Melatonin Membrane Receptors in Peripheral Tissues: Distribution and Functions Radomir M
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supporting Information
Supporting Information Koyanagi et al. 10.1073/pnas.0806215105 SI Materials and Methods CNGB, NM137763; human CNGB1, NM001297; human Amino Acid Sequences. The accession numbers of amino acid CNGB3, NM 019098; box jellyfish CNG, AB435552; fruit fly sequences used for analyses are as follows: Box jellyfish opsin, CNG4, NM 167441; fruit fly CNG3, NM 137871; human AB435549; sea anemone opsin, BR000662; hydra opsin, Con- CNGA4, NM 001037329; fruit fly CNGA, NM 057768; human CNGA1, NM000087; human CNGA2, NM005140; human tig39347:487820–488855 (http://hydrazome.metazome.net); hy- drozaon jellyfish opsin, AB332435; human encephalopsin, CNGA3, NM 001298. AF140242; ragworm c-opsin, AY692353; human rhodopsin, Western Blot Analysis. The proteins extracted from half a rhopalia U49742; human blue, M13299; human red, Z68193; lamprey were separated by 12% SDS/PAGE, transferred onto a PVDF parapinopsin, AB116380; lizard parietopsin, DQ100320; am- membrane, and incubated with 1:500 diluted anti-box jellyfish phioxus peropsin, AB050610; human peropsin, AF012270; hu- opsin antiserum. Visualization was carried out by the ABC man rgr, U15790; squid retinochrome, X57143; human melan- method (Vectastain) and by staining with 3,3Ј-diaminobenzidine opsin, AF147788; amphioxus melanopsin, AB205400; squid (Sigma). rhodopsin, X70498; fruit fly Rh1, K02315; human neuropsin, AY377391; amphioxus rhodopsin, AB050606; scallop Go- In Situ Hybridization. Digoxigenin-labeled antisense and sense RNA rhodopsin, AB006455; human muscarinic acetylcholine receptor probes for the box jellyfish opsin were synthesized by using the DIG M1 (CHRM1), NM000738; human melatonin receptor 1A RNA labeling kit (Roche). Sections were pretreated with protein- (MTNR1A), NM005958; Arabidopsis AKT1, U06745; Arabi- ase K and hybridized with each RNA probe. -

The G Protein-Coupled Receptor Subset of the Dog Genome Is More Similar
BMC Genomics BioMed Central Research article Open Access The G protein-coupled receptor subset of the dog genome is more similar to that in humans than rodents Tatjana Haitina1, Robert Fredriksson1, Steven M Foord2, Helgi B Schiöth*1 and David E Gloriam*2 Address: 1Department of Neuroscience, Functional Pharmacology, Uppsala University, BMC, Box 593, 751 24, Uppsala, Sweden and 2GlaxoSmithKline Pharmaceuticals, New Frontiers Science Park, 3rd Avenue, Harlow CM19 5AW, UK Email: Tatjana Haitina - [email protected]; Robert Fredriksson - [email protected]; Steven M Foord - [email protected]; Helgi B Schiöth* - [email protected]; David E Gloriam* - [email protected] * Corresponding authors Published: 15 January 2009 Received: 20 August 2008 Accepted: 15 January 2009 BMC Genomics 2009, 10:24 doi:10.1186/1471-2164-10-24 This article is available from: http://www.biomedcentral.com/1471-2164/10/24 © 2009 Haitina et al; licensee BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Abstract Background: The dog is an important model organism and it is considered to be closer to humans than rodents regarding metabolism and responses to drugs. The close relationship between humans and dogs over many centuries has lead to the diversity of the canine species, important genetic discoveries and an appreciation of the effects of old age in another species. The superfamily of G protein-coupled receptors (GPCRs) is one of the largest gene families in most mammals and the most exploited in terms of drug discovery. -

A 3' UTR SNP Rs885863, a Cis-Eqtl for the Circadian Gene VIPR2 and Lincrna 689, Is Associated with Opioid Addiction
