URINOGENITAL SYSTEM Part-1
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Te2, Part Iii
TERMINOLOGIA EMBRYOLOGICA Second Edition International Embryological Terminology FIPAT The Federative International Programme for Anatomical Terminology A programme of the International Federation of Associations of Anatomists (IFAA) TE2, PART III Contents Caput V: Organogenesis Chapter 5: Organogenesis (continued) Systema respiratorium Respiratory system Systema urinarium Urinary system Systemata genitalia Genital systems Coeloma Coelom Glandulae endocrinae Endocrine glands Systema cardiovasculare Cardiovascular system Systema lymphoideum Lymphoid system Bibliographic Reference Citation: FIPAT. Terminologia Embryologica. 2nd ed. FIPAT.library.dal.ca. Federative International Programme for Anatomical Terminology, February 2017 Published pending approval by the General Assembly at the next Congress of IFAA (2019) Creative Commons License: The publication of Terminologia Embryologica is under a Creative Commons Attribution-NoDerivatives 4.0 International (CC BY-ND 4.0) license The individual terms in this terminology are within the public domain. Statements about terms being part of this international standard terminology should use the above bibliographic reference to cite this terminology. The unaltered PDF files of this terminology may be freely copied and distributed by users. IFAA member societies are authorized to publish translations of this terminology. Authors of other works that might be considered derivative should write to the Chair of FIPAT for permission to publish a derivative work. Caput V: ORGANOGENESIS Chapter 5: ORGANOGENESIS -

Structure of Pronephros and Development of Mesonephric Kidney in Larvae of Russian Sturgeon, Acipenser Gueldenstaedtii Brandt (Acipenseridae)
Zoologica5 PRONEPHROS Poloniae-AND (2012)-MESONEPHRIC 57/1-4: 5-20-KIDNEY-IN-LARVAE-OF-A.-GUELDENSTAEDTII 5 DOI: 10.2478/v10049-012-0001-6 STRUCTURE OF PRONEPHROS AND DEVELOPMENT OF MESONEPHRIC KIDNEY IN LARVAE OF RUSSIAN STURGEON, ACIPENSER GUELDENSTAEDTII BRANDT (ACIPENSERIDAE) L.S. KRAYUSHKINA*1, A.A. GERASIMOV1, A.A. KIRSANOV1, M.V. MOSYAGINA1, A. OGORZA£EK2 1Department of Ichthyology and Hydrobiology, St. Petersburg State University, 16-th Line 29, 199178, St. Petersburg, Russia, [email protected] 2 Department of Animal Developmental Biology, Zoological Institute, University of Wroclaw, Sienkiewicza 21, 50-335 Wroclaw, Poland. *Corresponding author Abstract. The structure of the pronephros and development of mesonephric kidney in Russian sturgeon larvae, Acipenser gueldenstaedtii Brandt at different stages of early postembryonic development (from hatching to 14 days), were studied with histological and electronic microscopy methods. The larval pronephros is represented by the system of bilaterally located pronephric tubules with ciliated nephrostomes and funnels and exog- enous single glomus, which is not integrated directly into pronephric tubules and located in the pronephric chamber. The glomus is positioned below the dorsal aorta and vascular- ized by its capillaries. The glomus has the same features of the thin structure that are typical of and necessary for the function of a filtering organ. The structure of the prone- phros in acipenserids is discussed and compared with teleosts and amphibians. Histogen- esis of the mesonephric kidney is observed during the period of pronephros functioning; it is complete by the time the larvae transfer to exogenous feeding. At this moment, the pronephros undergoes significant structural degradation. -

Essential Roles of Inhibin Beta a in Mouse Epididymal Coiling
Essential roles of inhibin beta A in mouse epididymal coiling Jessica Tomaszewski*, Avenel Joseph†, Denise Archambeault†, and Humphrey Hung-Chang Yao†‡ *Department of Biology, School of Integrative Biology, and †Department of Veterinary Biosciences, College of Veterinary Medicine, University of Illinois at Urbana–Champaign, Urbana, IL 61802 Edited by Jean D. Wilson, University of Texas Southwestern Medical Center, Dallas, TX, and approved May 29, 2007 (received for review April 13, 2007) Testis-derived testosterone has been recognized as the key factor appear to be indirect via a mesenchyme-derived regulator(s). When for morphogenesis of the Wolffian duct, the precursor of several the upper Wolffian duct epithelium (the future epididymis) was male reproductive tract structures. Evidence supports that testos- grafted onto the lower Wolffian duct mesenchyme (the future terone is required for the maintenance of the Wolffian duct via its seminal vesicle), the epithelium underwent seminal vesicle mor- action on the mesenchyme. However, it remains uncertain how phogenesis and expressed markers specific for seminal vesicle testosterone alone is able to facilitate formation of regionally epithelium instead of those for epididymal epithelium (10). This specific structures such as the epididymis, vas deferens, and sem- inductive ability of Wolffian duct mesenchyme was also found in the inal vesicle from a straight Wolffian duct. In this study, we iden- prostate, providing further support that AR in the mesenchyme is tified inhibin beta A (or Inhba) as a regional paracrine factor in necessary to dictate androgenic actions of the epithelium (11). mouse mesonephroi that controls coiling of the epithelium in the Furthermore, when the epithelium from the AR-deficient testicular anterior Wolffian duct, the future epididymis. -

