter for SPECT technology (Lagunas-Solar medical radioisotopes in the frame of an IAEA Co- et al., 1981). -

THE NATURAL RADIOACTIVITY of the BIOSPHERE (Prirodnaya Radioaktivnost' Iosfery)
XA04N2887 INIS-XA-N--259 L.A. Pertsov TRANSLATED FROM RUSSIAN Published for the U.S. Atomic Energy Commission and the National Science Foundation, Washington, D.C. by the Israel Program for Scientific Translations L. A. PERTSOV THE NATURAL RADIOACTIVITY OF THE BIOSPHERE (Prirodnaya Radioaktivnost' iosfery) Atomizdat NMoskva 1964 Translated from Russian Israel Program for Scientific Translations Jerusalem 1967 18 02 AEC-tr- 6714 Published Pursuant to an Agreement with THE U. S. ATOMIC ENERGY COMMISSION and THE NATIONAL SCIENCE FOUNDATION, WASHINGTON, D. C. Copyright (D 1967 Israel Program for scientific Translations Ltd. IPST Cat. No. 1802 Translated and Edited by IPST Staff Printed in Jerusalem by S. Monison Available from the U.S. DEPARTMENT OF COMMERCE Clearinghouse for Federal Scientific and Technical Information Springfield, Va. 22151 VI/ Table of Contents Introduction .1..................... Bibliography ...................................... 5 Chapter 1. GENESIS OF THE NATURAL RADIOACTIVITY OF THE BIOSPHERE ......................... 6 § Some historical problems...................... 6 § 2. Formation of natural radioactive isotopes of the earth ..... 7 §3. Radioactive isotope creation by cosmic radiation. ....... 11 §4. Distribution of radioactive isotopes in the earth ........ 12 § 5. The spread of radioactive isotopes over the earth's surface. ................................. 16 § 6. The cycle of natural radioactive isotopes in the biosphere. ................................ 18 Bibliography ................ .................. 22 Chapter 2. PHYSICAL AND BIOCHEMICAL PROPERTIES OF NATURAL RADIOACTIVE ISOTOPES. ........... 24 § 1. The contribution of individual radioactive isotopes to the total radioactivity of the biosphere. ............... 24 § 2. Properties of radioactive isotopes not belonging to radio- active families . ............ I............ 27 § 3. Properties of radioactive isotopes of the radioactive families. ................................ 38 § 4. Properties of radioactive isotopes of rare-earth elements . -

12 Natural Isotopes of Elements Other Than H, C, O
12 NATURAL ISOTOPES OF ELEMENTS OTHER THAN H, C, O In this chapter we are dealing with the less common applications of natural isotopes. Our discussions will be restricted to their origin and isotopic abundances and the main characteristics. Only brief indications are given about possible applications. More details are presented in the other volumes of this series. A few isotopes are mentioned only briefly, as they are of little relevance to water studies. Based on their half-life, the isotopes concerned can be subdivided: 1) stable isotopes of some elements (He, Li, B, N, S, Cl), of which the abundance variations point to certain geochemical and hydrogeological processes, and which can be applied as tracers in the hydrological systems, 2) radioactive isotopes with half-lives exceeding the age of the universe (232Th, 235U, 238U), 3) radioactive isotopes with shorter half-lives, mainly daughter nuclides of the previous catagory of isotopes, 4) radioactive isotopes with shorter half-lives that are of cosmogenic origin, i.e. that are being produced in the atmosphere by interactions of cosmic radiation particles with atmospheric molecules (7Be, 10Be, 26Al, 32Si, 36Cl, 36Ar, 39Ar, 81Kr, 85Kr, 129I) (Lal and Peters, 1967). The isotopes can also be distinguished by their chemical characteristics: 1) the isotopes of noble gases (He, Ar, Kr) play an important role, because of their solubility in water and because of their chemically inert and thus conservative character. Table 12.1 gives the solubility values in water (data from Benson and Krause, 1976); the table also contains the atmospheric concentrations (Andrews, 1992: error in his Eq.4, where Ti/(T1) should read (Ti/T)1); 2) another category consists of the isotopes of elements that are only slightly soluble and have very low concentrations in water under moderate conditions (Be, Al). -
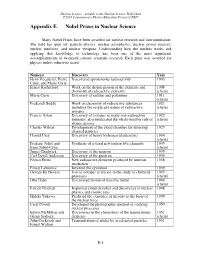
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

ARIE SKLODOWSKA CURIE Opened up the Science of Radioactivity
ARIE SKLODOWSKA CURIE opened up the science of radioactivity. She is best known as the discoverer of the radioactive elements polonium and radium and as the first person to win two Nobel prizes. For scientists and the public, her radium was a key to a basic change in our understanding of matter and energy. Her work not only influenced the development of fundamental science but also ushered in a new era in medical research and treatment. This file contains most of the text of the Web exhibit “Marie Curie and the Science of Radioactivity” at http://www.aip.org/history/curie/contents.htm. You must visit the Web exhibit to explore hyperlinks within the exhibit and to other exhibits. Material in this document is copyright © American Institute of Physics and Naomi Pasachoff and is based on the book Marie Curie and the Science of Radioactivity by Naomi Pasachoff, Oxford University Press, copyright © 1996 by Naomi Pasachoff. Site created 2000, revised May 2005 http://www.aip.org/history/curie/contents.htm Page 1 of 79 Table of Contents Polish Girlhood (1867-1891) 3 Nation and Family 3 The Floating University 6 The Governess 6 The Periodic Table of Elements 10 Dmitri Ivanovich Mendeleev (1834-1907) 10 Elements and Their Properties 10 Classifying the Elements 12 A Student in Paris (1891-1897) 13 Years of Study 13 Love and Marriage 15 Working Wife and Mother 18 Work and Family 20 Pierre Curie (1859-1906) 21 Radioactivity: The Unstable Nucleus and its Uses 23 Uses of Radioactivity 25 Radium and Radioactivity 26 On a New, Strongly Radio-active Substance -

Chapter 6 Free Electron Fermi Gas
理学院 物理系 沈嵘 Chapter 6 Free Electron Fermi Gas 6.1 Electron Gas Model and its Ground State 6.2 Thermal Properties of Electron Gas 6.3 Free Electrons in Electric Fields 6.4 Hall Effect 6.5 Thermal Conductivity of Metals 6.6 Failures of the free electron gas model 1 6.1 Electron Gas Model and its Ground State 6.1 Electron Gas Model and its Ground State I. Basic Assumptions of Electron Gas Model Metal: valence electrons → conduction electrons (moving freely) ü The simplest metals are the alkali metals—lithium, sodium, 2 potassium, cesium, and rubidium. 6.1 Electron Gas Model and its Ground State density of electrons: Zr n = N m A A where Z is # of conduction electrons per atom, A is relative atomic mass, rm is the density of mass in the metal. The spherical volume of each electron is, 1 3 1 V 4 3 æ 3 ö = = p rs rs = ç ÷ n N 3 è 4p nø Free electron gas model: Suppose, except the confining potential near surfaces of metals, conduction electrons are completely free. The conduction electrons thus behave just like gas atoms in an ideal gas --- free electron gas. 3 6.1 Electron Gas Model and its Ground State Basic Properties: ü Ignore interactions of electron-ion type (free electron approx.) ü And electron-eletron type (independent electron approx). Total energy are of kinetic type, ignore potential energy contribution. ü The classical theory had several conspicuous successes 4 6.1 Electron Gas Model and its Ground State Long Mean Free Path: ü From many types of experiments it is clear that a conduction electron in a metal can move freely in a straight path over many atomic distances.