RESEARCH ARTICLE A 3' UTR SNP rs885863, a cis-eQTL for the circadian gene VIPR2 and lincRNA 689, is associated with opioid addiction 1 1 2 3 4 Orna LevranID *, Matthew Randesi , John Rotrosen , Jurg Ott , Miriam Adelson , Mary Jeanne Kreek1 1 The Laboratory of the Biology of Addictive Diseases, The Rockefeller University, New York, New York, United States of America, 2 NYU School of Medicine, New York, New York, United States of America, 3 The Laboratory of Statistical Genetics, The Rockefeller University, New York, New York, United States of a1111111111 America, 4 Dr. Miriam and Sheldon G. Adelson Clinic for Drug Abuse Treatment and Research, Las Vegas, a1111111111 Nevada, United States of America a1111111111 a1111111111 * [email protected] a1111111111 Abstract There is a reciprocal relationship between the circadian and the reward systems. Polymor- OPEN ACCESS phisms in several circadian rhythm-related (clock) genes were associated with drug addic- Citation: Levran O, Randesi M, Rotrosen J, Ott J, tion. This study aims to search for associations between 895 variants in 39 circadian Adelson M, Kreek MJ (2019) A 3' UTR SNP rhythm-related genes and opioid addiction (OUD). Genotyping was performed with the rs885863, a cis-eQTL for the circadian gene VIPR2 ® and lincRNA 689, is associated with opioid Smokescreen array. Ancestry was verified by principal/MDS component analysis and the addiction. PLoS ONE 14(11): e0224399. https:// sample was limited to European Americans (EA) (OUD; n = 435, controls; n = 138). Nomi- doi.org/10.1371/journal.pone.0224399 nally significant associations (p < 0.01) were detected for several variants in genes encoding Editor: Huiping Zhang, Boston University, UNITED vasoactive intestinal peptide receptor 2 (VIPR2), period circadian regulator 2 (PER2), STATES casein kinase 1 epsilon (CSNK1E), and activator of transcription and developmental regula- Received: August 22, 2019 tor (AUTS2), but no signal survived correction for multiple testing. -

Genetic Basis of Idiopathic Scoliosis
Research & Review: Management of Cardiovascular and Orthopedic Complications Volume 1 Issue 1 Genetic Basis of Idiopathic Scoliosis S. Sreeremya Assistant Professor, Department of Biotechnology, Sree Narayana Guru College, Coimbatore, Tamil Nadu, India Email: [email protected] Abstract Idiopathic scoliosis (IS), the most usual spinal deformity, affects otherwise healthy children and adolescents during growth. The etiology is still not quiet understood, although genetic factors are believed to be important. This review corroborates the understanding of IS as a complex disease with a polygenic background. Presumably IS can be typically due to a spectrum of genetic risk variants, ranging from very rare or even private to very common. The most promising candidate genes are highlighted. Keywords: Idiopathic scoliosis, Genetics, Pathogenesis, Heredity INTRODUCTION marked by phenotypic complexity Idiopathic scoliosis (IS), the most general (variations in curve morphology and form of spinal deformity, affects otherwise magnitude, age of onset, rate of healthy children and adolescents during progression), and a prognosis mainly growth (Fig: 1). It usually presents as a rib ranging from increase in curve magnitude, hump visible at forward bending, together to stabilization, or to resolution with with unlevelled shoulders and an growth [5]. Genetic factors are known to asymmetrical waist [1]. According to play a pivotal role, as observed in twin Cobb, the diagnosis is specifically studies and their observation and singleton confirmed by a standing spinal radiograph multigenerational families [6]. A recent showing a lateral curvature of the spine research of monozygotic and dizygotic exceeding 10° [2]. A main concern in IS is twins from the Swedish twin registry the absence of reliable means by which to estimated that overall genetic effects predict risk of progression, leading to accounted for 39 % of the observed frequent follow-ups, radiographs, and phenotypic variance, leaving the remaining potentially unnecessary brace treatments. -