Development of the Urogenital System of the Dog
DEVELOPMENT OF THE UROGENITAL SYSTEM OF THE DOG MAJID AHMED AL-RADHAWI LICENCE, Higher Teachers' Training College, Baghdad, Iraq, 1954 A. THESIS submitted in partial fulfillment of the requirements for the degree MASTER OF SCIENCE Department of Zoology KANSAS STATE COLLEGE OF AGRICULTURE AND APPLIED SCIENCE 1958 LD C-2- TABLE OF CONTENTS INTRODUCTION AND HEVIEW OF LITERATURE 1 MATERIALS AND METHODS 3 OBSERVATIONS 5 Group I, Embryos from 8-16 Somites 5 Group II, Embryos from 17-26 Somites 10 Group III, Embryos from 27-29 Somites 12 Group 17, Embryos from 34-41 Somites 15 Group V , Embryos from 41-53 Somites 18 Subgroup A, Embryos from 41-<7 Somites 18 Subgroup B, Embryos from 4-7-53 Somites 20 Group VI, Embryos Showing Indifferent Gonad 22 Group VII, Embryos with Differentiatle Gonad 24 Subgroup A, Embryos with Seoondarily Divertioulated Pelvis ... 25 Subgroup B, Embryos with the Anlagen of the Uriniferous Tubules . 26 Subgroup C, Embryos with Advanced Gonad 27 DISCUSSION AND GENERAL CONSIDERATION 27 Formation of the Kidney .... 27 The Pronephros 31 The Mesonephros 35 The Metanephros 38 The Ureter 39 The Urogenital Sinus 4.0 The Mullerian Duot 40 The Gonad 41 1 iii TABLE OF CONTENTS The Genital Ridge Stage 41 The Indifferent Stage , 41 The Determining Stage, The Testes . 42 The Ovary 42 SUMMARI , 42 ACKNOWLEDGMENTS 46 LITERATURE CITED 47 APJENDEC 51 j INTRODUCTION AND REVIEW OF LITERATURE Nephrogenesis has been adequately described in only a few mammals, Buchanan and Fraser (1918), Fraser (1920), and MoCrady (1938) studied nephrogenesis in marsupials. Keibel (1903) reported on studies on Echidna Van der Strioht (1913) on the batj Torrey (1943) on the rat; and Bonnet (1888) and Davles and Davies (1950) on the sheep. -

Secreted Molecules in Metanephric Induction
J Am Soc Nephrol 11: S116–S119, 2000 Secreted Molecules in Metanephric Induction THOMAS J. CARROLL and ANDREW P. McMAHON Department of Molecular and Cellular Biology, Biological Laboratories, Harvard University, Cambridge, Massachusetts. Abstract. Nearly 50 yr ago, Clifford Grobstein made the ob- the classic model of metanephric induction. The studies of the servation that the ureteric bud induced the nephrogenic mes- classic ureteric inducer performed to date have most likely enchyme to undergo tubulogenesis. Since that discovery, sci- been characterizations of a mesenchyme-specific inducer, entists have attempted to characterize the molecular nature of Wnt-4, and its role in tubulogenesis. Ureteric induction most the inducer. To date, no single molecule that is both necessary likely involves a series of distinct events that provide prolif- and sufficient for nephric induction has been identified. Be- erative, survival, and condensation signals to the mesenchyme, cause of recent insights regarding the role of several secreted integrating the growth of the ureteric system with tubulogen- molecules in tubulogenesis, it has become necessary to revise esis. The developmental biologic processes of the kidney have been logenesis. The conclusion drawn from this discovery was that the subject of intense study for more than 100 yr (for review, the ureteric bud induces tubulogenesis within the surrounding see reference (1). All three vertebrate kidney types (pro- mesenchyme. During further investigation, it was discovered nephros, mesonephros, and metanephros) are derivatives of a that a number of tissues, including, most notably, a dorsal region of the embryo known as the intermediate mesoderm. In portion of the embryonic spinal cord, are able to substitute for mice, a portion of the mesonephric duct, known as the meta- the ureter in this inductive interaction. -