The Role of the Mtor Pathway in Developmental Reprogramming Of
THE ROLE OF THE MTOR PATHWAY IN DEVELOPMENTAL REPROGRAMMING OF HEPATIC LIPID METABOLISM AND THE HEPATIC TRANSCRIPTOME AFTER EXPOSURE TO 2,2',4,4'- TETRABROMODIPHENYL ETHER (BDE-47) An Honors Thesis Presented By JOSEPH PAUL MCGAUNN Approved as to style and content by: ________________________________________________________** Alexander Suvorov 05/18/20 10:40 ** Chair ________________________________________________________** Laura V Danai 05/18/20 10:51 ** Committee Member ________________________________________________________** Scott C Garman 05/18/20 10:57 ** Honors Program Director ABSTRACT An emerging hypothesis links the epidemic of metabolic diseases, such as non-alcoholic fatty liver disease (NAFLD) and diabetes with chemical exposures during development. Evidence from our lab and others suggests that developmental exposure to environmentally prevalent flame-retardant BDE47 may permanently reprogram hepatic lipid metabolism, resulting in an NAFLD-like phenotype. Additionally, we have demonstrated that BDE-47 alters the activity of both mTOR complexes (mTORC1 and 2) in hepatocytes. The mTOR pathway integrates environmental information from different signaling pathways, and regulates key cellular functions such as lipid metabolism, innate immunity, and ribosome biogenesis. Thus, we hypothesized that the developmental effects of BDE-47 on liver lipid metabolism are mTOR-dependent. To assess this, we generated mice with liver-specific deletions of mTORC1 or mTORC2 and exposed these mice and their respective controls perinatally to -

Expression of Melatonin Receptors 1A/1B, Calmodulin and Estrogen Receptor 2 in Deep Paravertebral Muscles Revisited
MOLECULAR MEDICINE REPORTS 14: 5719-5724, 2016 Etiopathogenesis of adolescent idiopathic scoliosis: Expression of melatonin receptors 1A/1B, calmodulin and estrogen receptor 2 in deep paravertebral muscles revisited JOSEF ZAMECNIK1*, LENKA KRSKOVA1*, JAROMIR HACEK1, IVANA STETKAROVA2 and MARTIN KRBEC3 1Department of Pathology and Molecular Medicine, 2nd Faculty of Medicine, Charles University in Prague and University Hospital Motol, 15006 Prague; 2Department of Neurology, Charles University; 3Department of Orthopedics and Traumatology, 3rd Faculty of Medicine, Charles University in Prague and University Hospital Královské Vinohrady, 10034 Prague, Czech Republic Received October 28, 2015; Accepted October 11, 2016 DOI: 10.3892/mmr.2016.5927 Abstract. The pathogenesis of adolescent idiopathic scoliosis addition, no difference in expression was detected between the (AIS), including the associated local changes in deep para- patients with AIS and the controls. With regards to MTNR1A vertebral muscles, is poorly understood. The asymmetric and MTNR1B, their expression was very weak in paravertebral expression of several molecules involved in the melatonin muscles, and in the majority of cases their expression could signaling pathway, including melatonin receptors 1A/1B not be detected by repeated RT-qPCR analysis. Therefore, (MTNR1A/MTNR1B), estrogen receptor 2 (ESR2) and these data do not support the previously suggested role of the calmodulin (CALM1), has previously been suggested to be asymmetric expression of molecules involved in the melatonin associated with AIS. However, this hypothesis is based on signaling pathway in deep paravertebral muscles in the patho- single studies in which the data were obtained by different genesis of AIS. methodological approaches. Therefore, to evaluate the symmetry of the mRNA expression levels of these molecules, Introduction 18 patients with AIS and 10 non-scoliotic controls were enrolled in the present study. -

Melatonin-The Hormone of Darkness - O
PHYSIOLOGY AND MAINTENANCE – Vol. III - Melatonin-The Hormone of Darkness - O. Vakkuri MELATONIN―THE HORMONE OF DARKNESS O. Vakkuri Department of Physiology, University of Oulu, Finland. Keywords: Pineal gland, retina, suprachiasmatic nuclei, circadian and circannual rhythms. Contents 1. Introduction 2. Melatonin as Pineal Hormone of Darkness 3. Melatonin in Other Tissues 4. Circadian Secretion Pattern of Melatonin 5. Seasonal Secretion of Melatonin 6. Metabolism of Melatonin 7. Melatonin Receptors 8. Biological Action Profile of Melatonin 8.1. Melatonin and Sleep 8.2. Melatonin as Antioxidant and Cancer 8.3. Melatonin, Mental Health and Aging 9. Future Perspectives 10. Conclusions Glossary Bibliography Biographical Sketch Summary Melatonin, the pineal hormone of darkness, was originally found and chemically characterized to N-acetyl-5-methoxytryptamine in bovine pineal extracts in the late 1950s. Since then melatonin has been studied more and more intensively and not only in humans and several animal species but lately also in plants. After its first-described biological effect, i.e. skin-lightening effect in lower vertebrates, melatonin was shortly known as a rhythm marker due to its circadian biosynthesis and secretion pattern in the pineal gland: melatonin is synthesized and secreted during the night, i.e. the dark period of the day.UNESCO This circadian rhythm is endoge – nouslyEOLSS regulated by the biological clock in the suprachiasmatic nuclei of the hypothalamus. Environmental light has a clear inhibiting effectSAMPLE on melatonin biosynthesis, CHAPTERS continuously entraining the melatonin rhythm so that endogenous and exogenous rhythms are maintained in the same phase. The entraining light information is transmitting via the eyes and the retinohypothalamic tract to the suprachiasmatic nuclei and then via the paraventricular nuclei to superior cervical ganglia from which along the sympathetic tract finally to the pineal gland. -

The Neuroprotective Effects of Melatonin: Possible Role in the Pathophysiology of Neuropsychiatric Disease
brain sciences Perspective The Neuroprotective Effects of Melatonin: Possible Role in the Pathophysiology of Neuropsychiatric Disease Jung Goo Lee 1,2 , Young Sup Woo 3, Sung Woo Park 2,4, Dae-Hyun Seog 5, Mi Kyoung Seo 6 and Won-Myong Bahk 3,* 1 Department of Psychiatry, College of Medicine, Haeundae Paik Hospital, Inje University, Busan 47392, Korea; [email protected] 2 Paik Institute for Clinical Research, Department of Health Science and Technology, Graduate School, Inje University, Busan 47392, Korea; [email protected] 3 Department of Psychiatry, College of Medicine, The Catholic University of Korea, Seoul 07345, Korea; [email protected] 4 Department of Convergence Biomedical Science, College of Medicine, Inje University, Busan 47392, Korea 5 Department of Biochemistry, College of Medicine, Inje University, Busan 47392, Korea; [email protected] 6 Paik Institute for Clinical Research, Inje University, Busan 47392, Korea; [email protected] * Correspondence: [email protected] Received: 16 September 2019; Accepted: 19 October 2019; Published: 21 October 2019 Abstract: Melatonin is a hormone that is secreted by the pineal gland. To date, melatonin is known to regulate the sleep cycle by controlling the circadian rhythm. However, recent advances in neuroscience and molecular biology have led to the discovery of new actions and effects of melatonin. In recent studies, melatonin was shown to have antioxidant activity and, possibly, to affect the development of Alzheimer’s disease (AD). In addition, melatonin has neuroprotective effects and affects neuroplasticity, thus indicating potential antidepressant properties. In the present review, the new functions of melatonin are summarized and a therapeutic target for the development of new drugs based on the mechanism of action of melatonin is proposed. -