Glomus Specification and Induction in Xenopus
Development 126, 5847-5856 (1999) 5847 Printed in Great Britain © The Company of Biologists Limited 1999 DEV6378 The specification and growth factor inducibility of the pronephric glomus in Xenopus laevis Hannah C. Brennan, Sarbjit Nijjar and Elizabeth A. Jones* Cell and Molecular Development Group, Department of Biological Sciences, Warwick University, Coventry, CV4 7AL, UK *Author for correspondence (e-mail: [email protected]) Accepted 23 September; published on WWW 24 November 1999 SUMMARY We report a study on the specification of the glomus, the Furthermore, we have analysed the growth factor filtration device of the amphibian pronephric kidney, using inducibility of the glomus in the presence or absence of an explant culturing strategy in Xenopus laevis. Explants of retinoic acid (RA) by RT-PCR. We define for the first time presumptive pronephric mesoderm were dissected from the conditions under which these growth factors induce embryos of mid-gastrula to swimming tadpole stages. These glomus tissue in animal cap tissue. Activin together with explants were cultured within ectodermal wraps and high concentrations of RA can induce glomus tissue from analysed by RT-PCR for the presence of the Wilm’s Tumour- animal cap ectoderm. Unlike the pronephric tubules, the 1 gene, xWT1, a marker specific for the glomus at the stages glomus can also be induced by FGF and RA. analysed, together with other mesodermal markers. We show that the glomus is specified at stage 12.5, the same stage at which pronephric tubules are specified. We have previously shown that pronephric duct is specified somewhat later, at stage 14. -

Urinary System Intermediate Mesoderm
Urinary System Intermediate mesoderm lateral mesoderm: somite ectoderm neural NOTE: Intermediate mesoderm splanchnic groove somatic is situated between somites and lateral mesoderm (somatic and splanchnic mesoderm bordering the coelom). All mesoderm is derived from the primary mesen- intermediate mesoderm endoderm chyme that migrated through the notochord coelom (becomes urogenital ridge) primitive streak. Intermediate mesoderm (plus adjacent mesothelium lining the coelom) forms a urogenital ridge, which consists of a laterally-positioned nephrogenic cord (that forms kidneys & ureter) and a medially-positioned gonadal ridge (for ovary/testis & female/male genital tract formation). Thus urinary & genital systems have a common embryonic origin; also, they share common ducts. NOTE: Urine production essentially requires an increased capillary surface area (glomeruli), epithelial tubules to collect plasma filtrate and extract desirable constituents, and a duct system to convey urine away from the body. Kidneys Bilateraly, three kid- mesonephric duct neys develop from the neph- metanephros pronephros rogenic cord. They develop mesonephric tubules chronologically in cranial- mesonephros caudal sequence, and are designated pro—, meso—, Nephrogenic Cord (left) and meta—, respectively. cloaca The pronephros and mesonephros develop similarly: the nephrogenic cord undergoes seg- mentation, segments become tubules, tubules drain into a duct & eventually tubules disintegrate. spinal ganglion 1] Pronephros—consists of (7-8) primitive tubules and a pronephric duct that grows caudally and terminates in the cloaca. The tubules soon degenerate, but the pronephric duct persists as the neural tube mesonephric duct. (The pronephros is not functional, somite except in sheep.) notochord mesonephric NOTE tubule The mesonephros is the functional kidney for fish and am- aorta phibians. The metanephros is the functional kidney body of reptiles, birds, & mammals. -

Embryology of the Kidney Rizaldy Paz Scott | Yoshiro Maezawa | Jordan Kreidberg | Susan E
1 Embryology of the Kidney Rizaldy Paz Scott | Yoshiro Maezawa | Jordan Kreidberg | Susan E. Quaggin CHAPTER OUTLINE MAMMALIAN KIDNEY DEVELOPMENT, 2 MOLECULAR GENETICS OF MODEL SYSTEMS TO STUDY KIDNEY NEPHROGENESIS, 22 DEVELOPMENT, 8 GENETIC ANALYSIS OF MAMMALIAN KIDNEY DEVELOPMENT, 15 KEY POINTS • The development of the kidney relies on reciprocal signaling and inductive interactions between neighboring cells. • Epithelial cells that comprise the tubular structures of the kidney are derived from two distinct cell lineages: the ureteric epithelia lineage that branches and gives rise to collecting ducts and the nephrogenic mesenchyme lineage that undergoes mesenchyme to epithelial transition to form connecting tubules, distal tubules, the loop of Henle, proximal tubules, parietal epithelial cells, and podocytes. • Nephrogenesis and nephron endowment requires an epigenetically regulated balance between nephron progenitor self-renewal and epithelial differentiation. • The timing of incorporation of nephron progenitor cells into nascent nephrons predicts their positional identity within the highly patterned mature nephron. • Stromal cells and their derivatives coregulate ureteric branching morphogenesis, nephrogenesis, and vascular development. • Endothelial cells track the development of the ureteric epithelia and establish the renal vasculature through a combination of vasculogenic and angiogenic processes. • Collecting duct epithelia have an inherent plasticity enabling them to switch between principal and intercalated cell identities. MAMMALIAN KIDNEY DEVELOPMENT The filtration function of the kidneys is accomplished by basic units called nephrons (Fig. 1.1). Humans on average have 1 million nephrons per adult kidney but the range of ANATOMIC OVERVIEW OF THE 4 MAMMALIAN KIDNEY total nephrons is highly variable across human populations. Each mouse kidney may contain up to 12,000–16,000 nephrons The kidney is a sophisticated, highly vascularized organ that depending on the strain.5 This wide range in nephron number plays a central role in overall body homeostasis. -
Genital System • Anatomically, the Genital System Is Subdivided Into
Genital system • Anatomically, the genital system is subdivided into: 1- sex glands ( Testis and Ovary). 2- duct system ( male or female duct system) 3- external genitalia ( scrotum and penis in male, vulva in female) . • Embryollogically, from the time of fertilization the sex of the embryo can be determined genetically (male XY and female XX). • In early stage of development the sex of embryo cannot be differentiated anatomically or histologically, this stage is termed indifferent stage. • Later on ,sex hormones induce differentiation into male or female and in the same time the structure of the other sex degenerate. Indifferent Stage:- • In this stage the sex of the embryo cannot be differentiated anatomically or histologically. • This stage includes development of : - - gonads. - duct system - external genitalia. A- Gonads 1. Due to enlargement of mesonephros, it bulges ventrolaterally into the coelum forming a longitudinal urogenital ridge. 2. This ridge subdivided longitudinally into a lateral urinary ridge and medial genital ridge. 2. The genital (gonadal) ridge or gonad results from proliferation of celomatic epithelium. 3. This celomatic epithelium forms sex cord which project in the underlying mesenchyme . 4. The primordial germ cells which are located between the endodermal cells of the yolk sac, migrate through the mesentery of the hind gut to be located in the sex cord of the gonads. 5. Therefore the gonad has three types of cells: -Primordial germ cells. -celomatic cells -mesenchymal cells. B- Duct system • Both male and female embryos have initially two pairs of genital ducts: Mesonephric and Paramesonephric. 1. Mesonephric (Wolffian) duct: • The Mesonephric duct and tubules remain after degeneration of mesonephros forming the male duct system. -