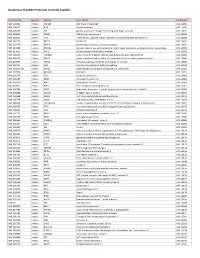
Quantigene Flowrna Probe Sets Currently Available
QuantiGene FlowRNA Probe Sets Currently Available Accession No. Species Symbol Gene Name Catalog No. NM_003452 Human ZNF189 zinc finger protein 189 VA1-10009 NM_000057 Human BLM Bloom syndrome VA1-10010 NM_005269 Human GLI glioma-associated oncogene homolog (zinc finger protein) VA1-10011 NM_002614 Human PDZK1 PDZ domain containing 1 VA1-10015 NM_003225 Human TFF1 Trefoil factor 1 (breast cancer, estrogen-inducible sequence expressed in) VA1-10016 NM_002276 Human KRT19 keratin 19 VA1-10022 NM_002659 Human PLAUR plasminogen activator, urokinase receptor VA1-10025 NM_017669 Human ERCC6L excision repair cross-complementing rodent repair deficiency, complementation group 6-like VA1-10029 NM_017699 Human SIDT1 SID1 transmembrane family, member 1 VA1-10032 NM_000077 Human CDKN2A cyclin-dependent kinase inhibitor 2A (melanoma, p16, inhibits CDK4) VA1-10040 NM_003150 Human STAT3 signal transducer and activator of transcripton 3 (acute-phase response factor) VA1-10046 NM_004707 Human ATG12 ATG12 autophagy related 12 homolog (S. cerevisiae) VA1-10047 NM_000737 Human CGB chorionic gonadotropin, beta polypeptide VA1-10048 NM_001017420 Human ESCO2 establishment of cohesion 1 homolog 2 (S. cerevisiae) VA1-10050 NM_197978 Human HEMGN hemogen VA1-10051 NM_001738 Human CA1 Carbonic anhydrase I VA1-10052 NM_000184 Human HBG2 Hemoglobin, gamma G VA1-10053 NM_005330 Human HBE1 Hemoglobin, epsilon 1 VA1-10054 NR_003367 Human PVT1 Pvt1 oncogene homolog (mouse) VA1-10061 NM_000454 Human SOD1 Superoxide dismutase 1, soluble (amyotrophic lateral sclerosis 1 (adult)) -

Hormonal Regulation of Oligodendrogenesis I: Effects Across the Lifespan
biomolecules Review Hormonal Regulation of Oligodendrogenesis I: Effects across the Lifespan Kimberly L. P. Long 1,*,†,‡ , Jocelyn M. Breton 1,‡,§ , Matthew K. Barraza 2 , Olga S. Perloff 3 and Daniela Kaufer 1,4,5 1 Helen Wills Neuroscience Institute, University of California, Berkeley, CA 94720, USA; [email protected] (J.M.B.); [email protected] (D.K.) 2 Department of Molecular and Cellular Biology, University of California, Berkeley, CA 94720, USA; [email protected] 3 Memory and Aging Center, Department of Neurology, University of California, San Francisco, CA 94143, USA; [email protected] 4 Department of Integrative Biology, University of California, Berkeley, CA 94720, USA 5 Canadian Institute for Advanced Research, Toronto, ON M5G 1M1, Canada * Correspondence: [email protected] † Current address: Department of Psychiatry and Behavioral Sciences, University of California, San Francisco, CA 94143, USA. ‡ These authors contributed equally to this work. § Current address: Department of Psychiatry, Columbia University, New York, NY 10027, USA. Abstract: The brain’s capacity to respond to changing environments via hormonal signaling is critical to fine-tuned function. An emerging body of literature highlights a role for myelin plasticity as a prominent type of experience-dependent plasticity in the adult brain. Myelin plasticity is driven by oligodendrocytes (OLs) and their precursor cells (OPCs). OPC differentiation regulates the trajectory of myelin production throughout development, and importantly, OPCs maintain the ability to proliferate and generate new OLs throughout adulthood. The process of oligodendrogenesis, Citation: Long, K.L.P.; Breton, J.M.; the‘creation of new OLs, can be dramatically influenced during early development and in adulthood Barraza, M.K.; Perloff, O.S.; Kaufer, D. -

Genetic Variations of Melatonin Receptor Type 1A Are Associated with the Clinicopathologic Development of Urothelial Cell Carcin