The Developing Renal, Reproductive, and Respiratory Systems of the African Elephant Suggest an Aquatic Ancestry
Proc. Natl. Acad. Sci. USA Vol. 96, pp. 5555–5558, May 1999 Developmental Biology, Evolution The developing renal, reproductive, and respiratory systems of the African elephant suggest an aquatic ancestry A. P. GAETH*†,R.V.SHORT‡, AND M. B. RENFREE* *Department of Zoology, University of Melbourne, Parkville, Victoria 3052, Australia; and ‡Department of Perinatal Medicine, University of Melbourne, The Royal Women’s Hospital, Carlton, Victoria 3053, Australia Communicated by Judith Kimble, University of Wisconsin, Madison, WI, March 18, 1999 (received for review December 28, 1998) ABSTRACT The early embryology of the elephant has never of the testes, and the precocious development of the trunk could been studied before. We have obtained a rare series of African all have been adaptations to an aquatic lifestyle. elephant (Loxodonta africana) embryos and fetuses ranging in weight from 0.04 to 18.5 g, estimated gestational ages 58–166 MATERIALS AND METHODS days (duration of gestation is '660 days). Nephrostomes, a We have been able to obtain some rare African elephant spec- feature of aquatic vertebrates, were found in the mesonephric imens consisting of one embryo (weight 0.04 g) and six fetuses kidneys at all stages of development whereas they have never (weight ranging from 0.79 to 18.5 g) that were collected from been recorded in the mesonephric kidneys of other viviparous adult females shot in the Kruger National Park, South Africa, mammals. The trunk was well developed even in the earliest between 1993 and 1995, as part of a culling operation to reduce fetus. The testes were intra-abdominal, and there was no evidence elephant numbers in the park. -
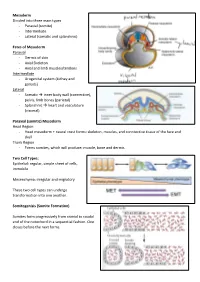
Mesoderm Divided Into Three Main Types - Paraxial (Somite) - Intermediate - Lateral (Somatic and Splanchnic)
Mesoderm Divided into three main types - Paraxial (somite) - Intermediate - Lateral (somatic and splanchnic) Fates of Mesoderm Paraxial - Dermis of skin - Axial Skeleton - Axial and limb muscles/tendons Intermediate - Urogenital system (kidney and gonads) Lateral - Somatic inner body wall (connective), pelvis, limb bones (parietal) - Splanchnic heart and vasculature (visceral) Paraxial (somitic) Mesoderm Head Region - Head mesoderm + neural crest forms: skeleton, muscles, and conntective tissue of the face and skull Trunk Region - Forms somites, which will produce: muscle, bone and dermis Two Cell Types: Epithelial: regular, simple sheet of cells, immobile Mesenchyma: irregular and migratory These two cell types can undergo transformation into one another. Somitogenisis (Somite Formation) Somites form progressively from cranial to caudal end of the notochord in a sequential fashion. One closes before the next forms. Somite Differentiation The somite splits into the epithelial dermamyotome (dermis/muscle) and the messenchymal sclerotome (skeletal). The somite is all paraxial mesoderm. Somite location determines the fate of its associates derma/myo/sclerotomes. Intermediate Mesoderm Urogenital system: - Kidneys - Gonads - Reproductive Duct Systems Runs alongside the paraxial mesoderm. Urogenital System Along mesonephric duct: - Pronephros, mesonephros, and metanephros - Pronephros fall away as gonad develops on ventral-medial side of mesonephros. - Metanephrogenic mesenchyme gives rise to kidney. The mesonephric duct will become the Wolffian duct forming at the nephric bud. The Mullerian duct forms via an invagination on the dorsal side of the nephric duct. The gonad will degenerate one of the two ducts depending on the hormones it produces. XX degenerates Wolffian duct – no testosterone, anti-Mullerian hormone (AMH) not produced, and Mullerian duct can develop in addition to female reproductive organs (ovaries, vagina) XY degenerates Mullerian duct – testosterone, AMH produced, Wolffian duct continues as male reproductive organs (testes, penis) develop. -

Renal Development
RENAL DEVELOPMENT Jon Barasch M.D., Ph.D. Telephone: 305-1890 e-mail: [email protected] SUGGESTED READING: Larsen, 3rd edition, pp 265 (first three paragraphs) - 266, 268-276 and figure 10-10 LEARNING OBJECTIVES: You should be able to: 1. Describe the three kidneys that are produced during development and know what happens to each one. 2. Explain what is meant by ‘reciprocal induction’ and why it poses problems in interpreting experiments in developing kidney. 3. Describe the stages of nephron formation from the renal vesicle. 4. Discuss the regulators of mesenchymal to epithelial transition in the intermediate mesoderm and metanephric mesenchyme and name three molecules mediating conversion. 5. Describe branching morphogenesis and name the three patterns in the developing metanephros. 6. Discuss three key important ligands and their receptors. 7. Discuss the classification of congenital renal abnormalities that are associated with urological abnormalities and the possible underlying mechanisms for their association. SUMMARY: The urogenital system derives from mesenchymal cells by a process of conversion to epithelia. The development of the kidney relies on three mechanisms of epithelial morphogenesis. 1. Some newborn epithelia migrate extensively (Wolffian duct), 2. some undergo branching morphogenesis (ureteric bud) and 3. some produce highly segmented tubules (nephrons). GLOSSARY: Angiotensin II: your favorite vasoconstrictor and regulator of proximal tubule reclamation of NaCl and water by receptor type 1. Receptor type-2 modulates cell growth and seems to play a role in congenital abnormalities. Arcade: a tubule of ureteric bud that induces a few nephrons simultaneously. The nephrons join to a common drainage called a connecting tubule that feeds into the ureteric’s collecting duct.