Int. J. Med. Sci. 2017, Vol. 14 1130 Ivyspring International Publisher International Journal of Medical Sciences 2017; 14(11): 1130-1135. doi: 10.7150/ijms.20629 Research Paper Genetic Variations of Melatonin Receptor Type 1A are Associated with the Clinicopathologic Development of Urothelial Cell Carcinoma Yung-Wei Lin1, 2, Shian-Shiang Wang3, 4, 5, Yu-Ching Wen2, 6, Min-Che Tung1, 7, Liang-Ming Lee2, 6, Shun-Fa Yang5, 8, Ming-Hsien Chien1, 9 1. Graduate Institute of Clinical Medicine, Taipei Medical University, Taipei, Taiwan; 2. Department of Urology, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan; 3. Division of Urology, Department of Surgery, Taichung Veterans General Hospital, Taichung, Taiwan; 4. School of Medicine, Chung Shan Medical University, Taichung, Taiwan; 5. Institute of Medicine, Chung Shan Medical University, Taichung, Taiwan; 6. Department of Urology, School of Medicine, College of Medicine, Taipei Medical University, Taipei, Taiwan; 7. Department of Surgery, Tungs' Taichung Metro Harbor Hospital, Taichung, Taiwan; 8. Department of Medical Research, Chung Shan Medical University Hospital, Taichung, Taiwan; 9. Department of Medical Education and Research, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan. Corresponding authors: Ming-Hsien Chien, PhD, Graduate Institute of Clinical Medicine, Taipei Medical University, 250 Wu-Hsing Street, Taipei 11031, Taiwan; Phone: 886-2-27361661, ext. 3237; Fax: 886-2-27390500; E-mail: [email protected] or Shun-Fa Yang, PhD, Institute of Medicine, Chung Shan Medical University, 110 Chien-Kuo N. Road, Section 1, Taichung 402, Taiwan; Phone: 886-4-2473959, ext. 34253; Fax: 886-4-24723229; E-mail: [email protected] © Ivyspring International Publisher. -

The Role of Melatonin in Diabetes: Therapeutic Implications
review The role of melatonin in diabetes: therapeutic implications Shweta Sharma1, Hemant Singh1, Nabeel Ahmad2, Priyanka Mishra1, Archana Tiwari1 ABSTRACT Melatonin referred as the hormone of darkness is mainly secreted by pineal gland, its levels being 1 School of Biotechnology, Rajiv elevated during night and low during the day. The effects of melatonin on insulin secretion are me- Gandhi Technical University, Gandhi diated through the melatonin receptors (MT1 and MT2). It decreases insulin secretion by inhibiting Nagar, Bhopal, Madhya Pradesh cAMP and cGMP pathways but activates the phospholipaseC/IP3 pathway, which mobilizes Ca2+ from 2 School of Biotechnology, organelles and, consequently increases insulin secretion. Both in vivo and in vitro, insulin secretion IFTM University, Lodhipur Rajput, Uttar Pradesh by the pancreatic islets in a circadian manner, is due to the melatonin action on the melatonin recep- tors inducing a phase shift in the cells. Melatonin may be involved in the genesis of diabetes as a Correspondence to: reduction in melatonin levels and a functional interrelationship between melatonin and insulin was Shweta Sharma School of Biotechnology observed in diabetic patients. Evidences from experimental studies proved that melatonin induces Rajiv Gandhi Technical University production of insulin growth factor and promotes insulin receptor tyrosine phosphorylation. The dis- Airport Bypass Road, Gandhi Nagar turbance of internal circadian system induces glucose intolerance and insulin resistance, which could 462036 – Bhopal, Madhya Pradesh [email protected] be restored by melatonin supplementation. Therefore, the presence of melatonin receptors on hu- man pancreatic islets may have an impact on pharmacotherapy of type 2 diabetes. Arch Endocrinol Metab. Received on June/8/2015 2015;59(5):391-9 Accepted on July/6/2015 DOI: 10.1590/2359-3997000000098 Keywords Melatonin; diabetes; insulin; beta cells; calcium; circadian rhythm INTRODUCTION tomy of rodents causes hyperinsulinemia (7).